User login
Vulvar Inflammatory Dermatoses: New Approaches for Diagnosis and Treatment
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
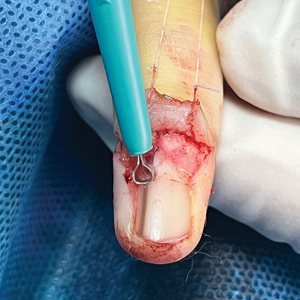
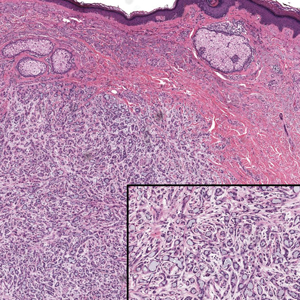
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
Optimizing Patient Care With Teledermatology: Improving Access, Efficiency, and Satisfaction
Telemedicine interest, which was relatively quiescent prior to the COVID-19 pandemic, has surged in popularity in the past few years.1 It can now be utilized seamlessly in dermatology practices to deliver exceptional patient care while reducing costs and travel time and offering dermatologists flexibility and improved work-life balance. Teledermatology applications include synchronous, asynchronous, and hybrid platforms.2 For synchronous teledermatology, patient visits are carried out in real time with audio and video technology.3 For asynchronous teledermatology—also known as the store-and-forward model—the dermatologist receives the patient’s history and photographs and then renders an assessment and treatment plan.2 Hybrid teledermatology uses real-time audio and video conferencing for history taking, assessment and treatment plan, and patient education, with photographs sent asynchronously.3 Telemedicine may not be initially intuitive or easy to integrate into clinical practice, but with time and effort, it will complement your dermatology practice, making it run more efficiently.
Patient Satisfaction With Teledermatology
Studies generally have shown very high patient satisfaction rates and shorter wait times with teledermatology vs in-person visits; for example, in a systematic review of 15 teledermatology studies including 7781 patients, more than 80% of participants reported high satisfaction with their telemedicine visit, with up to 92% reporting that they would choose to do a televisit again.4 In a retrospective analysis of 615 Zocdoc physicians, 65% of whom were dermatologists, mean wait times were 2.4 days for virtual appointments compared with 11.7 days for in-person appointments.5 Similarly, in a retrospective single-institution study, mean wait times for televisits were 14.3 days compared with 34.7 days for in-person referrals.6
Follow-Up Visits for Nail Disorders Via Teledermatology
Teledermatology may be particularly well suited for treating patients with nail disorders. In a prospective observational study, Onyeka et al7 accessed 813 images from 63 dermatology patients via teledermatology over a 6-month period to assess distance, focus, brightness, background, and image quality; of them, 83% were rated as high quality. Notably, images of nail disorders, skin growths, or pigmentation disorders were rated as having better image quality than images of inflammatory skin conditions (odds ratio [OR], 4.2-12.9 [P<.005]).7 In a retrospective study of 107 telemedicine visits for nail disorders during the COVID-19 pandemic, patients with longitudinal melanonychia were recommended for in-person visits for physical examination and dermoscopy, as were patients with suspected onychomycosis, who required nail plate sampling for diagnostic confirmation; however, approximately half of visits did not require in-person follow-up, including those patients with confirmed onychomycosis.8 Onychomycosis patients could be examined for clinical improvement and counseled on medication compliance via telemedicine. Other patients who did not require in-person follow-ups were those with traumatic nail disorders such as subungual hematoma and retronychia as well as those with body‐focused repetitive behaviors, including habit-tic nail deformity, onychophagia, and onychotillomania.8
Patients undergoing nail biopsies to rule out malignancies or to diagnose inflammatory nail disorders also may be managed via telemedicine. Patients for whom nail biopsies are recommended often are anxious about the procedure, which may be due to portrayal of nail trauma in the media9 or lack of accurate information on nail biopsies online.10 Therefore, counseling via telemedicine about the details of the procedure in a patient-friendly way (eg, showing an animated video and narrating it11) can allay anxiety without the inconvenience, cost, and time missed from work associated with traveling to an in-person visit. In addition, postoperative counseling ideally is performed via telemedicine because complications following nail procedures are uncommon. In a retrospective study of 502 patients who underwent a nail biopsy at a single academic center, only 14 developed surgical site infections within 8 days on average (range, 5–13 days), with a higher infection risk in patients with type 2 diabetes mellitus (P<.0003).12
Advantages and Limitations
There are many benefits to incorporating telemedicine into dermatology practices, including reduced overhead costs, convenience and time saved for patients, and flexibility and improved work-life balance for dermatologists. In addition, because the number of in-person visits seen generally is fixed due to space constraints and work-hour restrictions, delegating follow-up visits to telemedicine can free up in-person slots for new patients and those needing procedures. However, there also are some inherent limitations to telemedicine: technology access, vision or hearing difficulties or low digital health literacy, or language barriers. In the prospective observational study by Onyeka et al7 analyzing 813 teledermatology images, patients aged 65 to 74 years sent in more clinically useful images (OR, 7.9) and images that were more often in focus (OR, 2.6) compared with patients older than 85 years.
Final Thoughts
Incorporation of telemedicine into dermatologic practice is a valuable tool for triaging patients with acute issues, improving patient care and health care access, making practices more efficient, and improving dermatologist flexibility and work-life balance. Further development of teledermatology to provide access to underserved populations prioritizing dermatologist reimbursement and progress on technologic innovations will make teledermatology even more useful in the coming years.
- He A, Ti Kim T, Nguyen KD. Utilization of teledermatology services for dermatological diagnoses during the COVID-19 pandemic. Arch Dermatol Res. 2023;315:1059-1062.
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260.
- Wang RH, Barbieri JS, Kovarik CL, et al. Synchronous and asynchronous teledermatology: a narrative review of strengths and limitations. J Telemed Telecare. 2022;28:533-538.
- Miller J, Jones E. Shaping the future of teledermatology: a literature review of patient and provider satisfaction with synchronous teledermatology during the COVID-19 pandemic. Clin Exp Dermatol. 2022;47:1903-1909.
- Gu L, Xiang L, Lipner SR. Analysis of availability of online dermatology appointments during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:517-520.
- Wang RF, Trinidad J, Lawrence J, et al. Improved patient access and outcomes with the integration of an eConsult program (teledermatology) within a large academic medical center. J Am Acad Dermatol. 2019;83:1633-1638.
- Onyeka S, Kim J, Eid E, et al. Quality of images submitted by older patients to a teledermatology platform. Abstract presented at the Society of Investigative Dermatology Annual Meeting; May 15-18, 2024; Dallas, TX.
- Chang MJ, Stewart CR, Lipner SR. Retrospective study of nail telemedicine visits during the COVID-19 pandemic. Dermatol Ther. 2021;34:E14630.
- Albucker SJ, Falotico JM, Lipner SR. A real nail biter: a cross-sectional study of 75 nail trauma scenes in international films and television series. J Cutan Med Surg. 2023;27:288-291.
- Ishack S, Lipner SR. Evaluating the impact and educational value of YouTube videos on nail biopsy procedures. Cutis. 2020;105:148-149, E1.
- Hill RC, Ho B, Lipner SR. Assuaging patient anxiety about nail biopsies with an animated educational video. J Am Acad Dermatol. Published online March 29, 2024. doi:10.1016/j.jaad.2024.03.031.
- Axler E, Lu A, Darrell M, et al. Surgical site infections are uncommon following nail biopsies in a single-center case-control study of 502 patients. J Am Acad Dermatol. Published online May 15, 2024. doi:10.1016/j.jaad.2024.05.017
Telemedicine interest, which was relatively quiescent prior to the COVID-19 pandemic, has surged in popularity in the past few years.1 It can now be utilized seamlessly in dermatology practices to deliver exceptional patient care while reducing costs and travel time and offering dermatologists flexibility and improved work-life balance. Teledermatology applications include synchronous, asynchronous, and hybrid platforms.2 For synchronous teledermatology, patient visits are carried out in real time with audio and video technology.3 For asynchronous teledermatology—also known as the store-and-forward model—the dermatologist receives the patient’s history and photographs and then renders an assessment and treatment plan.2 Hybrid teledermatology uses real-time audio and video conferencing for history taking, assessment and treatment plan, and patient education, with photographs sent asynchronously.3 Telemedicine may not be initially intuitive or easy to integrate into clinical practice, but with time and effort, it will complement your dermatology practice, making it run more efficiently.
Patient Satisfaction With Teledermatology
Studies generally have shown very high patient satisfaction rates and shorter wait times with teledermatology vs in-person visits; for example, in a systematic review of 15 teledermatology studies including 7781 patients, more than 80% of participants reported high satisfaction with their telemedicine visit, with up to 92% reporting that they would choose to do a televisit again.4 In a retrospective analysis of 615 Zocdoc physicians, 65% of whom were dermatologists, mean wait times were 2.4 days for virtual appointments compared with 11.7 days for in-person appointments.5 Similarly, in a retrospective single-institution study, mean wait times for televisits were 14.3 days compared with 34.7 days for in-person referrals.6
Follow-Up Visits for Nail Disorders Via Teledermatology
Teledermatology may be particularly well suited for treating patients with nail disorders. In a prospective observational study, Onyeka et al7 accessed 813 images from 63 dermatology patients via teledermatology over a 6-month period to assess distance, focus, brightness, background, and image quality; of them, 83% were rated as high quality. Notably, images of nail disorders, skin growths, or pigmentation disorders were rated as having better image quality than images of inflammatory skin conditions (odds ratio [OR], 4.2-12.9 [P<.005]).7 In a retrospective study of 107 telemedicine visits for nail disorders during the COVID-19 pandemic, patients with longitudinal melanonychia were recommended for in-person visits for physical examination and dermoscopy, as were patients with suspected onychomycosis, who required nail plate sampling for diagnostic confirmation; however, approximately half of visits did not require in-person follow-up, including those patients with confirmed onychomycosis.8 Onychomycosis patients could be examined for clinical improvement and counseled on medication compliance via telemedicine. Other patients who did not require in-person follow-ups were those with traumatic nail disorders such as subungual hematoma and retronychia as well as those with body‐focused repetitive behaviors, including habit-tic nail deformity, onychophagia, and onychotillomania.8
Patients undergoing nail biopsies to rule out malignancies or to diagnose inflammatory nail disorders also may be managed via telemedicine. Patients for whom nail biopsies are recommended often are anxious about the procedure, which may be due to portrayal of nail trauma in the media9 or lack of accurate information on nail biopsies online.10 Therefore, counseling via telemedicine about the details of the procedure in a patient-friendly way (eg, showing an animated video and narrating it11) can allay anxiety without the inconvenience, cost, and time missed from work associated with traveling to an in-person visit. In addition, postoperative counseling ideally is performed via telemedicine because complications following nail procedures are uncommon. In a retrospective study of 502 patients who underwent a nail biopsy at a single academic center, only 14 developed surgical site infections within 8 days on average (range, 5–13 days), with a higher infection risk in patients with type 2 diabetes mellitus (P<.0003).12
Advantages and Limitations
There are many benefits to incorporating telemedicine into dermatology practices, including reduced overhead costs, convenience and time saved for patients, and flexibility and improved work-life balance for dermatologists. In addition, because the number of in-person visits seen generally is fixed due to space constraints and work-hour restrictions, delegating follow-up visits to telemedicine can free up in-person slots for new patients and those needing procedures. However, there also are some inherent limitations to telemedicine: technology access, vision or hearing difficulties or low digital health literacy, or language barriers. In the prospective observational study by Onyeka et al7 analyzing 813 teledermatology images, patients aged 65 to 74 years sent in more clinically useful images (OR, 7.9) and images that were more often in focus (OR, 2.6) compared with patients older than 85 years.
Final Thoughts
Incorporation of telemedicine into dermatologic practice is a valuable tool for triaging patients with acute issues, improving patient care and health care access, making practices more efficient, and improving dermatologist flexibility and work-life balance. Further development of teledermatology to provide access to underserved populations prioritizing dermatologist reimbursement and progress on technologic innovations will make teledermatology even more useful in the coming years.
Telemedicine interest, which was relatively quiescent prior to the COVID-19 pandemic, has surged in popularity in the past few years.1 It can now be utilized seamlessly in dermatology practices to deliver exceptional patient care while reducing costs and travel time and offering dermatologists flexibility and improved work-life balance. Teledermatology applications include synchronous, asynchronous, and hybrid platforms.2 For synchronous teledermatology, patient visits are carried out in real time with audio and video technology.3 For asynchronous teledermatology—also known as the store-and-forward model—the dermatologist receives the patient’s history and photographs and then renders an assessment and treatment plan.2 Hybrid teledermatology uses real-time audio and video conferencing for history taking, assessment and treatment plan, and patient education, with photographs sent asynchronously.3 Telemedicine may not be initially intuitive or easy to integrate into clinical practice, but with time and effort, it will complement your dermatology practice, making it run more efficiently.
Patient Satisfaction With Teledermatology
Studies generally have shown very high patient satisfaction rates and shorter wait times with teledermatology vs in-person visits; for example, in a systematic review of 15 teledermatology studies including 7781 patients, more than 80% of participants reported high satisfaction with their telemedicine visit, with up to 92% reporting that they would choose to do a televisit again.4 In a retrospective analysis of 615 Zocdoc physicians, 65% of whom were dermatologists, mean wait times were 2.4 days for virtual appointments compared with 11.7 days for in-person appointments.5 Similarly, in a retrospective single-institution study, mean wait times for televisits were 14.3 days compared with 34.7 days for in-person referrals.6
Follow-Up Visits for Nail Disorders Via Teledermatology
Teledermatology may be particularly well suited for treating patients with nail disorders. In a prospective observational study, Onyeka et al7 accessed 813 images from 63 dermatology patients via teledermatology over a 6-month period to assess distance, focus, brightness, background, and image quality; of them, 83% were rated as high quality. Notably, images of nail disorders, skin growths, or pigmentation disorders were rated as having better image quality than images of inflammatory skin conditions (odds ratio [OR], 4.2-12.9 [P<.005]).7 In a retrospective study of 107 telemedicine visits for nail disorders during the COVID-19 pandemic, patients with longitudinal melanonychia were recommended for in-person visits for physical examination and dermoscopy, as were patients with suspected onychomycosis, who required nail plate sampling for diagnostic confirmation; however, approximately half of visits did not require in-person follow-up, including those patients with confirmed onychomycosis.8 Onychomycosis patients could be examined for clinical improvement and counseled on medication compliance via telemedicine. Other patients who did not require in-person follow-ups were those with traumatic nail disorders such as subungual hematoma and retronychia as well as those with body‐focused repetitive behaviors, including habit-tic nail deformity, onychophagia, and onychotillomania.8
Patients undergoing nail biopsies to rule out malignancies or to diagnose inflammatory nail disorders also may be managed via telemedicine. Patients for whom nail biopsies are recommended often are anxious about the procedure, which may be due to portrayal of nail trauma in the media9 or lack of accurate information on nail biopsies online.10 Therefore, counseling via telemedicine about the details of the procedure in a patient-friendly way (eg, showing an animated video and narrating it11) can allay anxiety without the inconvenience, cost, and time missed from work associated with traveling to an in-person visit. In addition, postoperative counseling ideally is performed via telemedicine because complications following nail procedures are uncommon. In a retrospective study of 502 patients who underwent a nail biopsy at a single academic center, only 14 developed surgical site infections within 8 days on average (range, 5–13 days), with a higher infection risk in patients with type 2 diabetes mellitus (P<.0003).12
Advantages and Limitations
There are many benefits to incorporating telemedicine into dermatology practices, including reduced overhead costs, convenience and time saved for patients, and flexibility and improved work-life balance for dermatologists. In addition, because the number of in-person visits seen generally is fixed due to space constraints and work-hour restrictions, delegating follow-up visits to telemedicine can free up in-person slots for new patients and those needing procedures. However, there also are some inherent limitations to telemedicine: technology access, vision or hearing difficulties or low digital health literacy, or language barriers. In the prospective observational study by Onyeka et al7 analyzing 813 teledermatology images, patients aged 65 to 74 years sent in more clinically useful images (OR, 7.9) and images that were more often in focus (OR, 2.6) compared with patients older than 85 years.
Final Thoughts
Incorporation of telemedicine into dermatologic practice is a valuable tool for triaging patients with acute issues, improving patient care and health care access, making practices more efficient, and improving dermatologist flexibility and work-life balance. Further development of teledermatology to provide access to underserved populations prioritizing dermatologist reimbursement and progress on technologic innovations will make teledermatology even more useful in the coming years.
- He A, Ti Kim T, Nguyen KD. Utilization of teledermatology services for dermatological diagnoses during the COVID-19 pandemic. Arch Dermatol Res. 2023;315:1059-1062.
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260.
- Wang RH, Barbieri JS, Kovarik CL, et al. Synchronous and asynchronous teledermatology: a narrative review of strengths and limitations. J Telemed Telecare. 2022;28:533-538.
- Miller J, Jones E. Shaping the future of teledermatology: a literature review of patient and provider satisfaction with synchronous teledermatology during the COVID-19 pandemic. Clin Exp Dermatol. 2022;47:1903-1909.
- Gu L, Xiang L, Lipner SR. Analysis of availability of online dermatology appointments during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:517-520.
- Wang RF, Trinidad J, Lawrence J, et al. Improved patient access and outcomes with the integration of an eConsult program (teledermatology) within a large academic medical center. J Am Acad Dermatol. 2019;83:1633-1638.
- Onyeka S, Kim J, Eid E, et al. Quality of images submitted by older patients to a teledermatology platform. Abstract presented at the Society of Investigative Dermatology Annual Meeting; May 15-18, 2024; Dallas, TX.
- Chang MJ, Stewart CR, Lipner SR. Retrospective study of nail telemedicine visits during the COVID-19 pandemic. Dermatol Ther. 2021;34:E14630.
- Albucker SJ, Falotico JM, Lipner SR. A real nail biter: a cross-sectional study of 75 nail trauma scenes in international films and television series. J Cutan Med Surg. 2023;27:288-291.
- Ishack S, Lipner SR. Evaluating the impact and educational value of YouTube videos on nail biopsy procedures. Cutis. 2020;105:148-149, E1.
- Hill RC, Ho B, Lipner SR. Assuaging patient anxiety about nail biopsies with an animated educational video. J Am Acad Dermatol. Published online March 29, 2024. doi:10.1016/j.jaad.2024.03.031.
- Axler E, Lu A, Darrell M, et al. Surgical site infections are uncommon following nail biopsies in a single-center case-control study of 502 patients. J Am Acad Dermatol. Published online May 15, 2024. doi:10.1016/j.jaad.2024.05.017
- He A, Ti Kim T, Nguyen KD. Utilization of teledermatology services for dermatological diagnoses during the COVID-19 pandemic. Arch Dermatol Res. 2023;315:1059-1062.
- Lee JJ, English JC 3rd. Teledermatology: a review and update. Am J Clin Dermatol. 2018;19:253-260.
- Wang RH, Barbieri JS, Kovarik CL, et al. Synchronous and asynchronous teledermatology: a narrative review of strengths and limitations. J Telemed Telecare. 2022;28:533-538.
- Miller J, Jones E. Shaping the future of teledermatology: a literature review of patient and provider satisfaction with synchronous teledermatology during the COVID-19 pandemic. Clin Exp Dermatol. 2022;47:1903-1909.
- Gu L, Xiang L, Lipner SR. Analysis of availability of online dermatology appointments during the COVID-19 pandemic. J Am Acad Dermatol. 2021;84:517-520.
- Wang RF, Trinidad J, Lawrence J, et al. Improved patient access and outcomes with the integration of an eConsult program (teledermatology) within a large academic medical center. J Am Acad Dermatol. 2019;83:1633-1638.
- Onyeka S, Kim J, Eid E, et al. Quality of images submitted by older patients to a teledermatology platform. Abstract presented at the Society of Investigative Dermatology Annual Meeting; May 15-18, 2024; Dallas, TX.
- Chang MJ, Stewart CR, Lipner SR. Retrospective study of nail telemedicine visits during the COVID-19 pandemic. Dermatol Ther. 2021;34:E14630.
- Albucker SJ, Falotico JM, Lipner SR. A real nail biter: a cross-sectional study of 75 nail trauma scenes in international films and television series. J Cutan Med Surg. 2023;27:288-291.
- Ishack S, Lipner SR. Evaluating the impact and educational value of YouTube videos on nail biopsy procedures. Cutis. 2020;105:148-149, E1.
- Hill RC, Ho B, Lipner SR. Assuaging patient anxiety about nail biopsies with an animated educational video. J Am Acad Dermatol. Published online March 29, 2024. doi:10.1016/j.jaad.2024.03.031.
- Axler E, Lu A, Darrell M, et al. Surgical site infections are uncommon following nail biopsies in a single-center case-control study of 502 patients. J Am Acad Dermatol. Published online May 15, 2024. doi:10.1016/j.jaad.2024.05.017
Practice Points
- Incorporation of telemedicine into dermatologic practice can improve patient access, reduce costs, and offer dermatologists flexibility and improved work-life balance.
- Patient satisfaction with telemedicine is exceedingly high, and teledermatology may be particularly well suited for caring for patients with nail disorders.
Customized Dermal Curette: An Alternative and Effective Shaving Tool in Nail Surgery
Practice Gap
Longitudinal melanonychia (LM) is characterized by the presence of a dark brown, longitudinal, pigmented band on the nail unit, often caused by melanocytic activation or melanocytic hyperplasia in the nail matrix. Distinguishing between benign and early malignant LM is crucial due to their similar clinical presentations.1 Hence, surgical excision of the pigmented nail matrix followed by histopathologic examination is a common procedure aimed at managing LM and reducing the risk for delayed diagnosis of subungual melanoma.
Tangential matrix excision combined with the nail window technique has emerged as a common and favored surgical strategy for managing LM.2 This method is highly valued for its ability to minimize the risk for severe permanent nail dystrophy and effectively reduce postsurgical pigmentation recurrence.
The procedure begins with the creation of a matrix window along the lateral edge of the pigmented band followed by 1 lateral incision carefully made on each side of the nail fold. This meticulous approach allows for the complete exposure of the pigmented lesion. Subsequently, the nail fold is separated from the dorsal surface of the nail plate to facilitate access to the pigmented nail matrix. Finally, the target pigmented area is excised using a scalpel.
Despite the recognized efficacy of this procedure, challenges do arise, particularly when the width of the pigmented matrix lesion is narrow. Holding the scalpel horizontally to ensure precise excision can prove to be demanding, leading to difficulty achieving complete lesion removal and obtaining the desired cosmetic outcomes. As such, there is a clear need to explore alternative tools that can effectively address these challenges while ensuring optimal surgical outcomes for patients with LM. We propose the use of the customized dermal curette.
The Technique
An improved curette tool is a practical solution for complete removal of the pigmented nail matrix. This enhanced instrument is crafted from a sterile disposable dermal curette with its top flattened using a needle holder(Figure 1). Termed the customized dermal curette, this device is a simple yet accurate tool for the precise excision of pigmented lesions within the nail matrix. Importantly, it offers versatility by accommodating different widths of pigmented lesions through the availability of various sizes of dermal curettes (Figure 2).

Histopathologically, we have found that the scalpel technique may lead to variable tissue removal, resulting in differences in tissue thickness, fragility, and completeness (Figure 3A). Conversely, the customized dermal curette consistently provides more accurate tissue excision, resulting in uniform tissue thickness and integrity (Figure 3B).
Practice Implications
Compared to the traditional scalpel, this modified tool offers distinct advantages. Specifically, the customized dermal curette provides enhanced maneuverability and control during the procedure, thereby improving the overall efficacy of the excision process. It also offers a more accurate approach to completely remove pigmented bands, which reduces the risk for postoperative recurrence. The simplicity, affordability, and ease of operation associated with customized dermal curettes holds promise as an effective alternative for tissue shaving, especially in cases involving narrow pigmented matrix lesions, thereby addressing a notable practice gap and enhancing patient care.
- Tan WC, Wang DY, Seghers AC, et al. Should we biopsy melanonychia striata in Asian children? a retrospective observational study. Pediatr Dermatol. 2019;36:864-868. doi:10.1111/pde.13934
- Zhou Y, Chen W, Liu ZR, et al. Modified shave surgery combined with nail window technique for the treatment of longitudinal melanonychia: evaluation of the method on a series of 67 cases. J Am Acad Dermatol. 2019;81:717-722. doi:10.1016/j.jaad.2019.03.065
Practice Gap
Longitudinal melanonychia (LM) is characterized by the presence of a dark brown, longitudinal, pigmented band on the nail unit, often caused by melanocytic activation or melanocytic hyperplasia in the nail matrix. Distinguishing between benign and early malignant LM is crucial due to their similar clinical presentations.1 Hence, surgical excision of the pigmented nail matrix followed by histopathologic examination is a common procedure aimed at managing LM and reducing the risk for delayed diagnosis of subungual melanoma.
Tangential matrix excision combined with the nail window technique has emerged as a common and favored surgical strategy for managing LM.2 This method is highly valued for its ability to minimize the risk for severe permanent nail dystrophy and effectively reduce postsurgical pigmentation recurrence.
The procedure begins with the creation of a matrix window along the lateral edge of the pigmented band followed by 1 lateral incision carefully made on each side of the nail fold. This meticulous approach allows for the complete exposure of the pigmented lesion. Subsequently, the nail fold is separated from the dorsal surface of the nail plate to facilitate access to the pigmented nail matrix. Finally, the target pigmented area is excised using a scalpel.
Despite the recognized efficacy of this procedure, challenges do arise, particularly when the width of the pigmented matrix lesion is narrow. Holding the scalpel horizontally to ensure precise excision can prove to be demanding, leading to difficulty achieving complete lesion removal and obtaining the desired cosmetic outcomes. As such, there is a clear need to explore alternative tools that can effectively address these challenges while ensuring optimal surgical outcomes for patients with LM. We propose the use of the customized dermal curette.
The Technique
An improved curette tool is a practical solution for complete removal of the pigmented nail matrix. This enhanced instrument is crafted from a sterile disposable dermal curette with its top flattened using a needle holder(Figure 1). Termed the customized dermal curette, this device is a simple yet accurate tool for the precise excision of pigmented lesions within the nail matrix. Importantly, it offers versatility by accommodating different widths of pigmented lesions through the availability of various sizes of dermal curettes (Figure 2).


Histopathologically, we have found that the scalpel technique may lead to variable tissue removal, resulting in differences in tissue thickness, fragility, and completeness (Figure 3A). Conversely, the customized dermal curette consistently provides more accurate tissue excision, resulting in uniform tissue thickness and integrity (Figure 3B).

Practice Implications
Compared to the traditional scalpel, this modified tool offers distinct advantages. Specifically, the customized dermal curette provides enhanced maneuverability and control during the procedure, thereby improving the overall efficacy of the excision process. It also offers a more accurate approach to completely remove pigmented bands, which reduces the risk for postoperative recurrence. The simplicity, affordability, and ease of operation associated with customized dermal curettes holds promise as an effective alternative for tissue shaving, especially in cases involving narrow pigmented matrix lesions, thereby addressing a notable practice gap and enhancing patient care.
Practice Gap
Longitudinal melanonychia (LM) is characterized by the presence of a dark brown, longitudinal, pigmented band on the nail unit, often caused by melanocytic activation or melanocytic hyperplasia in the nail matrix. Distinguishing between benign and early malignant LM is crucial due to their similar clinical presentations.1 Hence, surgical excision of the pigmented nail matrix followed by histopathologic examination is a common procedure aimed at managing LM and reducing the risk for delayed diagnosis of subungual melanoma.
Tangential matrix excision combined with the nail window technique has emerged as a common and favored surgical strategy for managing LM.2 This method is highly valued for its ability to minimize the risk for severe permanent nail dystrophy and effectively reduce postsurgical pigmentation recurrence.
The procedure begins with the creation of a matrix window along the lateral edge of the pigmented band followed by 1 lateral incision carefully made on each side of the nail fold. This meticulous approach allows for the complete exposure of the pigmented lesion. Subsequently, the nail fold is separated from the dorsal surface of the nail plate to facilitate access to the pigmented nail matrix. Finally, the target pigmented area is excised using a scalpel.
Despite the recognized efficacy of this procedure, challenges do arise, particularly when the width of the pigmented matrix lesion is narrow. Holding the scalpel horizontally to ensure precise excision can prove to be demanding, leading to difficulty achieving complete lesion removal and obtaining the desired cosmetic outcomes. As such, there is a clear need to explore alternative tools that can effectively address these challenges while ensuring optimal surgical outcomes for patients with LM. We propose the use of the customized dermal curette.
The Technique
An improved curette tool is a practical solution for complete removal of the pigmented nail matrix. This enhanced instrument is crafted from a sterile disposable dermal curette with its top flattened using a needle holder(Figure 1). Termed the customized dermal curette, this device is a simple yet accurate tool for the precise excision of pigmented lesions within the nail matrix. Importantly, it offers versatility by accommodating different widths of pigmented lesions through the availability of various sizes of dermal curettes (Figure 2).


Histopathologically, we have found that the scalpel technique may lead to variable tissue removal, resulting in differences in tissue thickness, fragility, and completeness (Figure 3A). Conversely, the customized dermal curette consistently provides more accurate tissue excision, resulting in uniform tissue thickness and integrity (Figure 3B).

Practice Implications
Compared to the traditional scalpel, this modified tool offers distinct advantages. Specifically, the customized dermal curette provides enhanced maneuverability and control during the procedure, thereby improving the overall efficacy of the excision process. It also offers a more accurate approach to completely remove pigmented bands, which reduces the risk for postoperative recurrence. The simplicity, affordability, and ease of operation associated with customized dermal curettes holds promise as an effective alternative for tissue shaving, especially in cases involving narrow pigmented matrix lesions, thereby addressing a notable practice gap and enhancing patient care.
- Tan WC, Wang DY, Seghers AC, et al. Should we biopsy melanonychia striata in Asian children? a retrospective observational study. Pediatr Dermatol. 2019;36:864-868. doi:10.1111/pde.13934
- Zhou Y, Chen W, Liu ZR, et al. Modified shave surgery combined with nail window technique for the treatment of longitudinal melanonychia: evaluation of the method on a series of 67 cases. J Am Acad Dermatol. 2019;81:717-722. doi:10.1016/j.jaad.2019.03.065
- Tan WC, Wang DY, Seghers AC, et al. Should we biopsy melanonychia striata in Asian children? a retrospective observational study. Pediatr Dermatol. 2019;36:864-868. doi:10.1111/pde.13934
- Zhou Y, Chen W, Liu ZR, et al. Modified shave surgery combined with nail window technique for the treatment of longitudinal melanonychia: evaluation of the method on a series of 67 cases. J Am Acad Dermatol. 2019;81:717-722. doi:10.1016/j.jaad.2019.03.065
Slowly Enlarging Nodule on the Neck
The Diagnosis: Microsecretory Adenocarcinoma
Microscopically, the tumor was relatively well circumscribed but had irregular borders. It consisted of microcysts and tubules lined by flattened to plump eosinophilic cells with mildly enlarged nuclei and intraluminal basophilic secretions. Peripheral lymphocytic aggregates also were seen in the mid and deep reticular dermis. Tumor necrosis, lymphovascular invasion, and notable mitotic activity were absent. Immunohistochemistry was diffusely positive for cytokeratin (CK) 7 and CK5/6. Occasional tumor cells showed variable expression of alpha smooth muscle actin, S-100 protein, and p40 and p63 antibodies. Immunohistochemistry was negative for CK20; GATA binding protein 3; MYB proto-oncogene, transcription factor; and insulinoma-associated protein 1. A dual-color, break-apart fluorescence in situ hybridization probe identified a rearrangement of the SS18 (SYT) gene locus on chromosome 18. The nodule was excised with clear surgical margins, and the patient had no evidence of recurrent disease or metastasis at 2-year follow-up.
In recent years, there has been a growing recognition of the pivotal role played by gene fusions in driving oncogenesis, encompassing a diverse range of benign and malignant cutaneous neoplasms. These investigations have shed light on previously unknown mechanisms and pathways contributing to the pathogenesis of these neoplastic conditions, offering invaluable insights into their underlying biology. As a result, our ability to classify and diagnose these cutaneous tumors has improved. A notable example of how our current understanding has evolved is the discovery of the new cutaneous adnexal tumor microsecretory adenocarcinoma (MSA). Initially described by Bishop et al1 in 2019 as predominantly occurring in the intraoral minor salivary glands, rare instances of primary cutaneous MSA involving the head and neck regions also have been reported.2 Microsecretory adenocarcinoma represents an important addition to the group of fusion-driven tumors with both salivary gland and cutaneous adnexal analogues, characterized by a MEF2C::SS18 gene fusion. This entity is now recognized as a group of cutaneous adnexal tumors with distinct gene fusions, including both relatively recently discovered entities (eg, secretory carcinoma with NTRK fusions) and previously known entities with newly identified gene fusions (eg, poroid neoplasms with NUTM1, YAP1, or WWTR1 fusions; hidradenomatous neoplasms with CRTC1::MAML2 fusions; and adenoid cystic carcinoma with MYB, MYBL1, and/or NFIB rearrangements).3
Microsecretory adenocarcinoma exhibits a high degree of morphologic consistency, characterized by a microcystic-predominant growth pattern, uniform intercalated ductlike tumor cells with attenuated eosinophilic to clear cytoplasm, monotonous oval hyperchromatic nuclei with indistinct nucleoli, abundant basophilic luminal secretions, and a variably cellular fibromyxoid stroma. It also shows rounded borders with subtle infiltrative growth. Occasionally, pseudoepitheliomatous hyperplasia, tumor-associated lymphoid proliferation, or metaplastic bone formation may accompany MSA. Perineural invasion is rare, necrosis is absent, and mitotic rates generally are low, contributing to its distinctive histopathologic features that aid in accurate diagnosis and differentiation from other entities. Immunohistochemistry reveals diffuse positivity for CK7 and patchy to diffuse expression of S-100 in tumor cells as well as variable expression of p40 and p63. Highly specific SS18 gene translocations at chromosome 18q are useful for diagnosing MSA when found alongside its characteristic appearance, and SS18 break-apart fluorescence in situ hybridization can serve reliably as an accurate diagnostic method (Figure 1).4 Our case illustrates how molecular analysis assists in distinguishing MSA from other cutaneous adnexal tumors, exemplifying the power of our evolving understanding in refining diagnostic accuracy and guiding targeted therapies in clinical practice.
The differential diagnosis of MSA includes tubular adenoma, secretory carcinoma, cribriform tumor (previously carcinoma), and metastatic adenocarcinoma. Tubular adenoma is a rare benign neoplasm that predominantly affects females and can manifest at any age in adulthood. It typically manifests as a slow-growing, occasionally pedunculated nodule, often measuring less than 2 cm. Although it most commonly manifests on the scalp, tubular adenoma also may arise in diverse sites such as the face, axillae, lower extremities, or genitalia.
Notably, scalp lesions often are associated with nevus sebaceus of Jadassohn or syringocystadenoma papilliferum. Microscopically, tubular adenoma is well circumscribed within the dermis and may extend into the subcutis in some cases. Its distinctive appearance consists of variably sized tubules lined by a double or multilayered cuboidal to columnar epithelium, frequently displaying apocrine decapitation secretion (Figure 2). Cystic changes and intraluminal papillae devoid of true fibrovascular cores frequently are observed. Immunohistochemically, luminal epithelial cells express epithelial membrane antigen and carcinoembryonic antigen, while the myoepithelial layer expresses smooth muscle markers, p40, and S-100 protein. BRAF V600E mutation can be detected using immunohistochemistry, with excellent sensitivity and specificity using the anti-BRAF V600E antibody (clone VE1).5 Distinguishing tubular adenoma from MSA is achievable by observing its larger, more variable tubules, along with the consistent presence of a peripheral myoepithelial layer.
Secretory carcinoma is recognized as a low-grade gene fusion–driven carcinoma that primarily arises in salivary glands (both major and minor), with occasional occurrences in the breast and extremely rare instances in other locations such as the skin, thyroid gland, and lung.6 Although the axilla is the most common cutaneous site, diverse locations such as the neck, eyelids, extremities, and nipples also have been documented. Secretory carcinoma affects individuals across a wide age range (13–71 years).6 The hallmark tumors exhibit densely packed, sievelike microcystic glands and tubular spaces filled with abundant eosinophilic intraluminal secretions (Figure 3). Additionally, morphologic variants, such as predominantly papillary, papillary-cystic, macrocystic, solid, partially mucinous, and mixed-pattern neoplasms, have been described. Secretory carcinoma shares certain features with MSA; however, it is distinguished by the presence of pronounced eosinophilic secretions, plump and vacuolated cytoplasm, and a less conspicuous fibromyxoid stroma. Immunohistochemistry reveals tumor cells that are positive for CK7, SOX-10, S-100, mammaglobin, MUC4, and variably GATA-3. Genetically, secretory carcinoma exhibits distinct characteristics, commonly showing the ETV6::NTRK3 fusion, detectable through molecular techniques or pan-TRK immunohistochemistry, while RET fusions and other rare variants are less frequent.7
In 1998, Requena et al8 introduced the concept of primary cutaneous cribriform carcinoma. Despite initially being classified as a carcinoma, the malignant potential of this tumor remains uncertain. Consequently, the term cribriform tumor now has become the preferred terminology for denoting this rare entity.9 Primary cutaneous cribriform tumors are observed more commonly in women and typically affect individuals aged 20 to 55 years (mean, 44 years). Predominant locations include the upper and lower extremities, especially the thighs, knees, and legs, with additional cases occurring on the head and trunk. Microscopically, cribriform tumor is characterized by a partially circumscribed, unencapsulated dermal nodule composed of round or oval nuclei displaying hyperchromatism and mild pleomorphism. The defining aspect of its morphology revolves around interspersed small round cavities that give rise to the hallmark cribriform pattern (Figure 4). Although MSA occasionally may exhibit a cribriform architectural pattern, it typically lacks the distinctive feature of thin, threadlike, intraluminal bridging strands observed in cribriform tumors. Similarly, luminal cells within the cribriform tumor express CK7 and exhibit variable S-100 expression. It is recognized as an indolent neoplasm with uncertain malignant potential.
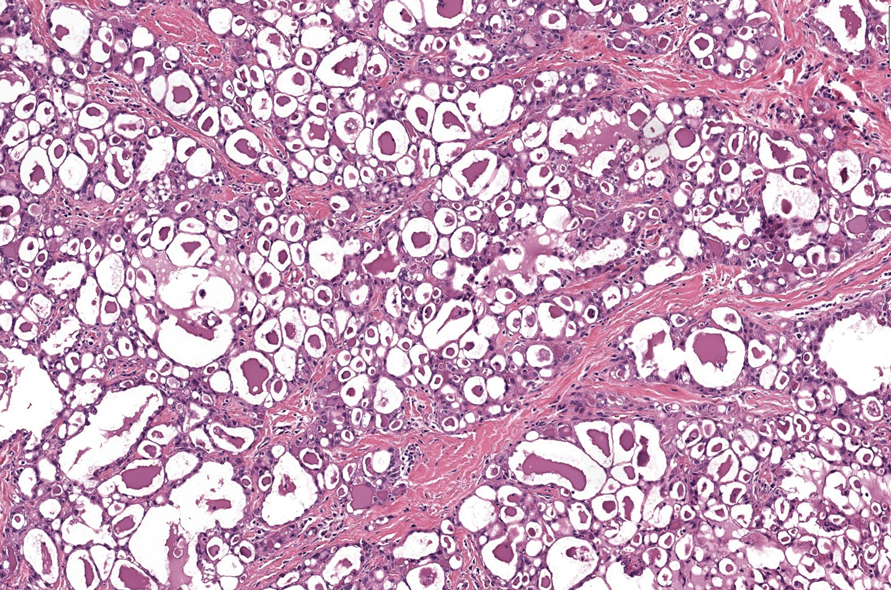
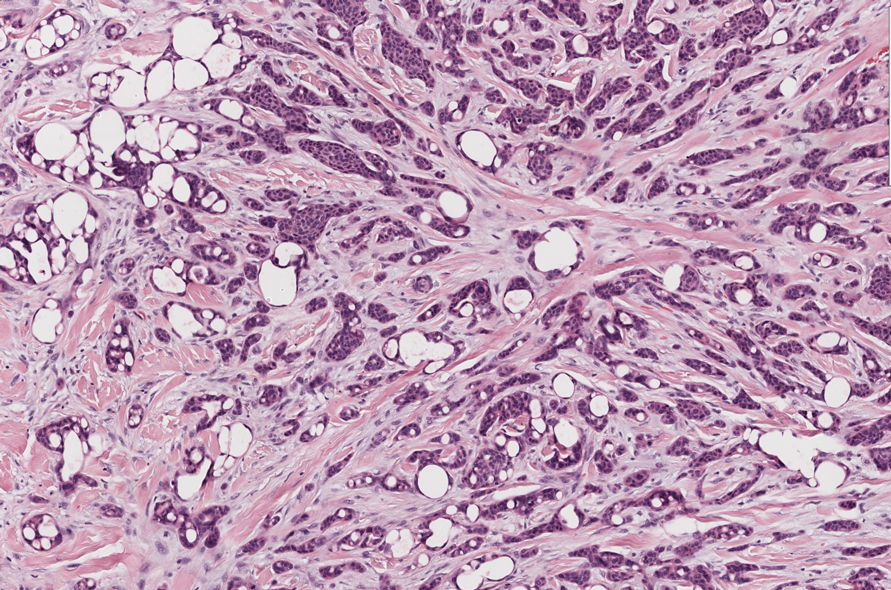
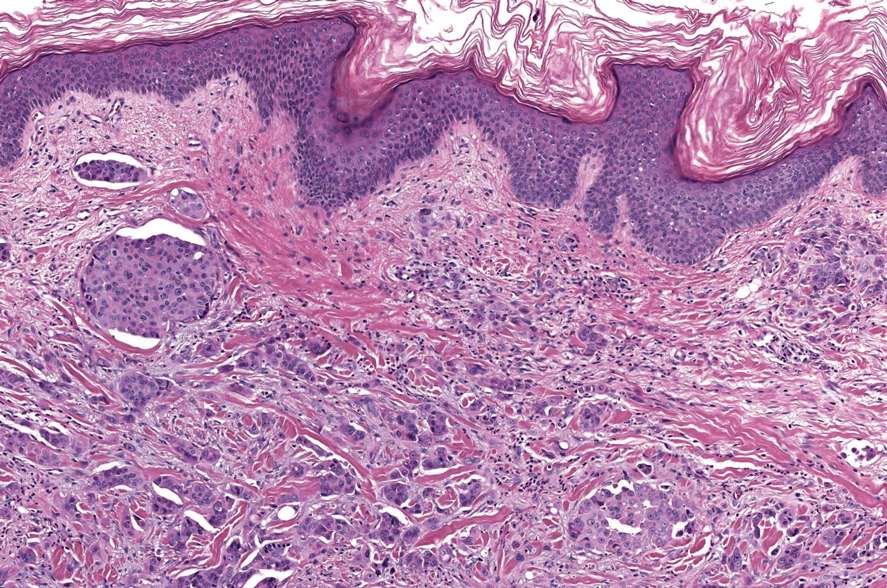
The histopathologic features of metastatic carcinomas can overlap with those of primary cutaneous tumors, particularly adnexal neoplasms.10 However, several key features can aid in the differentiation of cutaneous metastases, including a dermal-based growth pattern with or without subcutaneous involvement, the presence of multiple lesions, and the occurrence of lymphovascular invasion (Figure 5). Conversely, features that suggest a primary cutaneous adnexal neoplasm include the presence of superimposed in situ disease, carcinoma developing within a benign adnexal neoplasm, and notable stromal and/or vascular hyalinization within benign-appearing areas. In some cases, it can be difficult to determine the primary site of origin of a metastatic carcinoma to the skin based on morphologic features alone. In these cases, immunohistochemistry can be helpful. The most cost-effective and time-efficient approach to accurate diagnosis is to obtain a comprehensive clinical history. If there is a known history of cancer, a small panel of organ-specific immunohistochemical studies can be performed to confirm the diagnosis. If there is no known history, an algorithmic approach can be used to identify the primary site of origin. In all circumstances, it cannot be stressed enough that acquiring a thorough clinical history before conducting any diagnostic examinations is paramount.
- Bishop JA, Weinreb I, Swanson D, et al. Microsecretory adenocarcinoma: a novel salivary gland tumor characterized by a recurrent MEF2C-SS18 fusion. Am J Surg Pathol. 2019;43:1023-1032.
- Bishop JA, Williams EA, McLean AC, et al. Microsecretory adenocarcinoma of the skin harboring recurrent SS18 fusions: a cutaneous analog to a newly described salivary gland tumor. J Cutan Pathol. 2023;50:134-139.
- Macagno N, Sohier Pierre, Kervarrec T, et al. Recent advances on immunohistochemistry and molecular biology for the diagnosis of adnexal sweat gland tumors. Cancers (Basel). 2022;14:476.
- Bishop JA, Koduru P, Veremis BM, et al. SS18 break-apart fluorescence in situ hybridization is a practical and effective method for diagnosing microsecretory adenocarcinoma of salivary glands. Head Neck Pathol. 2021;15:723-726.
- Liau JY, Tsai JH, Huang WC, et al. BRAF and KRAS mutations in tubular apocrine adenoma and papillary eccrine adenoma of the skin. Hum Pathol. 2018;73:59-65.
- Chang MD, Arthur AK, Garcia JJ, et al. ETV6 rearrangement in a case of mammary analogue secretory carcinoma of the skin. J Cutan Pathol. 2016;43:1045-1049.
- Skalova A, Baneckova M, Thompson LDR, et al. Expanding the molecular spectrum of secretory carcinoma of salivary glands with a novel VIM-RET fusion. Am J Surg Pathol. 2020;44:1295-1307.
- Requena L, Kiryu H, Ackerman AB. Neoplasms With Apocrine Differentiation. Lippencott-Raven; 1998.
- Kazakov DV, Llamas-Velasco M, Fernandez-Flores A, et al. Cribriform tumour (previously carcinoma). In: WHO Classification of Tumours: Skin Tumours. 5th ed. International Agency for Research on Cancer; 2024.
- Habaermehl G, Ko J. Cutaneous metastases: a review and diagnostic approach to tumors of unknown origin. Arch Pathol Lab Med. 2019;143:943-957.
The Diagnosis: Microsecretory Adenocarcinoma
Microscopically, the tumor was relatively well circumscribed but had irregular borders. It consisted of microcysts and tubules lined by flattened to plump eosinophilic cells with mildly enlarged nuclei and intraluminal basophilic secretions. Peripheral lymphocytic aggregates also were seen in the mid and deep reticular dermis. Tumor necrosis, lymphovascular invasion, and notable mitotic activity were absent. Immunohistochemistry was diffusely positive for cytokeratin (CK) 7 and CK5/6. Occasional tumor cells showed variable expression of alpha smooth muscle actin, S-100 protein, and p40 and p63 antibodies. Immunohistochemistry was negative for CK20; GATA binding protein 3; MYB proto-oncogene, transcription factor; and insulinoma-associated protein 1. A dual-color, break-apart fluorescence in situ hybridization probe identified a rearrangement of the SS18 (SYT) gene locus on chromosome 18. The nodule was excised with clear surgical margins, and the patient had no evidence of recurrent disease or metastasis at 2-year follow-up.
In recent years, there has been a growing recognition of the pivotal role played by gene fusions in driving oncogenesis, encompassing a diverse range of benign and malignant cutaneous neoplasms. These investigations have shed light on previously unknown mechanisms and pathways contributing to the pathogenesis of these neoplastic conditions, offering invaluable insights into their underlying biology. As a result, our ability to classify and diagnose these cutaneous tumors has improved. A notable example of how our current understanding has evolved is the discovery of the new cutaneous adnexal tumor microsecretory adenocarcinoma (MSA). Initially described by Bishop et al1 in 2019 as predominantly occurring in the intraoral minor salivary glands, rare instances of primary cutaneous MSA involving the head and neck regions also have been reported.2 Microsecretory adenocarcinoma represents an important addition to the group of fusion-driven tumors with both salivary gland and cutaneous adnexal analogues, characterized by a MEF2C::SS18 gene fusion. This entity is now recognized as a group of cutaneous adnexal tumors with distinct gene fusions, including both relatively recently discovered entities (eg, secretory carcinoma with NTRK fusions) and previously known entities with newly identified gene fusions (eg, poroid neoplasms with NUTM1, YAP1, or WWTR1 fusions; hidradenomatous neoplasms with CRTC1::MAML2 fusions; and adenoid cystic carcinoma with MYB, MYBL1, and/or NFIB rearrangements).3
Microsecretory adenocarcinoma exhibits a high degree of morphologic consistency, characterized by a microcystic-predominant growth pattern, uniform intercalated ductlike tumor cells with attenuated eosinophilic to clear cytoplasm, monotonous oval hyperchromatic nuclei with indistinct nucleoli, abundant basophilic luminal secretions, and a variably cellular fibromyxoid stroma. It also shows rounded borders with subtle infiltrative growth. Occasionally, pseudoepitheliomatous hyperplasia, tumor-associated lymphoid proliferation, or metaplastic bone formation may accompany MSA. Perineural invasion is rare, necrosis is absent, and mitotic rates generally are low, contributing to its distinctive histopathologic features that aid in accurate diagnosis and differentiation from other entities. Immunohistochemistry reveals diffuse positivity for CK7 and patchy to diffuse expression of S-100 in tumor cells as well as variable expression of p40 and p63. Highly specific SS18 gene translocations at chromosome 18q are useful for diagnosing MSA when found alongside its characteristic appearance, and SS18 break-apart fluorescence in situ hybridization can serve reliably as an accurate diagnostic method (Figure 1).4 Our case illustrates how molecular analysis assists in distinguishing MSA from other cutaneous adnexal tumors, exemplifying the power of our evolving understanding in refining diagnostic accuracy and guiding targeted therapies in clinical practice.
The differential diagnosis of MSA includes tubular adenoma, secretory carcinoma, cribriform tumor (previously carcinoma), and metastatic adenocarcinoma. Tubular adenoma is a rare benign neoplasm that predominantly affects females and can manifest at any age in adulthood. It typically manifests as a slow-growing, occasionally pedunculated nodule, often measuring less than 2 cm. Although it most commonly manifests on the scalp, tubular adenoma also may arise in diverse sites such as the face, axillae, lower extremities, or genitalia.

Notably, scalp lesions often are associated with nevus sebaceus of Jadassohn or syringocystadenoma papilliferum. Microscopically, tubular adenoma is well circumscribed within the dermis and may extend into the subcutis in some cases. Its distinctive appearance consists of variably sized tubules lined by a double or multilayered cuboidal to columnar epithelium, frequently displaying apocrine decapitation secretion (Figure 2). Cystic changes and intraluminal papillae devoid of true fibrovascular cores frequently are observed. Immunohistochemically, luminal epithelial cells express epithelial membrane antigen and carcinoembryonic antigen, while the myoepithelial layer expresses smooth muscle markers, p40, and S-100 protein. BRAF V600E mutation can be detected using immunohistochemistry, with excellent sensitivity and specificity using the anti-BRAF V600E antibody (clone VE1).5 Distinguishing tubular adenoma from MSA is achievable by observing its larger, more variable tubules, along with the consistent presence of a peripheral myoepithelial layer.
Secretory carcinoma is recognized as a low-grade gene fusion–driven carcinoma that primarily arises in salivary glands (both major and minor), with occasional occurrences in the breast and extremely rare instances in other locations such as the skin, thyroid gland, and lung.6 Although the axilla is the most common cutaneous site, diverse locations such as the neck, eyelids, extremities, and nipples also have been documented. Secretory carcinoma affects individuals across a wide age range (13–71 years).6 The hallmark tumors exhibit densely packed, sievelike microcystic glands and tubular spaces filled with abundant eosinophilic intraluminal secretions (Figure 3). Additionally, morphologic variants, such as predominantly papillary, papillary-cystic, macrocystic, solid, partially mucinous, and mixed-pattern neoplasms, have been described. Secretory carcinoma shares certain features with MSA; however, it is distinguished by the presence of pronounced eosinophilic secretions, plump and vacuolated cytoplasm, and a less conspicuous fibromyxoid stroma. Immunohistochemistry reveals tumor cells that are positive for CK7, SOX-10, S-100, mammaglobin, MUC4, and variably GATA-3. Genetically, secretory carcinoma exhibits distinct characteristics, commonly showing the ETV6::NTRK3 fusion, detectable through molecular techniques or pan-TRK immunohistochemistry, while RET fusions and other rare variants are less frequent.7

In 1998, Requena et al8 introduced the concept of primary cutaneous cribriform carcinoma. Despite initially being classified as a carcinoma, the malignant potential of this tumor remains uncertain. Consequently, the term cribriform tumor now has become the preferred terminology for denoting this rare entity.9 Primary cutaneous cribriform tumors are observed more commonly in women and typically affect individuals aged 20 to 55 years (mean, 44 years). Predominant locations include the upper and lower extremities, especially the thighs, knees, and legs, with additional cases occurring on the head and trunk. Microscopically, cribriform tumor is characterized by a partially circumscribed, unencapsulated dermal nodule composed of round or oval nuclei displaying hyperchromatism and mild pleomorphism. The defining aspect of its morphology revolves around interspersed small round cavities that give rise to the hallmark cribriform pattern (Figure 4). Although MSA occasionally may exhibit a cribriform architectural pattern, it typically lacks the distinctive feature of thin, threadlike, intraluminal bridging strands observed in cribriform tumors. Similarly, luminal cells within the cribriform tumor express CK7 and exhibit variable S-100 expression. It is recognized as an indolent neoplasm with uncertain malignant potential.



The histopathologic features of metastatic carcinomas can overlap with those of primary cutaneous tumors, particularly adnexal neoplasms.10 However, several key features can aid in the differentiation of cutaneous metastases, including a dermal-based growth pattern with or without subcutaneous involvement, the presence of multiple lesions, and the occurrence of lymphovascular invasion (Figure 5). Conversely, features that suggest a primary cutaneous adnexal neoplasm include the presence of superimposed in situ disease, carcinoma developing within a benign adnexal neoplasm, and notable stromal and/or vascular hyalinization within benign-appearing areas. In some cases, it can be difficult to determine the primary site of origin of a metastatic carcinoma to the skin based on morphologic features alone. In these cases, immunohistochemistry can be helpful. The most cost-effective and time-efficient approach to accurate diagnosis is to obtain a comprehensive clinical history. If there is a known history of cancer, a small panel of organ-specific immunohistochemical studies can be performed to confirm the diagnosis. If there is no known history, an algorithmic approach can be used to identify the primary site of origin. In all circumstances, it cannot be stressed enough that acquiring a thorough clinical history before conducting any diagnostic examinations is paramount.
The Diagnosis: Microsecretory Adenocarcinoma
Microscopically, the tumor was relatively well circumscribed but had irregular borders. It consisted of microcysts and tubules lined by flattened to plump eosinophilic cells with mildly enlarged nuclei and intraluminal basophilic secretions. Peripheral lymphocytic aggregates also were seen in the mid and deep reticular dermis. Tumor necrosis, lymphovascular invasion, and notable mitotic activity were absent. Immunohistochemistry was diffusely positive for cytokeratin (CK) 7 and CK5/6. Occasional tumor cells showed variable expression of alpha smooth muscle actin, S-100 protein, and p40 and p63 antibodies. Immunohistochemistry was negative for CK20; GATA binding protein 3; MYB proto-oncogene, transcription factor; and insulinoma-associated protein 1. A dual-color, break-apart fluorescence in situ hybridization probe identified a rearrangement of the SS18 (SYT) gene locus on chromosome 18. The nodule was excised with clear surgical margins, and the patient had no evidence of recurrent disease or metastasis at 2-year follow-up.
In recent years, there has been a growing recognition of the pivotal role played by gene fusions in driving oncogenesis, encompassing a diverse range of benign and malignant cutaneous neoplasms. These investigations have shed light on previously unknown mechanisms and pathways contributing to the pathogenesis of these neoplastic conditions, offering invaluable insights into their underlying biology. As a result, our ability to classify and diagnose these cutaneous tumors has improved. A notable example of how our current understanding has evolved is the discovery of the new cutaneous adnexal tumor microsecretory adenocarcinoma (MSA). Initially described by Bishop et al1 in 2019 as predominantly occurring in the intraoral minor salivary glands, rare instances of primary cutaneous MSA involving the head and neck regions also have been reported.2 Microsecretory adenocarcinoma represents an important addition to the group of fusion-driven tumors with both salivary gland and cutaneous adnexal analogues, characterized by a MEF2C::SS18 gene fusion. This entity is now recognized as a group of cutaneous adnexal tumors with distinct gene fusions, including both relatively recently discovered entities (eg, secretory carcinoma with NTRK fusions) and previously known entities with newly identified gene fusions (eg, poroid neoplasms with NUTM1, YAP1, or WWTR1 fusions; hidradenomatous neoplasms with CRTC1::MAML2 fusions; and adenoid cystic carcinoma with MYB, MYBL1, and/or NFIB rearrangements).3
Microsecretory adenocarcinoma exhibits a high degree of morphologic consistency, characterized by a microcystic-predominant growth pattern, uniform intercalated ductlike tumor cells with attenuated eosinophilic to clear cytoplasm, monotonous oval hyperchromatic nuclei with indistinct nucleoli, abundant basophilic luminal secretions, and a variably cellular fibromyxoid stroma. It also shows rounded borders with subtle infiltrative growth. Occasionally, pseudoepitheliomatous hyperplasia, tumor-associated lymphoid proliferation, or metaplastic bone formation may accompany MSA. Perineural invasion is rare, necrosis is absent, and mitotic rates generally are low, contributing to its distinctive histopathologic features that aid in accurate diagnosis and differentiation from other entities. Immunohistochemistry reveals diffuse positivity for CK7 and patchy to diffuse expression of S-100 in tumor cells as well as variable expression of p40 and p63. Highly specific SS18 gene translocations at chromosome 18q are useful for diagnosing MSA when found alongside its characteristic appearance, and SS18 break-apart fluorescence in situ hybridization can serve reliably as an accurate diagnostic method (Figure 1).4 Our case illustrates how molecular analysis assists in distinguishing MSA from other cutaneous adnexal tumors, exemplifying the power of our evolving understanding in refining diagnostic accuracy and guiding targeted therapies in clinical practice.
The differential diagnosis of MSA includes tubular adenoma, secretory carcinoma, cribriform tumor (previously carcinoma), and metastatic adenocarcinoma. Tubular adenoma is a rare benign neoplasm that predominantly affects females and can manifest at any age in adulthood. It typically manifests as a slow-growing, occasionally pedunculated nodule, often measuring less than 2 cm. Although it most commonly manifests on the scalp, tubular adenoma also may arise in diverse sites such as the face, axillae, lower extremities, or genitalia.

Notably, scalp lesions often are associated with nevus sebaceus of Jadassohn or syringocystadenoma papilliferum. Microscopically, tubular adenoma is well circumscribed within the dermis and may extend into the subcutis in some cases. Its distinctive appearance consists of variably sized tubules lined by a double or multilayered cuboidal to columnar epithelium, frequently displaying apocrine decapitation secretion (Figure 2). Cystic changes and intraluminal papillae devoid of true fibrovascular cores frequently are observed. Immunohistochemically, luminal epithelial cells express epithelial membrane antigen and carcinoembryonic antigen, while the myoepithelial layer expresses smooth muscle markers, p40, and S-100 protein. BRAF V600E mutation can be detected using immunohistochemistry, with excellent sensitivity and specificity using the anti-BRAF V600E antibody (clone VE1).5 Distinguishing tubular adenoma from MSA is achievable by observing its larger, more variable tubules, along with the consistent presence of a peripheral myoepithelial layer.
Secretory carcinoma is recognized as a low-grade gene fusion–driven carcinoma that primarily arises in salivary glands (both major and minor), with occasional occurrences in the breast and extremely rare instances in other locations such as the skin, thyroid gland, and lung.6 Although the axilla is the most common cutaneous site, diverse locations such as the neck, eyelids, extremities, and nipples also have been documented. Secretory carcinoma affects individuals across a wide age range (13–71 years).6 The hallmark tumors exhibit densely packed, sievelike microcystic glands and tubular spaces filled with abundant eosinophilic intraluminal secretions (Figure 3). Additionally, morphologic variants, such as predominantly papillary, papillary-cystic, macrocystic, solid, partially mucinous, and mixed-pattern neoplasms, have been described. Secretory carcinoma shares certain features with MSA; however, it is distinguished by the presence of pronounced eosinophilic secretions, plump and vacuolated cytoplasm, and a less conspicuous fibromyxoid stroma. Immunohistochemistry reveals tumor cells that are positive for CK7, SOX-10, S-100, mammaglobin, MUC4, and variably GATA-3. Genetically, secretory carcinoma exhibits distinct characteristics, commonly showing the ETV6::NTRK3 fusion, detectable through molecular techniques or pan-TRK immunohistochemistry, while RET fusions and other rare variants are less frequent.7

In 1998, Requena et al8 introduced the concept of primary cutaneous cribriform carcinoma. Despite initially being classified as a carcinoma, the malignant potential of this tumor remains uncertain. Consequently, the term cribriform tumor now has become the preferred terminology for denoting this rare entity.9 Primary cutaneous cribriform tumors are observed more commonly in women and typically affect individuals aged 20 to 55 years (mean, 44 years). Predominant locations include the upper and lower extremities, especially the thighs, knees, and legs, with additional cases occurring on the head and trunk. Microscopically, cribriform tumor is characterized by a partially circumscribed, unencapsulated dermal nodule composed of round or oval nuclei displaying hyperchromatism and mild pleomorphism. The defining aspect of its morphology revolves around interspersed small round cavities that give rise to the hallmark cribriform pattern (Figure 4). Although MSA occasionally may exhibit a cribriform architectural pattern, it typically lacks the distinctive feature of thin, threadlike, intraluminal bridging strands observed in cribriform tumors. Similarly, luminal cells within the cribriform tumor express CK7 and exhibit variable S-100 expression. It is recognized as an indolent neoplasm with uncertain malignant potential.



The histopathologic features of metastatic carcinomas can overlap with those of primary cutaneous tumors, particularly adnexal neoplasms.10 However, several key features can aid in the differentiation of cutaneous metastases, including a dermal-based growth pattern with or without subcutaneous involvement, the presence of multiple lesions, and the occurrence of lymphovascular invasion (Figure 5). Conversely, features that suggest a primary cutaneous adnexal neoplasm include the presence of superimposed in situ disease, carcinoma developing within a benign adnexal neoplasm, and notable stromal and/or vascular hyalinization within benign-appearing areas. In some cases, it can be difficult to determine the primary site of origin of a metastatic carcinoma to the skin based on morphologic features alone. In these cases, immunohistochemistry can be helpful. The most cost-effective and time-efficient approach to accurate diagnosis is to obtain a comprehensive clinical history. If there is a known history of cancer, a small panel of organ-specific immunohistochemical studies can be performed to confirm the diagnosis. If there is no known history, an algorithmic approach can be used to identify the primary site of origin. In all circumstances, it cannot be stressed enough that acquiring a thorough clinical history before conducting any diagnostic examinations is paramount.
- Bishop JA, Weinreb I, Swanson D, et al. Microsecretory adenocarcinoma: a novel salivary gland tumor characterized by a recurrent MEF2C-SS18 fusion. Am J Surg Pathol. 2019;43:1023-1032.
- Bishop JA, Williams EA, McLean AC, et al. Microsecretory adenocarcinoma of the skin harboring recurrent SS18 fusions: a cutaneous analog to a newly described salivary gland tumor. J Cutan Pathol. 2023;50:134-139.
- Macagno N, Sohier Pierre, Kervarrec T, et al. Recent advances on immunohistochemistry and molecular biology for the diagnosis of adnexal sweat gland tumors. Cancers (Basel). 2022;14:476.
- Bishop JA, Koduru P, Veremis BM, et al. SS18 break-apart fluorescence in situ hybridization is a practical and effective method for diagnosing microsecretory adenocarcinoma of salivary glands. Head Neck Pathol. 2021;15:723-726.
- Liau JY, Tsai JH, Huang WC, et al. BRAF and KRAS mutations in tubular apocrine adenoma and papillary eccrine adenoma of the skin. Hum Pathol. 2018;73:59-65.
- Chang MD, Arthur AK, Garcia JJ, et al. ETV6 rearrangement in a case of mammary analogue secretory carcinoma of the skin. J Cutan Pathol. 2016;43:1045-1049.
- Skalova A, Baneckova M, Thompson LDR, et al. Expanding the molecular spectrum of secretory carcinoma of salivary glands with a novel VIM-RET fusion. Am J Surg Pathol. 2020;44:1295-1307.
- Requena L, Kiryu H, Ackerman AB. Neoplasms With Apocrine Differentiation. Lippencott-Raven; 1998.
- Kazakov DV, Llamas-Velasco M, Fernandez-Flores A, et al. Cribriform tumour (previously carcinoma). In: WHO Classification of Tumours: Skin Tumours. 5th ed. International Agency for Research on Cancer; 2024.
- Habaermehl G, Ko J. Cutaneous metastases: a review and diagnostic approach to tumors of unknown origin. Arch Pathol Lab Med. 2019;143:943-957.
- Bishop JA, Weinreb I, Swanson D, et al. Microsecretory adenocarcinoma: a novel salivary gland tumor characterized by a recurrent MEF2C-SS18 fusion. Am J Surg Pathol. 2019;43:1023-1032.
- Bishop JA, Williams EA, McLean AC, et al. Microsecretory adenocarcinoma of the skin harboring recurrent SS18 fusions: a cutaneous analog to a newly described salivary gland tumor. J Cutan Pathol. 2023;50:134-139.
- Macagno N, Sohier Pierre, Kervarrec T, et al. Recent advances on immunohistochemistry and molecular biology for the diagnosis of adnexal sweat gland tumors. Cancers (Basel). 2022;14:476.
- Bishop JA, Koduru P, Veremis BM, et al. SS18 break-apart fluorescence in situ hybridization is a practical and effective method for diagnosing microsecretory adenocarcinoma of salivary glands. Head Neck Pathol. 2021;15:723-726.
- Liau JY, Tsai JH, Huang WC, et al. BRAF and KRAS mutations in tubular apocrine adenoma and papillary eccrine adenoma of the skin. Hum Pathol. 2018;73:59-65.
- Chang MD, Arthur AK, Garcia JJ, et al. ETV6 rearrangement in a case of mammary analogue secretory carcinoma of the skin. J Cutan Pathol. 2016;43:1045-1049.
- Skalova A, Baneckova M, Thompson LDR, et al. Expanding the molecular spectrum of secretory carcinoma of salivary glands with a novel VIM-RET fusion. Am J Surg Pathol. 2020;44:1295-1307.
- Requena L, Kiryu H, Ackerman AB. Neoplasms With Apocrine Differentiation. Lippencott-Raven; 1998.
- Kazakov DV, Llamas-Velasco M, Fernandez-Flores A, et al. Cribriform tumour (previously carcinoma). In: WHO Classification of Tumours: Skin Tumours. 5th ed. International Agency for Research on Cancer; 2024.
- Habaermehl G, Ko J. Cutaneous metastases: a review and diagnostic approach to tumors of unknown origin. Arch Pathol Lab Med. 2019;143:943-957.
A 74-year-old man presented with an asymptomatic nodule on the left neck measuring approximately 2 cm. An excisional biopsy was obtained for histopathologic evaluation.
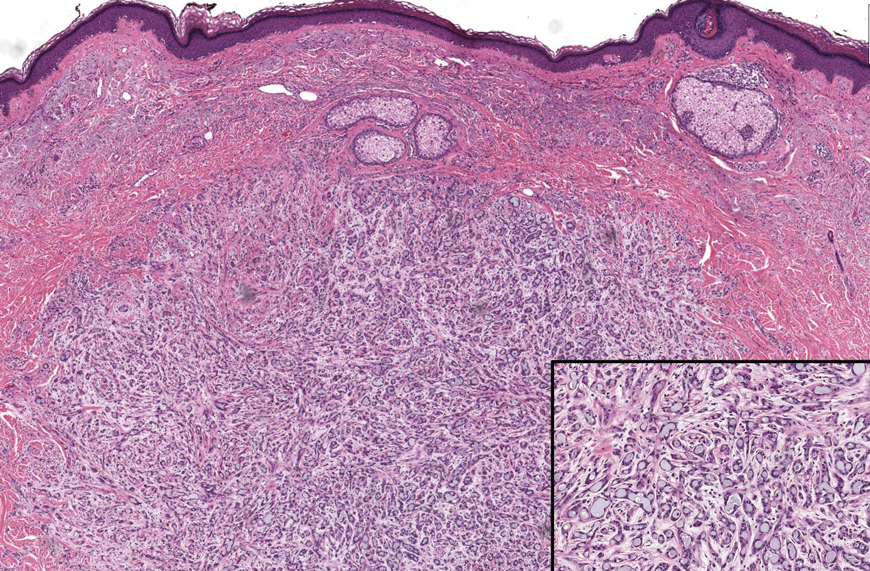
Weight gain despite dieting
Binge-eating disorder is more prevalent in women than men and has one of the strongest associations with obesity; among patients with obesity, lifetime prevalence of binge eating is approximately 5.5%. Large population studies suggest that binge-eating disorder may be present in 2%-4% of adolescents, with a mean age of onset of 12-13 years. This patient probably had milder binge-eating disorder as an adolescent and young adult, which was exacerbated by the pandemic.
Both new diagnoses and reports of clinical worsening in patients with preexisting diagnoses of binge-eating disorder during the pandemic have been documented. Food insecurity has been associated with binge eating, consistent with this patient's anxiety over food and grocery availability during the pandemic. The definition of binge-eating disorder includes recurrent specific episodes of overeating that are not consistent with the patient's usual behavior, eating to the point of being uncomfortably full, eating more quickly or when not hungry, and having feelings of loss of control during episodes and of guilt or disgust afterward.
Obesity and eating disorders share some common risk factors and approaches to management. Binge eating has been associated with type 2 diabetes, hypertension, asthma, sleep disorders, and menstrual disorders, all of which are also affected by obesity. The presence of both conditions increases the adverse outcomes associated with each, including negative impacts on cardiometabolic and psychological health. Workup of patients presenting with binge eating and obesity should always include complete blood/metabolic panels and cardiovascular and renal health, as well as assessments of nutrition status, electrolyte imbalances, gastrointestinal reflux disease, and chronic pain.
In general, where binge-eating disorder and obesity are concurrent, cognitive-behavioral therapy (CBT) for binge-eating disorder should be the first priority, with obesity management (medication or surgery) to follow. CBT has the strongest evidence of benefit for patients with binge-eating disorder and is the recommended treatment approach. Other psychotherapeutic interventions that may be of benefit include dialectical behavioral therapy (to reduce binge-eating frequency), technology-based options, and family-based therapy when symptoms are recognized in children or adolescents. Structured behavioral weight management strategies for management of obesity and overweight do not increase symptoms of eating disorders and may instead relieve some symptoms. An emerging approach to binge eating in patients with obesity is CBT that integrates therapeutic approaches to both issues.
Medications to treat binge-eating disorder are limited and should not be used without concurrent psychotherapy; lisdexamfetamine has demonstrated benefit, is recommended by the American Psychiatric Association, and is approved by the US Food and Drug Administration specifically to treat adults with binge-eating disorder.
The success of psychological interventions and lifestyle modifications for obesity is heavily dependent on the individual's ability and motivation to comply with recommended interventions. The American Gastroenterological Association and other organizations recommend treatment with antiobesity medications along with lifestyle modifications for patients with obesity (BMI ≥ 30) and weight-related complications (BMI > 27). Recommended medications include phentermine-topiramate and bupropion-naltrexone (which may benefit those with binge-eating disorder), as well as injectable glucagon-like peptide receptor agonists (GLP-1 RAs) at the approved dosage for obesity management (semaglutide 2.4 mg weekly or liraglutide 3.0 mg daily). Orlistat is not recommended. Ongoing research on the potential benefit of GLP-1 RAs in management of binge eating offers additional support for a role in patients, like this one, with binge-eating disorder and obesity.
Carolyn Newbury, MD, Assistant Professor of Medicine, Director of GI Nutrition, Innovative Center for Health and Nutrition in Gastroenterology (ICHANGE), Division of Gastroenterology, Weill Cornell Medical Center, New York, NY.
Disclosure: Carolyn Newberry, MD, has disclosed the following relevant financial relationships:
Serve(d) as a speaker or a member of a speakers bureau for: Baster International; InBody.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Binge-eating disorder is more prevalent in women than men and has one of the strongest associations with obesity; among patients with obesity, lifetime prevalence of binge eating is approximately 5.5%. Large population studies suggest that binge-eating disorder may be present in 2%-4% of adolescents, with a mean age of onset of 12-13 years. This patient probably had milder binge-eating disorder as an adolescent and young adult, which was exacerbated by the pandemic.
Both new diagnoses and reports of clinical worsening in patients with preexisting diagnoses of binge-eating disorder during the pandemic have been documented. Food insecurity has been associated with binge eating, consistent with this patient's anxiety over food and grocery availability during the pandemic. The definition of binge-eating disorder includes recurrent specific episodes of overeating that are not consistent with the patient's usual behavior, eating to the point of being uncomfortably full, eating more quickly or when not hungry, and having feelings of loss of control during episodes and of guilt or disgust afterward.
Obesity and eating disorders share some common risk factors and approaches to management. Binge eating has been associated with type 2 diabetes, hypertension, asthma, sleep disorders, and menstrual disorders, all of which are also affected by obesity. The presence of both conditions increases the adverse outcomes associated with each, including negative impacts on cardiometabolic and psychological health. Workup of patients presenting with binge eating and obesity should always include complete blood/metabolic panels and cardiovascular and renal health, as well as assessments of nutrition status, electrolyte imbalances, gastrointestinal reflux disease, and chronic pain.
In general, where binge-eating disorder and obesity are concurrent, cognitive-behavioral therapy (CBT) for binge-eating disorder should be the first priority, with obesity management (medication or surgery) to follow. CBT has the strongest evidence of benefit for patients with binge-eating disorder and is the recommended treatment approach. Other psychotherapeutic interventions that may be of benefit include dialectical behavioral therapy (to reduce binge-eating frequency), technology-based options, and family-based therapy when symptoms are recognized in children or adolescents. Structured behavioral weight management strategies for management of obesity and overweight do not increase symptoms of eating disorders and may instead relieve some symptoms. An emerging approach to binge eating in patients with obesity is CBT that integrates therapeutic approaches to both issues.
Medications to treat binge-eating disorder are limited and should not be used without concurrent psychotherapy; lisdexamfetamine has demonstrated benefit, is recommended by the American Psychiatric Association, and is approved by the US Food and Drug Administration specifically to treat adults with binge-eating disorder.
The success of psychological interventions and lifestyle modifications for obesity is heavily dependent on the individual's ability and motivation to comply with recommended interventions. The American Gastroenterological Association and other organizations recommend treatment with antiobesity medications along with lifestyle modifications for patients with obesity (BMI ≥ 30) and weight-related complications (BMI > 27). Recommended medications include phentermine-topiramate and bupropion-naltrexone (which may benefit those with binge-eating disorder), as well as injectable glucagon-like peptide receptor agonists (GLP-1 RAs) at the approved dosage for obesity management (semaglutide 2.4 mg weekly or liraglutide 3.0 mg daily). Orlistat is not recommended. Ongoing research on the potential benefit of GLP-1 RAs in management of binge eating offers additional support for a role in patients, like this one, with binge-eating disorder and obesity.
Carolyn Newbury, MD, Assistant Professor of Medicine, Director of GI Nutrition, Innovative Center for Health and Nutrition in Gastroenterology (ICHANGE), Division of Gastroenterology, Weill Cornell Medical Center, New York, NY.
Disclosure: Carolyn Newberry, MD, has disclosed the following relevant financial relationships:
Serve(d) as a speaker or a member of a speakers bureau for: Baster International; InBody.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Binge-eating disorder is more prevalent in women than men and has one of the strongest associations with obesity; among patients with obesity, lifetime prevalence of binge eating is approximately 5.5%. Large population studies suggest that binge-eating disorder may be present in 2%-4% of adolescents, with a mean age of onset of 12-13 years. This patient probably had milder binge-eating disorder as an adolescent and young adult, which was exacerbated by the pandemic.
Both new diagnoses and reports of clinical worsening in patients with preexisting diagnoses of binge-eating disorder during the pandemic have been documented. Food insecurity has been associated with binge eating, consistent with this patient's anxiety over food and grocery availability during the pandemic. The definition of binge-eating disorder includes recurrent specific episodes of overeating that are not consistent with the patient's usual behavior, eating to the point of being uncomfortably full, eating more quickly or when not hungry, and having feelings of loss of control during episodes and of guilt or disgust afterward.
Obesity and eating disorders share some common risk factors and approaches to management. Binge eating has been associated with type 2 diabetes, hypertension, asthma, sleep disorders, and menstrual disorders, all of which are also affected by obesity. The presence of both conditions increases the adverse outcomes associated with each, including negative impacts on cardiometabolic and psychological health. Workup of patients presenting with binge eating and obesity should always include complete blood/metabolic panels and cardiovascular and renal health, as well as assessments of nutrition status, electrolyte imbalances, gastrointestinal reflux disease, and chronic pain.
In general, where binge-eating disorder and obesity are concurrent, cognitive-behavioral therapy (CBT) for binge-eating disorder should be the first priority, with obesity management (medication or surgery) to follow. CBT has the strongest evidence of benefit for patients with binge-eating disorder and is the recommended treatment approach. Other psychotherapeutic interventions that may be of benefit include dialectical behavioral therapy (to reduce binge-eating frequency), technology-based options, and family-based therapy when symptoms are recognized in children or adolescents. Structured behavioral weight management strategies for management of obesity and overweight do not increase symptoms of eating disorders and may instead relieve some symptoms. An emerging approach to binge eating in patients with obesity is CBT that integrates therapeutic approaches to both issues.
Medications to treat binge-eating disorder are limited and should not be used without concurrent psychotherapy; lisdexamfetamine has demonstrated benefit, is recommended by the American Psychiatric Association, and is approved by the US Food and Drug Administration specifically to treat adults with binge-eating disorder.
The success of psychological interventions and lifestyle modifications for obesity is heavily dependent on the individual's ability and motivation to comply with recommended interventions. The American Gastroenterological Association and other organizations recommend treatment with antiobesity medications along with lifestyle modifications for patients with obesity (BMI ≥ 30) and weight-related complications (BMI > 27). Recommended medications include phentermine-topiramate and bupropion-naltrexone (which may benefit those with binge-eating disorder), as well as injectable glucagon-like peptide receptor agonists (GLP-1 RAs) at the approved dosage for obesity management (semaglutide 2.4 mg weekly or liraglutide 3.0 mg daily). Orlistat is not recommended. Ongoing research on the potential benefit of GLP-1 RAs in management of binge eating offers additional support for a role in patients, like this one, with binge-eating disorder and obesity.
Carolyn Newbury, MD, Assistant Professor of Medicine, Director of GI Nutrition, Innovative Center for Health and Nutrition in Gastroenterology (ICHANGE), Division of Gastroenterology, Weill Cornell Medical Center, New York, NY.
Disclosure: Carolyn Newberry, MD, has disclosed the following relevant financial relationships:
Serve(d) as a speaker or a member of a speakers bureau for: Baster International; InBody.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 28-year-old woman presents with concerns about weight gain despite dieting. She is 5 ft 4 in and weighs 180 lb (BMI 30.9). The patient lives alone and says she often feels isolated and has ongoing anxiety. She states that she has been overweight since her early teen years and had rare episodes of overeating. As an adult, her weight remained relatively stable (BMI ~26) until she began working remotely because of the COVID-19 pandemic in March 2020. She admits to becoming increasingly anxious and worried about food availability and grocery shopping during the early pandemic closures, feelings that have not completely resolved. While working from home, she has had more days where she compulsively overeats, even while trying to diet or use supplements she saw on TV or the internet. She stopped participating in a regular exercise walking group in mid-2020 and has not returned to it.
At presentation, she appears anxious and nervous. Her blood pressure is elevated (140/90 mm Hg), heart rate is 110 beats/min, and respiratory rate is 18 breaths/min. Her results on the Generalized Anxiety Disorder assessment indicate moderate symptoms of anxiety. Lab results indicate A1c = 6.5%, low-density lipoprotein cholesterol = 105 mg/dL, and estimated glomerular filtration rate = 90 mL/min/1.73 m2; all other results are within normal.
Epidermal Tumors Arising on Donor Sites From Autologous Skin Grafts: A Systematic Review
Skin grafting is a surgical technique used to cover skin defects resulting from the removal of skin tumors, ulcers, or burn injuries.1-3 Complications can occur at both donor and recipient sites and may include bleeding, hematoma/seroma formation, postoperative pain, infection, scarring, paresthesia, skin pigmentation, graft contracture, and graft failure.1,2,4,5 The development of epidermal tumors is not commonly reported among the complications of skin grafting; however, cases of epidermal tumor development on skin graft donor sites during the postoperative period have been reported.6-12
We performed a systematic review of the literature for cases of epidermal tumor development on skin graft donor sites in patients undergoing autologous skin graft surgery. We present the clinical characteristics of these cases and discuss the nature of these tumors.
Methods
Search Strategy and Study Selection—A literature search was conducted by 2 independent researchers (Z.P. and V.P.) for articles published before December 2022 in the following databases: MEDLINE/PubMed, Web of Science, Scopus, Cochrane Library, OpenGrey, Google Scholar, and WorldCat. Search terms included all possible combinations of the following: keratoacanthoma, molluscum sebaceum, basal cell carcinoma, squamous cell carcinoma, acanthoma, wart, Merkel cell carcinoma, verruca, Bowen disease, keratosis, skin cancer, cutaneous cancer, skin neoplasia, cutaneous neoplasia, and skin tumor. The literature search terms were selected based on the World Health Organization classification of skin tumors.13 Manual bibliography checks were performed on all eligible search results for possible relevant studies. Discrepancies were resolved through discussion and, if needed, mediation by a third researcher (N.C.). To be included, a study had to report a case(s) of epidermal tumor(s) that was confirmed by histopathology and arose on a graft donor site in a patient receiving autologous skin grafts for any reason. No language, geographic, or report date restrictions were set.
Data Extraction, Quality Assessment, and Statistical Analysis—We adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.14 Two independent researchers (Z.P. and V.P.) retrieved the data from the included studies. We have used the terms case and patient interchangeably, and 1 month was measured as 4 weeks for simplicity. Disagreements were resolved by discussion and mediation by a third researcher (N.C.). The quality of the included studies was assessed by 2 researchers (M.P. and V.P.) using the tool proposed by Murad et al.15
We used descriptive statistical analysis to analyze clinical characteristics of the included cases. We performed separate descriptive analyses based on the most frequently reported types of epidermal tumors and compared the differences between different groups using the Mann-Whitney U test, χ2 test, and Fisher exact test. The level of significance was set at P<.05. All statistical analyses were conducted using SPSS (version 29).
Results
Literature Search and Characteristics of Included Studies—The initial literature search identified 1378 studies, which were screened based on title and abstract. After removing duplicate and irrelevant studies and evaluating the full text of eligible studies, 31 studies (4 case series and 27 case reports) were included in the systematic review (Figure).6-12,16-39 Quality assessment of the included studies is presented in Table 1.
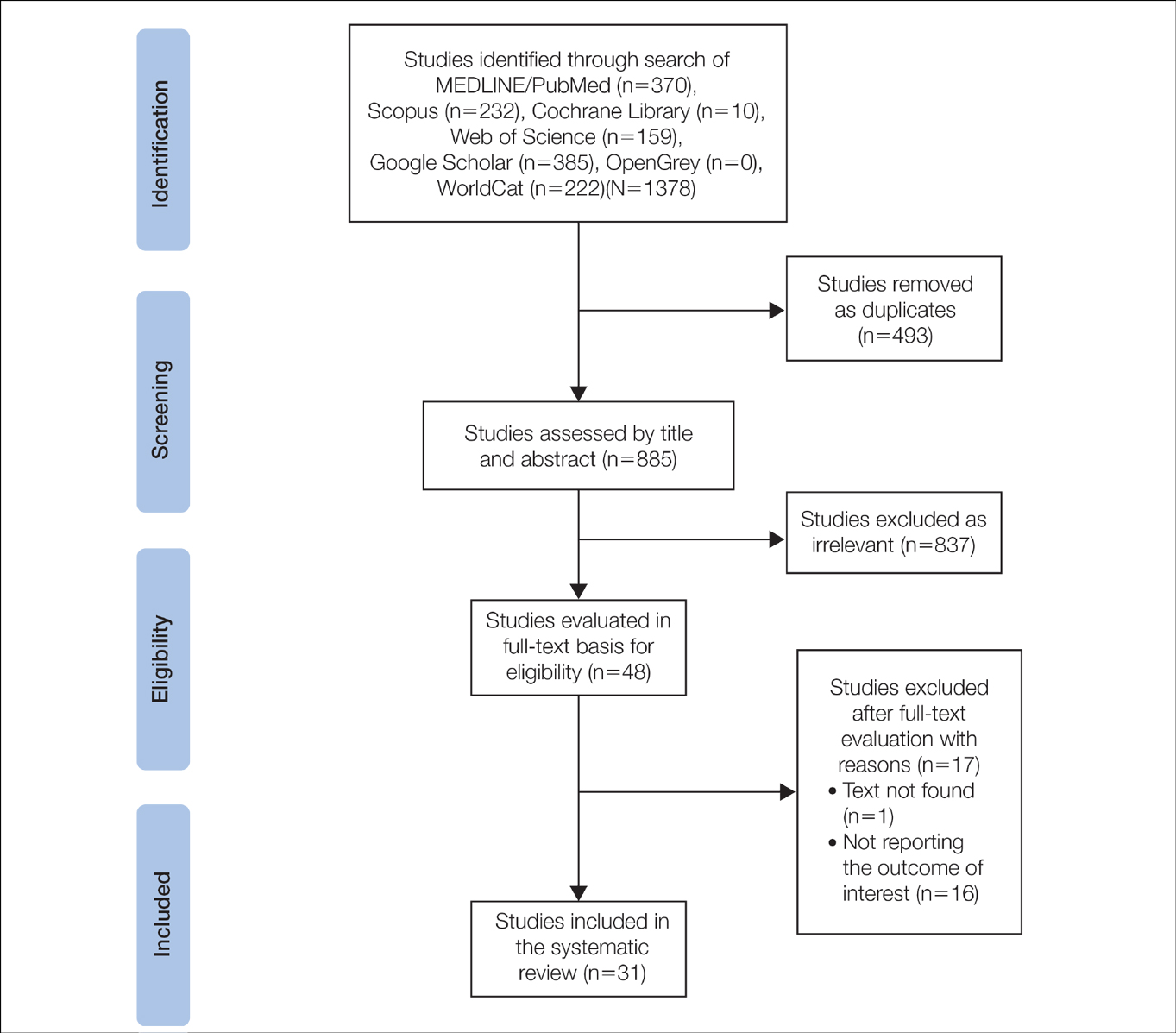
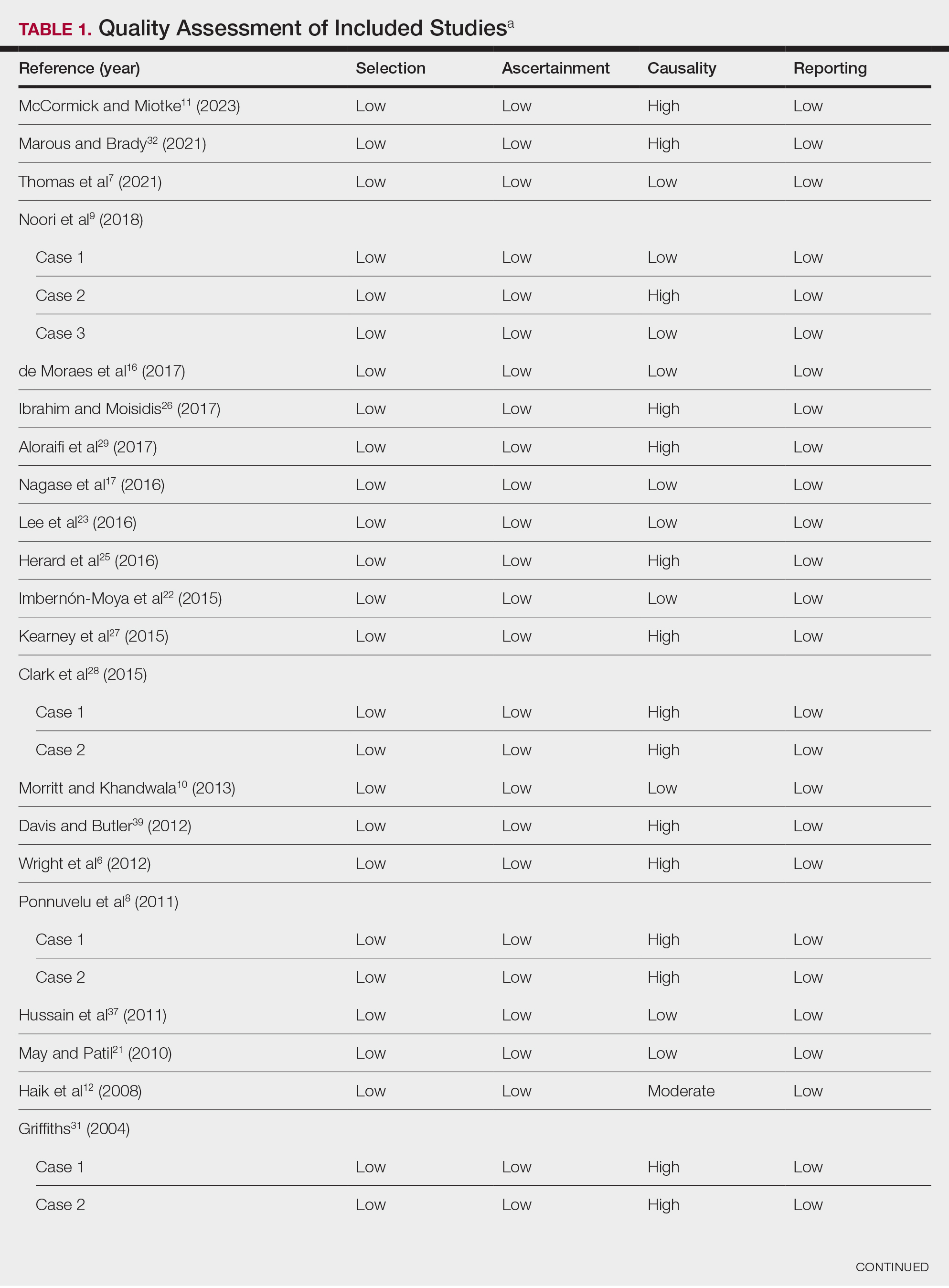
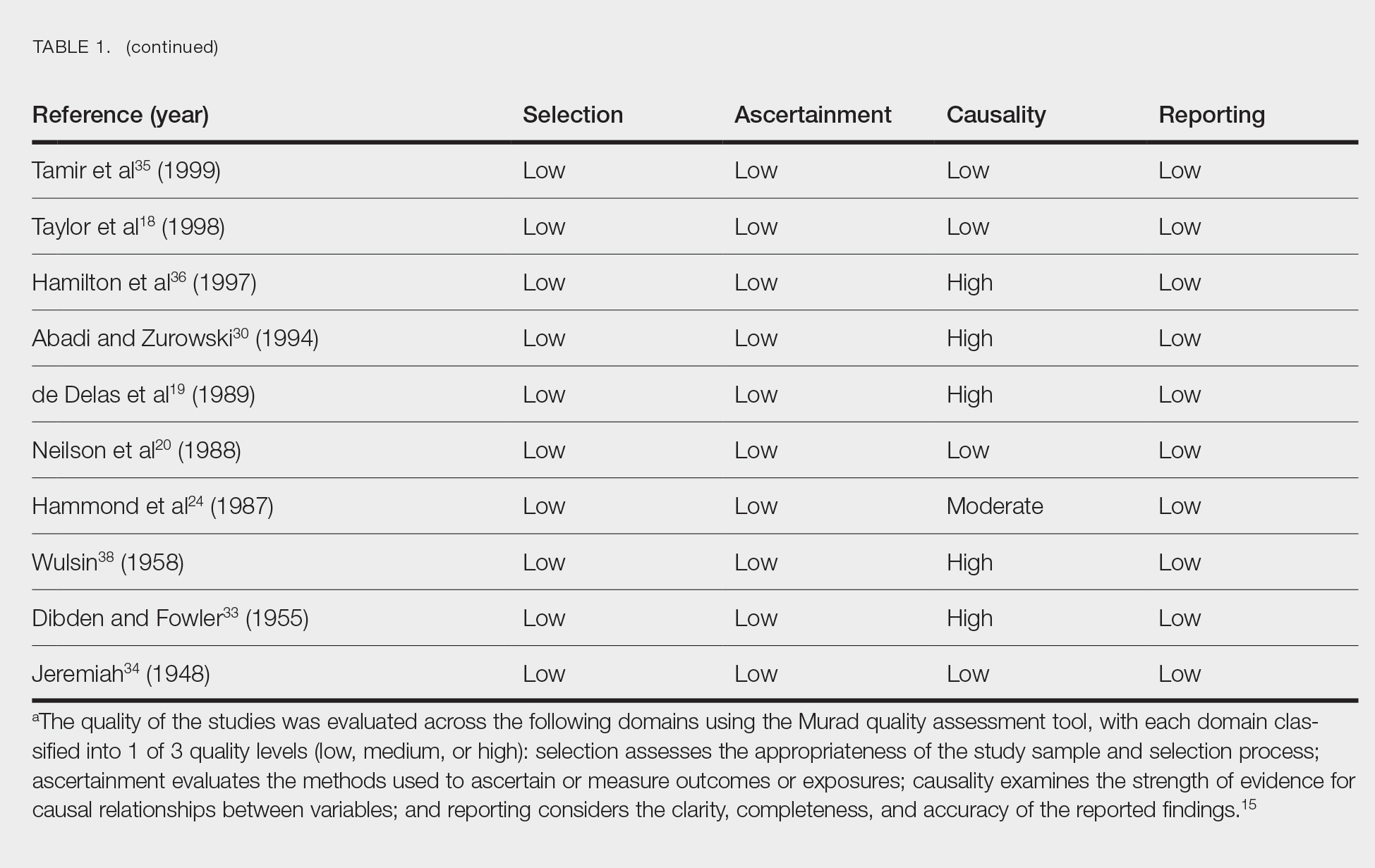
Clinical Characteristics of Included Patients—Our systematic review included 36 patients with a mean age of 63 years and a male to female ratio of 2:1. The 2 most common causes for skin grafting were burn wounds and surgical excision of skin tumors. Most grafts were harvested from the thighs. The development of a solitary lesion on the donor area was reported in two-thirds of the patients, while more than 1 lesion developed in the remaining one-third of patients. The median time to tumor development was 6.5 weeks. In most cases, a split-thickness skin graft was used.
Cutaneous squamous cell carcinomas (CSCCs) were found in 23 patients, with well-differentiated CSCCs in 19 of these cases. Additionally, keratoacanthomas (KAs) were found in 10 patients. The majority of patients underwent surgical excision of the tumor. The median follow-up time was 12 months, during which recurrences were noted in a small percentage of cases. Clinical characteristics of included patients are presented in Table 2.
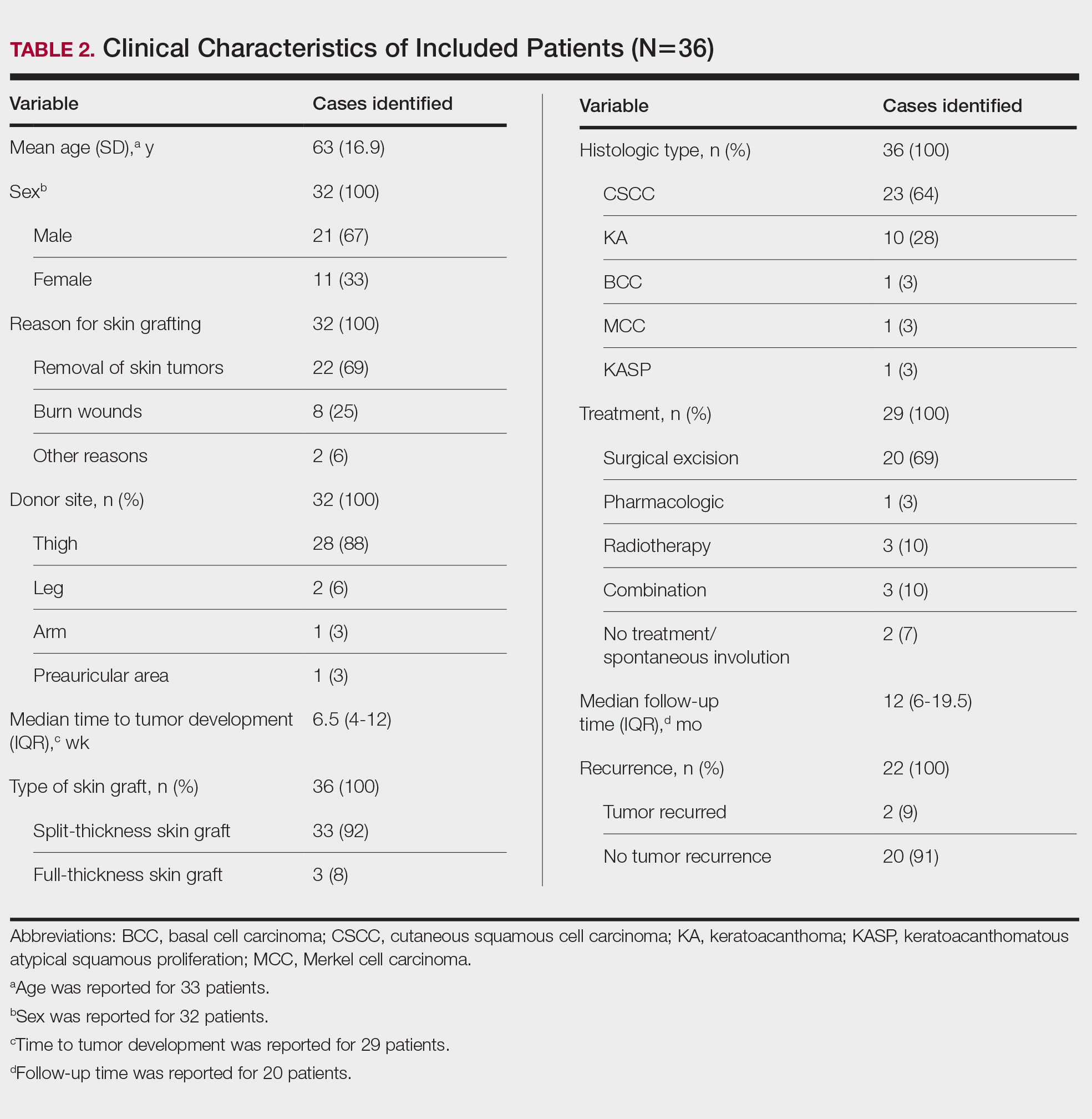
Comment
Reasons for Tumor Development on Skin Graft Donor Sites—The etiology behind epidermal tumor development on graft donor sites is unclear. According to one theory, iatrogenic contamination of the donor site during the removal of a primary epidermal tumor could be responsible. However, contemporary surgical procedures dictate the use of different sets of instruments for separate surgical sites. Moreover, this theory cannot explain the occurrence of epidermal tumors on donor sites in patients who have undergone skin grafting for the repair of burn wounds.37
Another theory suggests that hematogenous and/or lymphatic spread can occur from the site of the primary epidermal tumor to the donor site, which has increased vascularization.16,37 However, this theory also fails to provide an explanation for the development of epidermal tumors in patients who receive skin grafts for burn wounds.
A third theory states that the microenvironment of the donor site is key to tumor development. The donor site undergoes acute inflammation due to the trauma from harvesting the skin graft. According to this theory, acute inflammation could promote neoplastic growth and thus explain the development of epidermal tumors on the donor site.8,26 However, the relationship between acute inflammation and carcinogenesis remains unclear. What is known to date is that the development of CSCC has been documented primarily in chronically inflamed tissues, whereas the development of KA—a variant of CSCC with distinctive and more benign clinical characteristics—can be expected in the setting of acute trauma-related inflammation.13,40,41
Based on our systematic review, we propose that well-differentiated CSCC on graft donor sites might actually be misdiagnosed KA, given that the histopathologic differential diagnosis between CSCC and KA is extremely challenging.42 This hypothesis could explain the development of well-differentiated CSCC and KA on graft donor sites.
Conclusion
Development of CSCC and KA on graft donor sites can be listed among the postoperative complications of autologous skin grafting. Patients and physicians should be aware of this potential complication, and donor sites should be monitored for the occurrence of epidermal tumors.
- Adams DC, Ramsey ML. Grafts in dermatologic surgery: review and update on full- and split-thickness skin grafts, free cartilage grafts, and composite grafts. Dermatologic Surg. 2005;31(8, pt 2):1055-1067. doi:10.1111/j.1524-4725.2005.31831
- Shimizu R, Kishi K. Skin graft. Plast Surg Int. 2012;2012:563493. doi:10.1155/2012/563493
- Reddy S, El-Haddawi F, Fancourt M, et al. The incidence and risk factors for lower limb skin graft failure. Dermatol Res Pract. 2014;2014:582080. doi:10.1155/2014/582080
- Coughlin MJ, Dockery GD, Crawford ME, et al. Lower Extremity Soft Tissue & Cutaneous Plastic Surgery. 2nd ed. Saunders Ltd; 2012.
- Herskovitz I, Hughes OB, Macquhae F, et al. Epidermal skin grafting. Int Wound J. 2016;13(suppl 3):52-56. doi:10.1111/iwj.12631
- Wright H, McKinnell TH, Dunkin C. Recurrence of cutaneous squamous cell carcinoma at remote limb donor site. J Plast Reconstr Aesthet Surg. 2012;65:1265-1266. doi:10.1016/j.bjps.2012.01.022
- Thomas W, Rezzadeh K, Rossi K, et al. Squamous cell carcinoma arising at a skin graft donor site: case report and review of the literature. Plast Surg Case Stud. 2021;7:2513826X211008425. doi:10.1177/2513826X211008425
- Ponnuvelu G, Ng MFY, Connolly CM, et al. Inflammation to skin malignancy, time to rethink the link: SCC in skin graft donor sites. Surgeon. 2011;9:168-169. doi:10.1016/j.surge.2010.08.006
- Noori VJ, Trehan K, Savetamal A, et al. New onset squamous cell carcinoma in previous split-thickness skin graft donor site. Int J Surg. 2018;52:16-19. doi:10.1016/j.ijsu.2018.01.047
- Morritt DG, Khandwala AR. The development of squamous cell carcinomas in split-thickness skin graft donor sites. Eur J Plast Surg. 2013;36:377-380.
- McCormick M, Miotke S. Squamous cell carcinoma at split thickness skin graft donor site: a case report and review of the literature. J Burn Care Res. 2023;44:210-213. doi:10.1093/jbcr/irac137
- Haik J, Georgiou I, Farber N, et al. Squamous cell carcinoma arising in a split-thickness skin graft donor site. Burns. 2008;34:891-893. doi:10.1016/j.burns.2007.06.006
- Elder DE, Massi D, Scolyer RA WR. WHO Classification of Skin Tumours. 4th ed. IARC Press; 2018.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264-269, W64. doi:10.7326/0003-4819-151-4-200908180-00135
- Murad MH, Sultan S, Haffar S, et al. Methodological quality and synthesis of case series and case reports. BMJ. 2018;23:60-63. doi:10.1136/bmjebm-2017-110853
- de Moraes LPB, Burchett I, Nicholls S, et al. Large solitary distant metastasis of cutaneous squamous cell carcinoma to skin graft site with complete response following definitive radiotherapy. Int J Bioautomation. 2017;21:103-108.
- Nagase K, Suzuki Y, Misago N, et al. Acute development of keratoacanthoma at a full-thickness skin graft donor site shortly after surgery. J Dermatol. 2016;43:1232-1233. doi:10.1111/1346-8138.13368
- Taylor CD, Snelling CF, Nickerson D, et al. Acute development of invasive squamous cell carcinoma in a split-thickness skin graft donor site. J Burn Care Rehabil. 1998;19:382-385. doi:10.1097/00004630-199809000-00004
- de Delas J, Leache A, Vazquez Doval J, et al. Keratoacanthoma over the donor site of a laminar skin graft. Med Cutan Ibero Lat Am. 1989;17:225-228.
- Neilson D, Emerson DJ, Dunn L. Squamous cell carcinoma of skin developing in a skin graft donor site. Br J Plast Surg. 1988;41:417-419. doi:10.1016/0007-1226(88)90086-0
- May JT, Patil YJ. Keratoacanthoma-type squamous cell carcinoma developing in a skin graft donor site after tumor extirpation at a distant site. Ear Nose Throat J. 2010;89:E11-E13.
- Imbernón-Moya A, Vargas-Laguna E, Lobato-Berezo A, et al. Simultaneous onset of basal cell carcinoma over skin graft and donor site. JAAD Case Rep. 2015;1:244-246. doi:10.1016/j.jdcr.2015.05.004
- Lee S, Coutts I, Ryan A, et al. Keratoacanthoma formation after skin grafting: a brief report and pathophysiological hypothesis. Australas J Dermatol. 2017;58:e117-e119. doi:10.1111/ajd.12501
- Hammond JS, Thomsen S, Ward CG. Scar carcinoma arising acutelyin a skin graft donor site. J Trauma. 1987;27:681-683. doi:10.1097/00005373-198706000-00017
- Herard C, Arnaud D, Goga D, et al. Rapid onset of squamous cell carcinoma in a thin skin graft donor site. Ann Dermatol Venereol. 2016;143:457-461. doi:10.1016/j.annder.2015.03.027
- Ibrahim A, Moisidis E. Case series: rapidly growing squamous cell carcinoma after cutaneous surgical intervention. JPRAS Open. 2017;14:27-32. doi:10.1016/j.jpra.2017.08.004
- Kearney L, Dolan RT, Parfrey NA, et al. Squamous cell carcinoma arising in a skin graft donor site following melanoma extirpation at a distant site: a case report and review of the literature. JPRAS Open. 2015;3:35-38. doi:10.1016/j.jpra.2015.02.002
- Clark MA, Guitart J, Gerami P, et al. Eruptive keratoacanthomatous atypical squamous proliferations (KASPs) arising in skin graft sites. JAAD Case Rep. 2015;1:274-276. doi:10.1016/j.jdcr.2015.06.009
- Aloraifi F, Mulgrew S, James NK. Secondary Merkel cell carcinoma arising from a graft donor site. J Cutan Med Surg. 2017;21:167-169. doi:10.1177/1203475416676805
- Abadir R, Zurowski S. Case report: squamous cell carcinoma of the skin in both palms, axillary node, donor skin graft site and both soles—associated hyperkeratosis and porokeratosis. Br J Radiol. 1994;67:507-510. doi:10.1259/0007-1285-67-797-507
- Griffiths RW. Keratoacanthoma observed. Br J Plast Surg. 2004;57:485-501. doi:10.1016/j.bjps.2004.05.007
- Marous M, Brady K. Cutaneous squamous cell carcinoma arising in a split thickness skin graft donor site in a patient with systemic lupus erythematosus. Dermatologic Surg. 2021;47:1106-1107. doi:10.1097/DSS.0000000000002955
- Dibden FA, Fowler M. The multiple growth of molluscum sebaceum in donor and recipient sites of skin graft. Aust N Z J Surg. 1955;25:157-159. doi:10.1111/j.1445-2197.1955.tb05122.x
- Jeremiah BS. Squamous cell carcinoma development on donor area following removal of a split thickness skin graft. Plast Reconstr Surg. 1948;3:718-721.
- Tamir G, Morgenstern S, Ben-Amitay D, et al. Synchronous appearance of keratoacanthomas in burn scar and skin graft donor site shortly after injury. J Am Acad Dermatol. 1999;40(5, pt 2):870-871. doi:10.1053/jd.1999.v40.a94419
- Hamilton SA, Dickson WA, O’Brien CJ. Keratoacanthoma developing in a split skin graft donor site. Br J Plast Surg. 1997;50:560-561. doi:10.1016/s0007-1226(97)91308-4
- Hussain A, Ekwobi C, Watson S. Metastatic implantation squamous cell carcinoma in a split-thickness skin graft donor site. J Plast Reconstr Aesthet Surg. 2011;64:690-692. doi:10.1016/j.bjps.2010.06.004
- Wulsin JH. Keratoacanthoma: a benign cutaneous tumors arising in a skin graft donor site. Am Surg. 1958;24:689-692.
- Davis L, Butler D. Acute development of squamous cell carcinoma in a split-thickness skin graft donor site [abstract]. J Am Acad Dermatol. 2012;66:AB208. doi:10.1016/j.jaad.2011.11.874
- Shacter E, Weitzman SA. Chronic inflammation and cancer. Oncology (Williston Park). 2002;16:217-226, 229; discussion 230-232.
- Piotrowski I, Kulcenty K, Suchorska W. Interplay between inflammation and cancer. Reports Pract Oncol Radiother. 2020;25:422-427. doi:10.1016/j.rpor.2020.04.004
- Carr RA, Houghton JP. Histopathologists’ approach to keratoacanthoma: a multisite survey of regional variation in Great Britain and Ireland. J Clin Pathol. 2014;67:637-638. doi:10.1136/jclinpath-2014-202255
Skin grafting is a surgical technique used to cover skin defects resulting from the removal of skin tumors, ulcers, or burn injuries.1-3 Complications can occur at both donor and recipient sites and may include bleeding, hematoma/seroma formation, postoperative pain, infection, scarring, paresthesia, skin pigmentation, graft contracture, and graft failure.1,2,4,5 The development of epidermal tumors is not commonly reported among the complications of skin grafting; however, cases of epidermal tumor development on skin graft donor sites during the postoperative period have been reported.6-12
We performed a systematic review of the literature for cases of epidermal tumor development on skin graft donor sites in patients undergoing autologous skin graft surgery. We present the clinical characteristics of these cases and discuss the nature of these tumors.
Methods
Search Strategy and Study Selection—A literature search was conducted by 2 independent researchers (Z.P. and V.P.) for articles published before December 2022 in the following databases: MEDLINE/PubMed, Web of Science, Scopus, Cochrane Library, OpenGrey, Google Scholar, and WorldCat. Search terms included all possible combinations of the following: keratoacanthoma, molluscum sebaceum, basal cell carcinoma, squamous cell carcinoma, acanthoma, wart, Merkel cell carcinoma, verruca, Bowen disease, keratosis, skin cancer, cutaneous cancer, skin neoplasia, cutaneous neoplasia, and skin tumor. The literature search terms were selected based on the World Health Organization classification of skin tumors.13 Manual bibliography checks were performed on all eligible search results for possible relevant studies. Discrepancies were resolved through discussion and, if needed, mediation by a third researcher (N.C.). To be included, a study had to report a case(s) of epidermal tumor(s) that was confirmed by histopathology and arose on a graft donor site in a patient receiving autologous skin grafts for any reason. No language, geographic, or report date restrictions were set.
Data Extraction, Quality Assessment, and Statistical Analysis—We adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.14 Two independent researchers (Z.P. and V.P.) retrieved the data from the included studies. We have used the terms case and patient interchangeably, and 1 month was measured as 4 weeks for simplicity. Disagreements were resolved by discussion and mediation by a third researcher (N.C.). The quality of the included studies was assessed by 2 researchers (M.P. and V.P.) using the tool proposed by Murad et al.15
We used descriptive statistical analysis to analyze clinical characteristics of the included cases. We performed separate descriptive analyses based on the most frequently reported types of epidermal tumors and compared the differences between different groups using the Mann-Whitney U test, χ2 test, and Fisher exact test. The level of significance was set at P<.05. All statistical analyses were conducted using SPSS (version 29).
Results
Literature Search and Characteristics of Included Studies—The initial literature search identified 1378 studies, which were screened based on title and abstract. After removing duplicate and irrelevant studies and evaluating the full text of eligible studies, 31 studies (4 case series and 27 case reports) were included in the systematic review (Figure).6-12,16-39 Quality assessment of the included studies is presented in Table 1.



Clinical Characteristics of Included Patients—Our systematic review included 36 patients with a mean age of 63 years and a male to female ratio of 2:1. The 2 most common causes for skin grafting were burn wounds and surgical excision of skin tumors. Most grafts were harvested from the thighs. The development of a solitary lesion on the donor area was reported in two-thirds of the patients, while more than 1 lesion developed in the remaining one-third of patients. The median time to tumor development was 6.5 weeks. In most cases, a split-thickness skin graft was used.
Cutaneous squamous cell carcinomas (CSCCs) were found in 23 patients, with well-differentiated CSCCs in 19 of these cases. Additionally, keratoacanthomas (KAs) were found in 10 patients. The majority of patients underwent surgical excision of the tumor. The median follow-up time was 12 months, during which recurrences were noted in a small percentage of cases. Clinical characteristics of included patients are presented in Table 2.


Comment
Reasons for Tumor Development on Skin Graft Donor Sites—The etiology behind epidermal tumor development on graft donor sites is unclear. According to one theory, iatrogenic contamination of the donor site during the removal of a primary epidermal tumor could be responsible. However, contemporary surgical procedures dictate the use of different sets of instruments for separate surgical sites. Moreover, this theory cannot explain the occurrence of epidermal tumors on donor sites in patients who have undergone skin grafting for the repair of burn wounds.37
Another theory suggests that hematogenous and/or lymphatic spread can occur from the site of the primary epidermal tumor to the donor site, which has increased vascularization.16,37 However, this theory also fails to provide an explanation for the development of epidermal tumors in patients who receive skin grafts for burn wounds.
A third theory states that the microenvironment of the donor site is key to tumor development. The donor site undergoes acute inflammation due to the trauma from harvesting the skin graft. According to this theory, acute inflammation could promote neoplastic growth and thus explain the development of epidermal tumors on the donor site.8,26 However, the relationship between acute inflammation and carcinogenesis remains unclear. What is known to date is that the development of CSCC has been documented primarily in chronically inflamed tissues, whereas the development of KA—a variant of CSCC with distinctive and more benign clinical characteristics—can be expected in the setting of acute trauma-related inflammation.13,40,41
Based on our systematic review, we propose that well-differentiated CSCC on graft donor sites might actually be misdiagnosed KA, given that the histopathologic differential diagnosis between CSCC and KA is extremely challenging.42 This hypothesis could explain the development of well-differentiated CSCC and KA on graft donor sites.
Conclusion
Development of CSCC and KA on graft donor sites can be listed among the postoperative complications of autologous skin grafting. Patients and physicians should be aware of this potential complication, and donor sites should be monitored for the occurrence of epidermal tumors.
Skin grafting is a surgical technique used to cover skin defects resulting from the removal of skin tumors, ulcers, or burn injuries.1-3 Complications can occur at both donor and recipient sites and may include bleeding, hematoma/seroma formation, postoperative pain, infection, scarring, paresthesia, skin pigmentation, graft contracture, and graft failure.1,2,4,5 The development of epidermal tumors is not commonly reported among the complications of skin grafting; however, cases of epidermal tumor development on skin graft donor sites during the postoperative period have been reported.6-12
We performed a systematic review of the literature for cases of epidermal tumor development on skin graft donor sites in patients undergoing autologous skin graft surgery. We present the clinical characteristics of these cases and discuss the nature of these tumors.
Methods
Search Strategy and Study Selection—A literature search was conducted by 2 independent researchers (Z.P. and V.P.) for articles published before December 2022 in the following databases: MEDLINE/PubMed, Web of Science, Scopus, Cochrane Library, OpenGrey, Google Scholar, and WorldCat. Search terms included all possible combinations of the following: keratoacanthoma, molluscum sebaceum, basal cell carcinoma, squamous cell carcinoma, acanthoma, wart, Merkel cell carcinoma, verruca, Bowen disease, keratosis, skin cancer, cutaneous cancer, skin neoplasia, cutaneous neoplasia, and skin tumor. The literature search terms were selected based on the World Health Organization classification of skin tumors.13 Manual bibliography checks were performed on all eligible search results for possible relevant studies. Discrepancies were resolved through discussion and, if needed, mediation by a third researcher (N.C.). To be included, a study had to report a case(s) of epidermal tumor(s) that was confirmed by histopathology and arose on a graft donor site in a patient receiving autologous skin grafts for any reason. No language, geographic, or report date restrictions were set.
Data Extraction, Quality Assessment, and Statistical Analysis—We adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.14 Two independent researchers (Z.P. and V.P.) retrieved the data from the included studies. We have used the terms case and patient interchangeably, and 1 month was measured as 4 weeks for simplicity. Disagreements were resolved by discussion and mediation by a third researcher (N.C.). The quality of the included studies was assessed by 2 researchers (M.P. and V.P.) using the tool proposed by Murad et al.15
We used descriptive statistical analysis to analyze clinical characteristics of the included cases. We performed separate descriptive analyses based on the most frequently reported types of epidermal tumors and compared the differences between different groups using the Mann-Whitney U test, χ2 test, and Fisher exact test. The level of significance was set at P<.05. All statistical analyses were conducted using SPSS (version 29).
Results
Literature Search and Characteristics of Included Studies—The initial literature search identified 1378 studies, which were screened based on title and abstract. After removing duplicate and irrelevant studies and evaluating the full text of eligible studies, 31 studies (4 case series and 27 case reports) were included in the systematic review (Figure).6-12,16-39 Quality assessment of the included studies is presented in Table 1.



Clinical Characteristics of Included Patients—Our systematic review included 36 patients with a mean age of 63 years and a male to female ratio of 2:1. The 2 most common causes for skin grafting were burn wounds and surgical excision of skin tumors. Most grafts were harvested from the thighs. The development of a solitary lesion on the donor area was reported in two-thirds of the patients, while more than 1 lesion developed in the remaining one-third of patients. The median time to tumor development was 6.5 weeks. In most cases, a split-thickness skin graft was used.
Cutaneous squamous cell carcinomas (CSCCs) were found in 23 patients, with well-differentiated CSCCs in 19 of these cases. Additionally, keratoacanthomas (KAs) were found in 10 patients. The majority of patients underwent surgical excision of the tumor. The median follow-up time was 12 months, during which recurrences were noted in a small percentage of cases. Clinical characteristics of included patients are presented in Table 2.


Comment
Reasons for Tumor Development on Skin Graft Donor Sites—The etiology behind epidermal tumor development on graft donor sites is unclear. According to one theory, iatrogenic contamination of the donor site during the removal of a primary epidermal tumor could be responsible. However, contemporary surgical procedures dictate the use of different sets of instruments for separate surgical sites. Moreover, this theory cannot explain the occurrence of epidermal tumors on donor sites in patients who have undergone skin grafting for the repair of burn wounds.37
Another theory suggests that hematogenous and/or lymphatic spread can occur from the site of the primary epidermal tumor to the donor site, which has increased vascularization.16,37 However, this theory also fails to provide an explanation for the development of epidermal tumors in patients who receive skin grafts for burn wounds.
A third theory states that the microenvironment of the donor site is key to tumor development. The donor site undergoes acute inflammation due to the trauma from harvesting the skin graft. According to this theory, acute inflammation could promote neoplastic growth and thus explain the development of epidermal tumors on the donor site.8,26 However, the relationship between acute inflammation and carcinogenesis remains unclear. What is known to date is that the development of CSCC has been documented primarily in chronically inflamed tissues, whereas the development of KA—a variant of CSCC with distinctive and more benign clinical characteristics—can be expected in the setting of acute trauma-related inflammation.13,40,41
Based on our systematic review, we propose that well-differentiated CSCC on graft donor sites might actually be misdiagnosed KA, given that the histopathologic differential diagnosis between CSCC and KA is extremely challenging.42 This hypothesis could explain the development of well-differentiated CSCC and KA on graft donor sites.
Conclusion
Development of CSCC and KA on graft donor sites can be listed among the postoperative complications of autologous skin grafting. Patients and physicians should be aware of this potential complication, and donor sites should be monitored for the occurrence of epidermal tumors.
- Adams DC, Ramsey ML. Grafts in dermatologic surgery: review and update on full- and split-thickness skin grafts, free cartilage grafts, and composite grafts. Dermatologic Surg. 2005;31(8, pt 2):1055-1067. doi:10.1111/j.1524-4725.2005.31831
- Shimizu R, Kishi K. Skin graft. Plast Surg Int. 2012;2012:563493. doi:10.1155/2012/563493
- Reddy S, El-Haddawi F, Fancourt M, et al. The incidence and risk factors for lower limb skin graft failure. Dermatol Res Pract. 2014;2014:582080. doi:10.1155/2014/582080
- Coughlin MJ, Dockery GD, Crawford ME, et al. Lower Extremity Soft Tissue & Cutaneous Plastic Surgery. 2nd ed. Saunders Ltd; 2012.
- Herskovitz I, Hughes OB, Macquhae F, et al. Epidermal skin grafting. Int Wound J. 2016;13(suppl 3):52-56. doi:10.1111/iwj.12631
- Wright H, McKinnell TH, Dunkin C. Recurrence of cutaneous squamous cell carcinoma at remote limb donor site. J Plast Reconstr Aesthet Surg. 2012;65:1265-1266. doi:10.1016/j.bjps.2012.01.022
- Thomas W, Rezzadeh K, Rossi K, et al. Squamous cell carcinoma arising at a skin graft donor site: case report and review of the literature. Plast Surg Case Stud. 2021;7:2513826X211008425. doi:10.1177/2513826X211008425
- Ponnuvelu G, Ng MFY, Connolly CM, et al. Inflammation to skin malignancy, time to rethink the link: SCC in skin graft donor sites. Surgeon. 2011;9:168-169. doi:10.1016/j.surge.2010.08.006
- Noori VJ, Trehan K, Savetamal A, et al. New onset squamous cell carcinoma in previous split-thickness skin graft donor site. Int J Surg. 2018;52:16-19. doi:10.1016/j.ijsu.2018.01.047
- Morritt DG, Khandwala AR. The development of squamous cell carcinomas in split-thickness skin graft donor sites. Eur J Plast Surg. 2013;36:377-380.
- McCormick M, Miotke S. Squamous cell carcinoma at split thickness skin graft donor site: a case report and review of the literature. J Burn Care Res. 2023;44:210-213. doi:10.1093/jbcr/irac137
- Haik J, Georgiou I, Farber N, et al. Squamous cell carcinoma arising in a split-thickness skin graft donor site. Burns. 2008;34:891-893. doi:10.1016/j.burns.2007.06.006
- Elder DE, Massi D, Scolyer RA WR. WHO Classification of Skin Tumours. 4th ed. IARC Press; 2018.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264-269, W64. doi:10.7326/0003-4819-151-4-200908180-00135
- Murad MH, Sultan S, Haffar S, et al. Methodological quality and synthesis of case series and case reports. BMJ. 2018;23:60-63. doi:10.1136/bmjebm-2017-110853
- de Moraes LPB, Burchett I, Nicholls S, et al. Large solitary distant metastasis of cutaneous squamous cell carcinoma to skin graft site with complete response following definitive radiotherapy. Int J Bioautomation. 2017;21:103-108.
- Nagase K, Suzuki Y, Misago N, et al. Acute development of keratoacanthoma at a full-thickness skin graft donor site shortly after surgery. J Dermatol. 2016;43:1232-1233. doi:10.1111/1346-8138.13368
- Taylor CD, Snelling CF, Nickerson D, et al. Acute development of invasive squamous cell carcinoma in a split-thickness skin graft donor site. J Burn Care Rehabil. 1998;19:382-385. doi:10.1097/00004630-199809000-00004
- de Delas J, Leache A, Vazquez Doval J, et al. Keratoacanthoma over the donor site of a laminar skin graft. Med Cutan Ibero Lat Am. 1989;17:225-228.
- Neilson D, Emerson DJ, Dunn L. Squamous cell carcinoma of skin developing in a skin graft donor site. Br J Plast Surg. 1988;41:417-419. doi:10.1016/0007-1226(88)90086-0
- May JT, Patil YJ. Keratoacanthoma-type squamous cell carcinoma developing in a skin graft donor site after tumor extirpation at a distant site. Ear Nose Throat J. 2010;89:E11-E13.
- Imbernón-Moya A, Vargas-Laguna E, Lobato-Berezo A, et al. Simultaneous onset of basal cell carcinoma over skin graft and donor site. JAAD Case Rep. 2015;1:244-246. doi:10.1016/j.jdcr.2015.05.004
- Lee S, Coutts I, Ryan A, et al. Keratoacanthoma formation after skin grafting: a brief report and pathophysiological hypothesis. Australas J Dermatol. 2017;58:e117-e119. doi:10.1111/ajd.12501
- Hammond JS, Thomsen S, Ward CG. Scar carcinoma arising acutelyin a skin graft donor site. J Trauma. 1987;27:681-683. doi:10.1097/00005373-198706000-00017
- Herard C, Arnaud D, Goga D, et al. Rapid onset of squamous cell carcinoma in a thin skin graft donor site. Ann Dermatol Venereol. 2016;143:457-461. doi:10.1016/j.annder.2015.03.027
- Ibrahim A, Moisidis E. Case series: rapidly growing squamous cell carcinoma after cutaneous surgical intervention. JPRAS Open. 2017;14:27-32. doi:10.1016/j.jpra.2017.08.004
- Kearney L, Dolan RT, Parfrey NA, et al. Squamous cell carcinoma arising in a skin graft donor site following melanoma extirpation at a distant site: a case report and review of the literature. JPRAS Open. 2015;3:35-38. doi:10.1016/j.jpra.2015.02.002
- Clark MA, Guitart J, Gerami P, et al. Eruptive keratoacanthomatous atypical squamous proliferations (KASPs) arising in skin graft sites. JAAD Case Rep. 2015;1:274-276. doi:10.1016/j.jdcr.2015.06.009
- Aloraifi F, Mulgrew S, James NK. Secondary Merkel cell carcinoma arising from a graft donor site. J Cutan Med Surg. 2017;21:167-169. doi:10.1177/1203475416676805
- Abadir R, Zurowski S. Case report: squamous cell carcinoma of the skin in both palms, axillary node, donor skin graft site and both soles—associated hyperkeratosis and porokeratosis. Br J Radiol. 1994;67:507-510. doi:10.1259/0007-1285-67-797-507
- Griffiths RW. Keratoacanthoma observed. Br J Plast Surg. 2004;57:485-501. doi:10.1016/j.bjps.2004.05.007
- Marous M, Brady K. Cutaneous squamous cell carcinoma arising in a split thickness skin graft donor site in a patient with systemic lupus erythematosus. Dermatologic Surg. 2021;47:1106-1107. doi:10.1097/DSS.0000000000002955
- Dibden FA, Fowler M. The multiple growth of molluscum sebaceum in donor and recipient sites of skin graft. Aust N Z J Surg. 1955;25:157-159. doi:10.1111/j.1445-2197.1955.tb05122.x
- Jeremiah BS. Squamous cell carcinoma development on donor area following removal of a split thickness skin graft. Plast Reconstr Surg. 1948;3:718-721.
- Tamir G, Morgenstern S, Ben-Amitay D, et al. Synchronous appearance of keratoacanthomas in burn scar and skin graft donor site shortly after injury. J Am Acad Dermatol. 1999;40(5, pt 2):870-871. doi:10.1053/jd.1999.v40.a94419
- Hamilton SA, Dickson WA, O’Brien CJ. Keratoacanthoma developing in a split skin graft donor site. Br J Plast Surg. 1997;50:560-561. doi:10.1016/s0007-1226(97)91308-4
- Hussain A, Ekwobi C, Watson S. Metastatic implantation squamous cell carcinoma in a split-thickness skin graft donor site. J Plast Reconstr Aesthet Surg. 2011;64:690-692. doi:10.1016/j.bjps.2010.06.004
- Wulsin JH. Keratoacanthoma: a benign cutaneous tumors arising in a skin graft donor site. Am Surg. 1958;24:689-692.
- Davis L, Butler D. Acute development of squamous cell carcinoma in a split-thickness skin graft donor site [abstract]. J Am Acad Dermatol. 2012;66:AB208. doi:10.1016/j.jaad.2011.11.874
- Shacter E, Weitzman SA. Chronic inflammation and cancer. Oncology (Williston Park). 2002;16:217-226, 229; discussion 230-232.
- Piotrowski I, Kulcenty K, Suchorska W. Interplay between inflammation and cancer. Reports Pract Oncol Radiother. 2020;25:422-427. doi:10.1016/j.rpor.2020.04.004
- Carr RA, Houghton JP. Histopathologists’ approach to keratoacanthoma: a multisite survey of regional variation in Great Britain and Ireland. J Clin Pathol. 2014;67:637-638. doi:10.1136/jclinpath-2014-202255
- Adams DC, Ramsey ML. Grafts in dermatologic surgery: review and update on full- and split-thickness skin grafts, free cartilage grafts, and composite grafts. Dermatologic Surg. 2005;31(8, pt 2):1055-1067. doi:10.1111/j.1524-4725.2005.31831
- Shimizu R, Kishi K. Skin graft. Plast Surg Int. 2012;2012:563493. doi:10.1155/2012/563493
- Reddy S, El-Haddawi F, Fancourt M, et al. The incidence and risk factors for lower limb skin graft failure. Dermatol Res Pract. 2014;2014:582080. doi:10.1155/2014/582080
- Coughlin MJ, Dockery GD, Crawford ME, et al. Lower Extremity Soft Tissue & Cutaneous Plastic Surgery. 2nd ed. Saunders Ltd; 2012.
- Herskovitz I, Hughes OB, Macquhae F, et al. Epidermal skin grafting. Int Wound J. 2016;13(suppl 3):52-56. doi:10.1111/iwj.12631
- Wright H, McKinnell TH, Dunkin C. Recurrence of cutaneous squamous cell carcinoma at remote limb donor site. J Plast Reconstr Aesthet Surg. 2012;65:1265-1266. doi:10.1016/j.bjps.2012.01.022
- Thomas W, Rezzadeh K, Rossi K, et al. Squamous cell carcinoma arising at a skin graft donor site: case report and review of the literature. Plast Surg Case Stud. 2021;7:2513826X211008425. doi:10.1177/2513826X211008425
- Ponnuvelu G, Ng MFY, Connolly CM, et al. Inflammation to skin malignancy, time to rethink the link: SCC in skin graft donor sites. Surgeon. 2011;9:168-169. doi:10.1016/j.surge.2010.08.006
- Noori VJ, Trehan K, Savetamal A, et al. New onset squamous cell carcinoma in previous split-thickness skin graft donor site. Int J Surg. 2018;52:16-19. doi:10.1016/j.ijsu.2018.01.047
- Morritt DG, Khandwala AR. The development of squamous cell carcinomas in split-thickness skin graft donor sites. Eur J Plast Surg. 2013;36:377-380.
- McCormick M, Miotke S. Squamous cell carcinoma at split thickness skin graft donor site: a case report and review of the literature. J Burn Care Res. 2023;44:210-213. doi:10.1093/jbcr/irac137
- Haik J, Georgiou I, Farber N, et al. Squamous cell carcinoma arising in a split-thickness skin graft donor site. Burns. 2008;34:891-893. doi:10.1016/j.burns.2007.06.006
- Elder DE, Massi D, Scolyer RA WR. WHO Classification of Skin Tumours. 4th ed. IARC Press; 2018.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264-269, W64. doi:10.7326/0003-4819-151-4-200908180-00135
- Murad MH, Sultan S, Haffar S, et al. Methodological quality and synthesis of case series and case reports. BMJ. 2018;23:60-63. doi:10.1136/bmjebm-2017-110853
- de Moraes LPB, Burchett I, Nicholls S, et al. Large solitary distant metastasis of cutaneous squamous cell carcinoma to skin graft site with complete response following definitive radiotherapy. Int J Bioautomation. 2017;21:103-108.
- Nagase K, Suzuki Y, Misago N, et al. Acute development of keratoacanthoma at a full-thickness skin graft donor site shortly after surgery. J Dermatol. 2016;43:1232-1233. doi:10.1111/1346-8138.13368
- Taylor CD, Snelling CF, Nickerson D, et al. Acute development of invasive squamous cell carcinoma in a split-thickness skin graft donor site. J Burn Care Rehabil. 1998;19:382-385. doi:10.1097/00004630-199809000-00004
- de Delas J, Leache A, Vazquez Doval J, et al. Keratoacanthoma over the donor site of a laminar skin graft. Med Cutan Ibero Lat Am. 1989;17:225-228.
- Neilson D, Emerson DJ, Dunn L. Squamous cell carcinoma of skin developing in a skin graft donor site. Br J Plast Surg. 1988;41:417-419. doi:10.1016/0007-1226(88)90086-0
- May JT, Patil YJ. Keratoacanthoma-type squamous cell carcinoma developing in a skin graft donor site after tumor extirpation at a distant site. Ear Nose Throat J. 2010;89:E11-E13.
- Imbernón-Moya A, Vargas-Laguna E, Lobato-Berezo A, et al. Simultaneous onset of basal cell carcinoma over skin graft and donor site. JAAD Case Rep. 2015;1:244-246. doi:10.1016/j.jdcr.2015.05.004
- Lee S, Coutts I, Ryan A, et al. Keratoacanthoma formation after skin grafting: a brief report and pathophysiological hypothesis. Australas J Dermatol. 2017;58:e117-e119. doi:10.1111/ajd.12501
- Hammond JS, Thomsen S, Ward CG. Scar carcinoma arising acutelyin a skin graft donor site. J Trauma. 1987;27:681-683. doi:10.1097/00005373-198706000-00017
- Herard C, Arnaud D, Goga D, et al. Rapid onset of squamous cell carcinoma in a thin skin graft donor site. Ann Dermatol Venereol. 2016;143:457-461. doi:10.1016/j.annder.2015.03.027
- Ibrahim A, Moisidis E. Case series: rapidly growing squamous cell carcinoma after cutaneous surgical intervention. JPRAS Open. 2017;14:27-32. doi:10.1016/j.jpra.2017.08.004
- Kearney L, Dolan RT, Parfrey NA, et al. Squamous cell carcinoma arising in a skin graft donor site following melanoma extirpation at a distant site: a case report and review of the literature. JPRAS Open. 2015;3:35-38. doi:10.1016/j.jpra.2015.02.002
- Clark MA, Guitart J, Gerami P, et al. Eruptive keratoacanthomatous atypical squamous proliferations (KASPs) arising in skin graft sites. JAAD Case Rep. 2015;1:274-276. doi:10.1016/j.jdcr.2015.06.009
- Aloraifi F, Mulgrew S, James NK. Secondary Merkel cell carcinoma arising from a graft donor site. J Cutan Med Surg. 2017;21:167-169. doi:10.1177/1203475416676805
- Abadir R, Zurowski S. Case report: squamous cell carcinoma of the skin in both palms, axillary node, donor skin graft site and both soles—associated hyperkeratosis and porokeratosis. Br J Radiol. 1994;67:507-510. doi:10.1259/0007-1285-67-797-507
- Griffiths RW. Keratoacanthoma observed. Br J Plast Surg. 2004;57:485-501. doi:10.1016/j.bjps.2004.05.007
- Marous M, Brady K. Cutaneous squamous cell carcinoma arising in a split thickness skin graft donor site in a patient with systemic lupus erythematosus. Dermatologic Surg. 2021;47:1106-1107. doi:10.1097/DSS.0000000000002955
- Dibden FA, Fowler M. The multiple growth of molluscum sebaceum in donor and recipient sites of skin graft. Aust N Z J Surg. 1955;25:157-159. doi:10.1111/j.1445-2197.1955.tb05122.x
- Jeremiah BS. Squamous cell carcinoma development on donor area following removal of a split thickness skin graft. Plast Reconstr Surg. 1948;3:718-721.
- Tamir G, Morgenstern S, Ben-Amitay D, et al. Synchronous appearance of keratoacanthomas in burn scar and skin graft donor site shortly after injury. J Am Acad Dermatol. 1999;40(5, pt 2):870-871. doi:10.1053/jd.1999.v40.a94419
- Hamilton SA, Dickson WA, O’Brien CJ. Keratoacanthoma developing in a split skin graft donor site. Br J Plast Surg. 1997;50:560-561. doi:10.1016/s0007-1226(97)91308-4
- Hussain A, Ekwobi C, Watson S. Metastatic implantation squamous cell carcinoma in a split-thickness skin graft donor site. J Plast Reconstr Aesthet Surg. 2011;64:690-692. doi:10.1016/j.bjps.2010.06.004
- Wulsin JH. Keratoacanthoma: a benign cutaneous tumors arising in a skin graft donor site. Am Surg. 1958;24:689-692.
- Davis L, Butler D. Acute development of squamous cell carcinoma in a split-thickness skin graft donor site [abstract]. J Am Acad Dermatol. 2012;66:AB208. doi:10.1016/j.jaad.2011.11.874
- Shacter E, Weitzman SA. Chronic inflammation and cancer. Oncology (Williston Park). 2002;16:217-226, 229; discussion 230-232.
- Piotrowski I, Kulcenty K, Suchorska W. Interplay between inflammation and cancer. Reports Pract Oncol Radiother. 2020;25:422-427. doi:10.1016/j.rpor.2020.04.004
- Carr RA, Houghton JP. Histopathologists’ approach to keratoacanthoma: a multisite survey of regional variation in Great Britain and Ireland. J Clin Pathol. 2014;67:637-638. doi:10.1136/jclinpath-2014-202255
Practice Points
- Donor site cutaneous squamous cell carcinoma (CSCC) and keratoacanthoma (KA) can be postoperative complications of autologous skin grafting.
- Surgical excision of donor site CSCC and KA typically is curative.
Obesity: The Basics
Editor's Note: This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Editor's Note: This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Editor's Note: This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Impact of a Pharmacist-Led Emergency Department Urinary Tract Infection Aftercare Program
The emergency department (ED) is estimated to provide half of all medical care in the United States, serving as a conduit between ambulatory care and inpatient settings.1 According to the Centers for Disease Control and Prevention, around 11 million antibiotic prescriptions were written in EDs in 2021.2 A previous study conducted at a US Department of Veterans (VA) Affairs medical center found that about 40% of all antimicrobial use in the ED was inappropriate.3 The ED is a critical and high-yield space for antimicrobial stewardship efforts.4
Urinary tract infections (UTIs) are one of the most common reasons for ED visits.4 In 2018, there were about 3 million UTI discharge diagnoses reported in the US.5 Diagnosis and management of UTIs can vary depending on patient sex, upper or lower urinary tract involvement, and the severity of the infection.6 Most UTIs are uncomplicated and can be safely treated with oral antibiotics at home; however, if mismanaged, they can lead to increased morbidity and mortality.6
Antimicrobial prescribing in the ED is predominantly empiric with challenges such as diverse patient needs, rising antimicrobial resistance, and limited microbiologic data at the time of discharge.6 The lack of a standardized process for urine culture follow-up after discharge represents another major complicating factor in the outpatient management of UTIs. Studies have shown that ED pharmacists play a vital role in providing quality follow-up care by optimizing antimicrobial use, resulting in improved patient outcomes in various infectious syndromes, including UTIs.7-13
Program Description
In June 2021, the VA Greater Los Angeles Healthcare System (VAGLAHS) piloted an ED pharmacist-led aftercare program to optimize postdischarge antimicrobial therapy management of UTIs. After a patient is discharged from the ED, the clinical pharmacist reviews urine culture results, interprets available antimicrobial susceptibility, conducts patient interviews, adjusts for patient-specific factors, and addresses potential antibiotic-associated adverse events. The ED pharmacist is then responsible for managing therapy changes in consultation with an ED health care practitioner (HCP).
Methods
This single center, retrospective chart review included veterans who were discharged with an oral antibiotic for UTI treatment from the VAGLAHS ED and evaluated by clinical pharmacists between June 1, 2021, and June 30, 2022. For patients with multiple ED visits, only the initial ED encounter was reviewed. Patients were excluded if they had a complicated UTI diagnosis requiring intravenous antibiotics or if they were admitted to the hospital. Data were generated through the Corporate Data Warehouse by VAGLAHS Pharmacy Informatics Service. Each patient was assigned a random number using the Microsoft Excel formula =RAND( ) and then sorted in chronological order to ensure randomization at baseline prior to data collection.
The primary aim of this quality improvement project was to characterize the impact of ED pharmacist-led interventions by evaluating the proportion of empiric to targeted therapy adjustments, antibiotic therapy discontinuation, and unmodified index treatment. The secondary objectives evaluated time to ED pharmacist aftercare follow-up, days of antibiotic exposure avoided, 30-day ED visits related to a urinary source, and transition of care documentation. Descriptive statistics were performed; median and IQR were calculated in Microsoft Excel.
Results
A total of 548 ED UTI encounters were identified, including 449 patients with an index ED UTI aftercare follow-up evaluation. Of the 246 randomly screened patients, 200 veterans met inclusion criteria. The median age of included patients was 73 years and most (83.0%) were male (Table 1). One hundred thirty-two patients (66.0%) had a cystitis diagnosis, followed by complicated UTI (14.0%) and catheter-associated UTI (11.0%). The most frequently isolated uropathogen was Escherichia coli (30.5%). ß-lactams were prescribed for empiric treatment to 121 patients (60.5%), followed by 36 fluoroquinolones prescriptions (18.0%). The median treatment duration was 7 days.
The median time to ED pharmacist UTI aftercare evaluation was 2 days (Table 2). Sixty-seven cases required pharmacist intervention, which included 34 transitions to targeted therapy (17.0%) and 33 antibiotic discontinuations (16.5%). A total of 144 days of antibiotic exposure was avoided (ie, days antibiotic was prescribed minus days therapy administered). The majority of cases without modification to index therapy were due to appropriate empiric treatment selection (49.0%). Twelve (6.0%) patients had a subsequent urinary-related ED visit within 30 days due to 8 cases of persistent and/or worsening urinary symptoms (66.7%) and 2 cases of recurrent UTI (16.7%).
Discussion
Outpatient antibiotic prescribing for UTI management in the ED is challenging due to the absence of microbiologic data at time of diagnosis and lack of consistent transition of care follow-up.6 The VAGLAHS ED UTI aftercare program piloted a pharmacist-driven protocol for review of all urine cultures and optimization of antibiotic therapy.
Most ED UTI discharges that did not require pharmacist intervention had empiric treatment selection active against the clinical isolates. This suggests that the ED prescribing practices concur with theVAGLAHS antibiogram and treatment guidelines. Clinical pharmacists intervened in about one-third of UTI cases, which included modification or discontinuation of therapy. Further review of these cases demonstrated that about half of those with a subsequent 30-day ED visit related to a urinary source had therapy modification. Most patients with a 30-day ED visit had persistent and/or worsening urinary symptoms, prompting further exploratory workup.
Although this project did not evaluate time from urine culture results to aftercare review, the VAGLAHS ED pharmacists had a median follow-up time of 48 hours. This timeline mirrors the typical duration for urine culture results, suggesting that the pilot program allowed for real time pharmacist review and intervention. Consequently, this initiative resulted in the avoidance of 144 unnecessary days of antibiotic exposure.
While the current protocol highlights the work that ED pharmacists provide postdischarge, there are additional opportunities for pharmacist intervention. For example, one-third of these clinical encounters were completed without HCP notification, indicating an ongoing need to ensure continuity of care. Additionally, all 16 patients diagnosed with asymptomatic bacteriuria were discharged with an oral antibiotic, highlighting an opportunity to further optimize antibiotic prescribing prior to discharge. ED pharmacists continue to play an important role in mitigating inappropriate and unnecessary antibiotic use, which will reduce antibiotic-related adverse drug reactions, Clostridioides difficile infection, and antimicrobial resistance.
Limitations
Inconsistent and incomplete documentation of clinical data in the electronic health record made the characterization of patient encounters challenging. Furthermore, ED HCPs varying clinical practices may have impacted the heterogeneity of UTI diagnosis and management at VAGLAHS.
Conclusions
Implementation of an ED pharmacist-driven UTI aftercare program at VAGLAHS reduced unnecessary antimicrobial exposure, improved antibiotic management, and ensured continuity of care postdischarge. Findings from our project implicate possible future pharmacist involvement predischarge, such as targeting inappropriate asymptomatic bacteriuria treatment.14-16 This pilot program suggested the feasibility of integrating antimicrobial stewardship practices within the ED setting in an ongoing effort to improve the quality of care for veterans.
1. Marcozzi D, Carr B, Liferidge A, Baehr N, Browne B.. Trends in the contribution of emergency departments to the provision of hospital-associated health care in the USA. Int J Health Serv. 2018;48(2):267–288. doi:10.1177/0020731417734498
2. Centers for Disease Control and Prevention. Outpatient antibiotic prescriptions — United States, 2021. Updated October 4, 2022. Accessed May 22, 2024. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/antibiotic-use/data/report-2021.html
3. Timbrook TT, Caffrey AR, Ovalle A, et al. Assessments of opportunities to improve antibiotic prescribing in an emergency department: a period prevalence survey. Infect Dis Ther. 2017;6(4):497-505. doi:10.1007/s40121-017-0175-9
4. Pulia M, Redwood R, May L. Antimicrobial stewardship in the emergency department. Emerg Med Clin North. 2018;36(4):853-872. doi:10.1016/j.emc.2018.06.012
5. Weiss A, Jiang H. Most frequent reasons for emergency department visits, 2018. December 16, 2021. Accessed May 22, 2024. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb286-ED-Frequent-Conditions-2018.pdf
6. Abrahamian FM, Moran GJ, Talan DA. Urinary tract infections in the emergency department. Infect Dis Clin North Am. 2008;22(1):73-87. doi:10.1016/j.idc.2007.10.002
7. Dumkow LE, Kenney RM, MacDonald NC, Carreno JJ, Malhotra MK, Davis SL. Impact of a multidisciplinary culture follow-up program of antimicrobial therapy in the emergency department. Infect Dis Ther. 2014;3(1):45-53. doi:10.1007/s40121-014-0026-x
8. Davis LC, Covey RB, Weston JS, Hu BB, Laine GA. Pharmacist-driven antimicrobial optimization in the emergency department. Am J Health Syst Pharm. 2016;73(5 Suppl 1):S49-S56. doi:10.2146/sp150036
9. Lingenfelter E, Darkin Z, Fritz K, Youngquist S, Madsen T, Fix M. ED pharmacist monitoring of provider antibiotic selection aids appropriate treatment for outpatient UTI. Am J Emerg Med. 2016;34(8):1600-1603. doi:10.1016/j.ajem.2016.05.076
10. Zhang X, Rowan N, Pflugeisen BM, Alajbegovic S. Urine culture guided antibiotic interventions: a pharmacist driven antimicrobial stewardship effort in the ED. Am J Emerg Med. 2017;35(4):594-598. doi:10.1016/j.ajem.2016.12.036
11. Percival KM, Valenti KM, Schmittling SE, Strader BD, Lopez RR, Bergman SJ. Impact of an antimicrobial stewardship intervention on urinary tract infection treatment in the ED. Am J Emerg Med. 2015;33(9):1129-1133. doi:10.1016/j.ajem.2015.04.067
12. Almulhim AS, Aldayyen A, Yenina K, Chiappini A, Khan TM. Optimization of antibiotic selection in the emergency department for urine culture follow ups, a retrospective pre-post intervention study: clinical pharmacist efforts. J Pharm Policy Pract. 2019;12(1):8. Published online April 9, 2019. doi:10.1186/s40545-019-0168-z
13. Stoll K, Feltz E, Ebert S. Pharmacist-driven implementation of outpatient antibiotic prescribing algorithms improves guideline adherence in the emergency department. J Pharm Pract. 2021;34(6):875-881. doi:10.1177/0897190020930979
14. Petty LA, Vaughn VM, Flanders SA, et al. Assessment of testing and treatment of asymptomatic bacteriuria initiated in the emergency department. Open Forum Infect Dis. 2020;7(12):ofaa537. Published online November 3, 2020. doi:10.1093/ofid/ofaa537
15. Ingalls EM, Veillette JJ, Olson J, et al. Impact of a multifaceted intervention on antibiotic prescribing for cystitis and asymptomatic bacteriuria in 23 community hospital emergency departments. Hosp Pharm. 2023;58(4):401-407. doi:10.1177/00185787231159578
16. Daniel M, Keller S, Mozafarihashjin M, Pahwa A, Soong C. An implementation guide to reducing overtreatment of asymptomatic bacteriuria. JAMA Intern Med. 2018;178(2):271-276.doi:10.1001/jamainternmed.2017.7290
The emergency department (ED) is estimated to provide half of all medical care in the United States, serving as a conduit between ambulatory care and inpatient settings.1 According to the Centers for Disease Control and Prevention, around 11 million antibiotic prescriptions were written in EDs in 2021.2 A previous study conducted at a US Department of Veterans (VA) Affairs medical center found that about 40% of all antimicrobial use in the ED was inappropriate.3 The ED is a critical and high-yield space for antimicrobial stewardship efforts.4
Urinary tract infections (UTIs) are one of the most common reasons for ED visits.4 In 2018, there were about 3 million UTI discharge diagnoses reported in the US.5 Diagnosis and management of UTIs can vary depending on patient sex, upper or lower urinary tract involvement, and the severity of the infection.6 Most UTIs are uncomplicated and can be safely treated with oral antibiotics at home; however, if mismanaged, they can lead to increased morbidity and mortality.6
Antimicrobial prescribing in the ED is predominantly empiric with challenges such as diverse patient needs, rising antimicrobial resistance, and limited microbiologic data at the time of discharge.6 The lack of a standardized process for urine culture follow-up after discharge represents another major complicating factor in the outpatient management of UTIs. Studies have shown that ED pharmacists play a vital role in providing quality follow-up care by optimizing antimicrobial use, resulting in improved patient outcomes in various infectious syndromes, including UTIs.7-13
Program Description
In June 2021, the VA Greater Los Angeles Healthcare System (VAGLAHS) piloted an ED pharmacist-led aftercare program to optimize postdischarge antimicrobial therapy management of UTIs. After a patient is discharged from the ED, the clinical pharmacist reviews urine culture results, interprets available antimicrobial susceptibility, conducts patient interviews, adjusts for patient-specific factors, and addresses potential antibiotic-associated adverse events. The ED pharmacist is then responsible for managing therapy changes in consultation with an ED health care practitioner (HCP).
Methods
This single center, retrospective chart review included veterans who were discharged with an oral antibiotic for UTI treatment from the VAGLAHS ED and evaluated by clinical pharmacists between June 1, 2021, and June 30, 2022. For patients with multiple ED visits, only the initial ED encounter was reviewed. Patients were excluded if they had a complicated UTI diagnosis requiring intravenous antibiotics or if they were admitted to the hospital. Data were generated through the Corporate Data Warehouse by VAGLAHS Pharmacy Informatics Service. Each patient was assigned a random number using the Microsoft Excel formula =RAND( ) and then sorted in chronological order to ensure randomization at baseline prior to data collection.
The primary aim of this quality improvement project was to characterize the impact of ED pharmacist-led interventions by evaluating the proportion of empiric to targeted therapy adjustments, antibiotic therapy discontinuation, and unmodified index treatment. The secondary objectives evaluated time to ED pharmacist aftercare follow-up, days of antibiotic exposure avoided, 30-day ED visits related to a urinary source, and transition of care documentation. Descriptive statistics were performed; median and IQR were calculated in Microsoft Excel.
Results
A total of 548 ED UTI encounters were identified, including 449 patients with an index ED UTI aftercare follow-up evaluation. Of the 246 randomly screened patients, 200 veterans met inclusion criteria. The median age of included patients was 73 years and most (83.0%) were male (Table 1). One hundred thirty-two patients (66.0%) had a cystitis diagnosis, followed by complicated UTI (14.0%) and catheter-associated UTI (11.0%). The most frequently isolated uropathogen was Escherichia coli (30.5%). ß-lactams were prescribed for empiric treatment to 121 patients (60.5%), followed by 36 fluoroquinolones prescriptions (18.0%). The median treatment duration was 7 days.
The median time to ED pharmacist UTI aftercare evaluation was 2 days (Table 2). Sixty-seven cases required pharmacist intervention, which included 34 transitions to targeted therapy (17.0%) and 33 antibiotic discontinuations (16.5%). A total of 144 days of antibiotic exposure was avoided (ie, days antibiotic was prescribed minus days therapy administered). The majority of cases without modification to index therapy were due to appropriate empiric treatment selection (49.0%). Twelve (6.0%) patients had a subsequent urinary-related ED visit within 30 days due to 8 cases of persistent and/or worsening urinary symptoms (66.7%) and 2 cases of recurrent UTI (16.7%).
Discussion
Outpatient antibiotic prescribing for UTI management in the ED is challenging due to the absence of microbiologic data at time of diagnosis and lack of consistent transition of care follow-up.6 The VAGLAHS ED UTI aftercare program piloted a pharmacist-driven protocol for review of all urine cultures and optimization of antibiotic therapy.
Most ED UTI discharges that did not require pharmacist intervention had empiric treatment selection active against the clinical isolates. This suggests that the ED prescribing practices concur with theVAGLAHS antibiogram and treatment guidelines. Clinical pharmacists intervened in about one-third of UTI cases, which included modification or discontinuation of therapy. Further review of these cases demonstrated that about half of those with a subsequent 30-day ED visit related to a urinary source had therapy modification. Most patients with a 30-day ED visit had persistent and/or worsening urinary symptoms, prompting further exploratory workup.
Although this project did not evaluate time from urine culture results to aftercare review, the VAGLAHS ED pharmacists had a median follow-up time of 48 hours. This timeline mirrors the typical duration for urine culture results, suggesting that the pilot program allowed for real time pharmacist review and intervention. Consequently, this initiative resulted in the avoidance of 144 unnecessary days of antibiotic exposure.
While the current protocol highlights the work that ED pharmacists provide postdischarge, there are additional opportunities for pharmacist intervention. For example, one-third of these clinical encounters were completed without HCP notification, indicating an ongoing need to ensure continuity of care. Additionally, all 16 patients diagnosed with asymptomatic bacteriuria were discharged with an oral antibiotic, highlighting an opportunity to further optimize antibiotic prescribing prior to discharge. ED pharmacists continue to play an important role in mitigating inappropriate and unnecessary antibiotic use, which will reduce antibiotic-related adverse drug reactions, Clostridioides difficile infection, and antimicrobial resistance.
Limitations
Inconsistent and incomplete documentation of clinical data in the electronic health record made the characterization of patient encounters challenging. Furthermore, ED HCPs varying clinical practices may have impacted the heterogeneity of UTI diagnosis and management at VAGLAHS.
Conclusions
Implementation of an ED pharmacist-driven UTI aftercare program at VAGLAHS reduced unnecessary antimicrobial exposure, improved antibiotic management, and ensured continuity of care postdischarge. Findings from our project implicate possible future pharmacist involvement predischarge, such as targeting inappropriate asymptomatic bacteriuria treatment.14-16 This pilot program suggested the feasibility of integrating antimicrobial stewardship practices within the ED setting in an ongoing effort to improve the quality of care for veterans.
The emergency department (ED) is estimated to provide half of all medical care in the United States, serving as a conduit between ambulatory care and inpatient settings.1 According to the Centers for Disease Control and Prevention, around 11 million antibiotic prescriptions were written in EDs in 2021.2 A previous study conducted at a US Department of Veterans (VA) Affairs medical center found that about 40% of all antimicrobial use in the ED was inappropriate.3 The ED is a critical and high-yield space for antimicrobial stewardship efforts.4
Urinary tract infections (UTIs) are one of the most common reasons for ED visits.4 In 2018, there were about 3 million UTI discharge diagnoses reported in the US.5 Diagnosis and management of UTIs can vary depending on patient sex, upper or lower urinary tract involvement, and the severity of the infection.6 Most UTIs are uncomplicated and can be safely treated with oral antibiotics at home; however, if mismanaged, they can lead to increased morbidity and mortality.6
Antimicrobial prescribing in the ED is predominantly empiric with challenges such as diverse patient needs, rising antimicrobial resistance, and limited microbiologic data at the time of discharge.6 The lack of a standardized process for urine culture follow-up after discharge represents another major complicating factor in the outpatient management of UTIs. Studies have shown that ED pharmacists play a vital role in providing quality follow-up care by optimizing antimicrobial use, resulting in improved patient outcomes in various infectious syndromes, including UTIs.7-13
Program Description
In June 2021, the VA Greater Los Angeles Healthcare System (VAGLAHS) piloted an ED pharmacist-led aftercare program to optimize postdischarge antimicrobial therapy management of UTIs. After a patient is discharged from the ED, the clinical pharmacist reviews urine culture results, interprets available antimicrobial susceptibility, conducts patient interviews, adjusts for patient-specific factors, and addresses potential antibiotic-associated adverse events. The ED pharmacist is then responsible for managing therapy changes in consultation with an ED health care practitioner (HCP).
Methods
This single center, retrospective chart review included veterans who were discharged with an oral antibiotic for UTI treatment from the VAGLAHS ED and evaluated by clinical pharmacists between June 1, 2021, and June 30, 2022. For patients with multiple ED visits, only the initial ED encounter was reviewed. Patients were excluded if they had a complicated UTI diagnosis requiring intravenous antibiotics or if they were admitted to the hospital. Data were generated through the Corporate Data Warehouse by VAGLAHS Pharmacy Informatics Service. Each patient was assigned a random number using the Microsoft Excel formula =RAND( ) and then sorted in chronological order to ensure randomization at baseline prior to data collection.
The primary aim of this quality improvement project was to characterize the impact of ED pharmacist-led interventions by evaluating the proportion of empiric to targeted therapy adjustments, antibiotic therapy discontinuation, and unmodified index treatment. The secondary objectives evaluated time to ED pharmacist aftercare follow-up, days of antibiotic exposure avoided, 30-day ED visits related to a urinary source, and transition of care documentation. Descriptive statistics were performed; median and IQR were calculated in Microsoft Excel.
Results
A total of 548 ED UTI encounters were identified, including 449 patients with an index ED UTI aftercare follow-up evaluation. Of the 246 randomly screened patients, 200 veterans met inclusion criteria. The median age of included patients was 73 years and most (83.0%) were male (Table 1). One hundred thirty-two patients (66.0%) had a cystitis diagnosis, followed by complicated UTI (14.0%) and catheter-associated UTI (11.0%). The most frequently isolated uropathogen was Escherichia coli (30.5%). ß-lactams were prescribed for empiric treatment to 121 patients (60.5%), followed by 36 fluoroquinolones prescriptions (18.0%). The median treatment duration was 7 days.
The median time to ED pharmacist UTI aftercare evaluation was 2 days (Table 2). Sixty-seven cases required pharmacist intervention, which included 34 transitions to targeted therapy (17.0%) and 33 antibiotic discontinuations (16.5%). A total of 144 days of antibiotic exposure was avoided (ie, days antibiotic was prescribed minus days therapy administered). The majority of cases without modification to index therapy were due to appropriate empiric treatment selection (49.0%). Twelve (6.0%) patients had a subsequent urinary-related ED visit within 30 days due to 8 cases of persistent and/or worsening urinary symptoms (66.7%) and 2 cases of recurrent UTI (16.7%).
Discussion
Outpatient antibiotic prescribing for UTI management in the ED is challenging due to the absence of microbiologic data at time of diagnosis and lack of consistent transition of care follow-up.6 The VAGLAHS ED UTI aftercare program piloted a pharmacist-driven protocol for review of all urine cultures and optimization of antibiotic therapy.
Most ED UTI discharges that did not require pharmacist intervention had empiric treatment selection active against the clinical isolates. This suggests that the ED prescribing practices concur with theVAGLAHS antibiogram and treatment guidelines. Clinical pharmacists intervened in about one-third of UTI cases, which included modification or discontinuation of therapy. Further review of these cases demonstrated that about half of those with a subsequent 30-day ED visit related to a urinary source had therapy modification. Most patients with a 30-day ED visit had persistent and/or worsening urinary symptoms, prompting further exploratory workup.
Although this project did not evaluate time from urine culture results to aftercare review, the VAGLAHS ED pharmacists had a median follow-up time of 48 hours. This timeline mirrors the typical duration for urine culture results, suggesting that the pilot program allowed for real time pharmacist review and intervention. Consequently, this initiative resulted in the avoidance of 144 unnecessary days of antibiotic exposure.
While the current protocol highlights the work that ED pharmacists provide postdischarge, there are additional opportunities for pharmacist intervention. For example, one-third of these clinical encounters were completed without HCP notification, indicating an ongoing need to ensure continuity of care. Additionally, all 16 patients diagnosed with asymptomatic bacteriuria were discharged with an oral antibiotic, highlighting an opportunity to further optimize antibiotic prescribing prior to discharge. ED pharmacists continue to play an important role in mitigating inappropriate and unnecessary antibiotic use, which will reduce antibiotic-related adverse drug reactions, Clostridioides difficile infection, and antimicrobial resistance.
Limitations
Inconsistent and incomplete documentation of clinical data in the electronic health record made the characterization of patient encounters challenging. Furthermore, ED HCPs varying clinical practices may have impacted the heterogeneity of UTI diagnosis and management at VAGLAHS.
Conclusions
Implementation of an ED pharmacist-driven UTI aftercare program at VAGLAHS reduced unnecessary antimicrobial exposure, improved antibiotic management, and ensured continuity of care postdischarge. Findings from our project implicate possible future pharmacist involvement predischarge, such as targeting inappropriate asymptomatic bacteriuria treatment.14-16 This pilot program suggested the feasibility of integrating antimicrobial stewardship practices within the ED setting in an ongoing effort to improve the quality of care for veterans.
1. Marcozzi D, Carr B, Liferidge A, Baehr N, Browne B.. Trends in the contribution of emergency departments to the provision of hospital-associated health care in the USA. Int J Health Serv. 2018;48(2):267–288. doi:10.1177/0020731417734498
2. Centers for Disease Control and Prevention. Outpatient antibiotic prescriptions — United States, 2021. Updated October 4, 2022. Accessed May 22, 2024. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/antibiotic-use/data/report-2021.html
3. Timbrook TT, Caffrey AR, Ovalle A, et al. Assessments of opportunities to improve antibiotic prescribing in an emergency department: a period prevalence survey. Infect Dis Ther. 2017;6(4):497-505. doi:10.1007/s40121-017-0175-9
4. Pulia M, Redwood R, May L. Antimicrobial stewardship in the emergency department. Emerg Med Clin North. 2018;36(4):853-872. doi:10.1016/j.emc.2018.06.012
5. Weiss A, Jiang H. Most frequent reasons for emergency department visits, 2018. December 16, 2021. Accessed May 22, 2024. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb286-ED-Frequent-Conditions-2018.pdf
6. Abrahamian FM, Moran GJ, Talan DA. Urinary tract infections in the emergency department. Infect Dis Clin North Am. 2008;22(1):73-87. doi:10.1016/j.idc.2007.10.002
7. Dumkow LE, Kenney RM, MacDonald NC, Carreno JJ, Malhotra MK, Davis SL. Impact of a multidisciplinary culture follow-up program of antimicrobial therapy in the emergency department. Infect Dis Ther. 2014;3(1):45-53. doi:10.1007/s40121-014-0026-x
8. Davis LC, Covey RB, Weston JS, Hu BB, Laine GA. Pharmacist-driven antimicrobial optimization in the emergency department. Am J Health Syst Pharm. 2016;73(5 Suppl 1):S49-S56. doi:10.2146/sp150036
9. Lingenfelter E, Darkin Z, Fritz K, Youngquist S, Madsen T, Fix M. ED pharmacist monitoring of provider antibiotic selection aids appropriate treatment for outpatient UTI. Am J Emerg Med. 2016;34(8):1600-1603. doi:10.1016/j.ajem.2016.05.076
10. Zhang X, Rowan N, Pflugeisen BM, Alajbegovic S. Urine culture guided antibiotic interventions: a pharmacist driven antimicrobial stewardship effort in the ED. Am J Emerg Med. 2017;35(4):594-598. doi:10.1016/j.ajem.2016.12.036
11. Percival KM, Valenti KM, Schmittling SE, Strader BD, Lopez RR, Bergman SJ. Impact of an antimicrobial stewardship intervention on urinary tract infection treatment in the ED. Am J Emerg Med. 2015;33(9):1129-1133. doi:10.1016/j.ajem.2015.04.067
12. Almulhim AS, Aldayyen A, Yenina K, Chiappini A, Khan TM. Optimization of antibiotic selection in the emergency department for urine culture follow ups, a retrospective pre-post intervention study: clinical pharmacist efforts. J Pharm Policy Pract. 2019;12(1):8. Published online April 9, 2019. doi:10.1186/s40545-019-0168-z
13. Stoll K, Feltz E, Ebert S. Pharmacist-driven implementation of outpatient antibiotic prescribing algorithms improves guideline adherence in the emergency department. J Pharm Pract. 2021;34(6):875-881. doi:10.1177/0897190020930979
14. Petty LA, Vaughn VM, Flanders SA, et al. Assessment of testing and treatment of asymptomatic bacteriuria initiated in the emergency department. Open Forum Infect Dis. 2020;7(12):ofaa537. Published online November 3, 2020. doi:10.1093/ofid/ofaa537
15. Ingalls EM, Veillette JJ, Olson J, et al. Impact of a multifaceted intervention on antibiotic prescribing for cystitis and asymptomatic bacteriuria in 23 community hospital emergency departments. Hosp Pharm. 2023;58(4):401-407. doi:10.1177/00185787231159578
16. Daniel M, Keller S, Mozafarihashjin M, Pahwa A, Soong C. An implementation guide to reducing overtreatment of asymptomatic bacteriuria. JAMA Intern Med. 2018;178(2):271-276.doi:10.1001/jamainternmed.2017.7290
1. Marcozzi D, Carr B, Liferidge A, Baehr N, Browne B.. Trends in the contribution of emergency departments to the provision of hospital-associated health care in the USA. Int J Health Serv. 2018;48(2):267–288. doi:10.1177/0020731417734498
2. Centers for Disease Control and Prevention. Outpatient antibiotic prescriptions — United States, 2021. Updated October 4, 2022. Accessed May 22, 2024. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/antibiotic-use/data/report-2021.html
3. Timbrook TT, Caffrey AR, Ovalle A, et al. Assessments of opportunities to improve antibiotic prescribing in an emergency department: a period prevalence survey. Infect Dis Ther. 2017;6(4):497-505. doi:10.1007/s40121-017-0175-9
4. Pulia M, Redwood R, May L. Antimicrobial stewardship in the emergency department. Emerg Med Clin North. 2018;36(4):853-872. doi:10.1016/j.emc.2018.06.012
5. Weiss A, Jiang H. Most frequent reasons for emergency department visits, 2018. December 16, 2021. Accessed May 22, 2024. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb286-ED-Frequent-Conditions-2018.pdf
6. Abrahamian FM, Moran GJ, Talan DA. Urinary tract infections in the emergency department. Infect Dis Clin North Am. 2008;22(1):73-87. doi:10.1016/j.idc.2007.10.002
7. Dumkow LE, Kenney RM, MacDonald NC, Carreno JJ, Malhotra MK, Davis SL. Impact of a multidisciplinary culture follow-up program of antimicrobial therapy in the emergency department. Infect Dis Ther. 2014;3(1):45-53. doi:10.1007/s40121-014-0026-x
8. Davis LC, Covey RB, Weston JS, Hu BB, Laine GA. Pharmacist-driven antimicrobial optimization in the emergency department. Am J Health Syst Pharm. 2016;73(5 Suppl 1):S49-S56. doi:10.2146/sp150036
9. Lingenfelter E, Darkin Z, Fritz K, Youngquist S, Madsen T, Fix M. ED pharmacist monitoring of provider antibiotic selection aids appropriate treatment for outpatient UTI. Am J Emerg Med. 2016;34(8):1600-1603. doi:10.1016/j.ajem.2016.05.076
10. Zhang X, Rowan N, Pflugeisen BM, Alajbegovic S. Urine culture guided antibiotic interventions: a pharmacist driven antimicrobial stewardship effort in the ED. Am J Emerg Med. 2017;35(4):594-598. doi:10.1016/j.ajem.2016.12.036
11. Percival KM, Valenti KM, Schmittling SE, Strader BD, Lopez RR, Bergman SJ. Impact of an antimicrobial stewardship intervention on urinary tract infection treatment in the ED. Am J Emerg Med. 2015;33(9):1129-1133. doi:10.1016/j.ajem.2015.04.067
12. Almulhim AS, Aldayyen A, Yenina K, Chiappini A, Khan TM. Optimization of antibiotic selection in the emergency department for urine culture follow ups, a retrospective pre-post intervention study: clinical pharmacist efforts. J Pharm Policy Pract. 2019;12(1):8. Published online April 9, 2019. doi:10.1186/s40545-019-0168-z
13. Stoll K, Feltz E, Ebert S. Pharmacist-driven implementation of outpatient antibiotic prescribing algorithms improves guideline adherence in the emergency department. J Pharm Pract. 2021;34(6):875-881. doi:10.1177/0897190020930979
14. Petty LA, Vaughn VM, Flanders SA, et al. Assessment of testing and treatment of asymptomatic bacteriuria initiated in the emergency department. Open Forum Infect Dis. 2020;7(12):ofaa537. Published online November 3, 2020. doi:10.1093/ofid/ofaa537
15. Ingalls EM, Veillette JJ, Olson J, et al. Impact of a multifaceted intervention on antibiotic prescribing for cystitis and asymptomatic bacteriuria in 23 community hospital emergency departments. Hosp Pharm. 2023;58(4):401-407. doi:10.1177/00185787231159578
16. Daniel M, Keller S, Mozafarihashjin M, Pahwa A, Soong C. An implementation guide to reducing overtreatment of asymptomatic bacteriuria. JAMA Intern Med. 2018;178(2):271-276.doi:10.1001/jamainternmed.2017.7290
A Crisis in Scope: Recruitment and Retention Challenges Reported by VA Gastroenterology Section Chiefs
Veterans have a high burden of digestive diseases, and gastroenterologists are needed for the diagnosis and management of these conditions.1-4 According to the Veterans Health Administration (VHA) Workforce Management and Consulting (WMC) office, the physician specialties with the greatest shortages are psychiatry, primary care, and gastroenterology.5 The VHA estimates it must hire 70 new gastroenterologists annually between fiscal years 2023 and 2027 to provide timely digestive care.5
Filling these positions will be increasingly difficult as competition for gastroenterologists is fierce. A recent Merritt Hawkins review states, “Gastroenterologists were the most in-demand type of provider during the 2022 review period.”6 In 2022, the median annual salary for US gastroenterologists was reported to be $561,375.7 Currently, the US Department of Veterans Affairs (VA) has an aggregate annual pay limit of $400,000 for all federal employees and cannot compete based on salary alone.
Retention of existing VA gastroenterologists also is challenging. The WMC has reported that 21.6% of VA gastroenterologists are eligible to retire, and in 2021, 8.2% left the VA to retire or seek non-VA positions.5 While not specific to the VA, a survey of practicing gastroenterologists conducted by the American College of Gastroenterology found a 49% burnout rate among respondents.8 Factors contributing to burnout at all career stages included administrative nonclinical work and a lack of clinical support staff.8 Burnout is also linked with higher rates of medical errors, interpersonal conflicts, and patient dissatisfaction. Burnout is more common among those with an innate strong sense of purpose and responsibility for their patients, characteristics we have observed in our VA colleagues.9
As members of the Section Chief Subcommittee of the VA Gastroenterology Field Advisory Board (GI FAB), we are passionate about providing outstanding gastroenterology care to US veterans, and we are alarmed at the struggles we are observing with recruiting and retaining a qualified national gastroenterology physician workforce. As such, we set out to survey the VA gastroenterology section chief community to gain insights into recruitment and retention challenges they have faced and identify potential solutions to these problems.
Methods
The GI FAB Section Chief Subcommittee developed a survey on gastroenterologist recruitment and retention using Microsoft Forms (Appendix). A link to the survey, which included 11 questions about facility location, current vacancies, and free text responses on barriers to recruitment and retention and potential solutions, was sent via email to all gastroenterology section chiefs on the National Gastroenterology and Hepatology Program Office’s email list of section chiefs on January 31, 2023. A reminder to complete the survey was sent to all section chiefs on February 8, 2023. Survey responses were aggregated and analyzed by the authors using descriptive statistics.
Results
The VA gastroenterologist recruitment and retention survey was emailed to 131 gastroenterology section chiefs and completed by 55 respondents (42%) (Figure). Of the responding section chiefs, 36 (65%) reported gastroenterologist vacancies at their facilities. Seventeen respondents (47%) reported a single vacancy, 12 (33%) reported 2 vacancies, 4 (11%) reported 3 vacancies, and 3 (8%) reported 4 vacancies. Of the sites with reported vacancies, 32 (89%) reported a need for a general gastroenterologist, 12 (33%) reported a need for a hepatologist, 11 (31%) reported a need for an advanced endoscopist, 9 (25%) reported a need for a gastroenterologist with specialized expertise in inflammatory bowel diseases, and 1 (3%) reported a need for a gastrointestinal motility specialist.
Numerous barriers to the recruitment and retention of gastroenterologists were reported. Given the large number of respondents that reported a unique barrier (ie, being the only respondents to report the barrier), a decision was made to include only barriers to recruitment and retention that were reported by at least 2 sites (Table). While there were some common themes, the reported barriers to retention differed from those to recruitment. The most reported barriers to recruitment were 46 respondents who noted salary, 23 reported human resources-related challenges, and 12 reported location. Respondents also noted various retention barriers, including 32 respondents who reported salary barriers; 13 reported administrative burden barriers, 6 reported medical center leadership, and 4 reported burnout.
Survey respondents provided multiple recommendations on how the VA can best support the recruitment and retention of gastroenterologists. The most frequent recommendations were to increase financial compensation by increasing the current aggregate salary cap to > $400,000, increasing the use of recruitment and retention incentives, and ensuring that gastroenterology is on the national Educational Debt Reduction Program (EDRP) list, which facilitates student loan repayment. It was recommended that a third-party company assist with hiring to overcome perceived issues with human resources. Additionally, there were multiple recommendations for improving administrative and clinical support. These included mandating how many support staff should be assigned to each gastroenterologist and providing best practice recommendations for support staff so that gastroenterologists can focus on physician-level work. Recommendations also included having a dedicated gastroenterology practice manager, nurse care coordinators, a colorectal cancer screening/surveillance coordinator, sufficient medical support assistants, and quality improvement personnel tracking ongoing professional practice evaluation data. Survey respondents also highlighted specific suggestions for recruiting recent graduates. These included offering a 4-day work week, as recent graduates place a premium on work-life balance, and ensuring gastroenterologists have individual offices. One respondent commented that gastroenterology fellows seeing VA gastroenterology attendings in cramped, shared offices, contrasted with private practice gastroenterologists in large private offices, may contribute to choosing private practice over joining the VA.
Discussion
Gastroenterology is currently listed by VHA WMC as 1 of the top 3 medical specialties in the VA with the most physician shortages.5 Working as a physician in the VA has long been recognized to have many benefits. First and foremost, many physicians are motivated by the VA mission to serve veterans, as this offers personal fulfillment and other intangible benefits. In addition, the VA can provide work-life balance, which is often not possible in fee-for-service settings, with patient panels and call volumes typically lower than in comparable private hospital settings. Moreover, VA physicians have outstanding teaching opportunities, as the VA is the largest supporter of medical education, with postgraduate trainees rotating through > 150 VA medical centers. Likewise, the VA offers a variety of student loan repayment programs (eg, the Specialty Education Loan Repayment Program and the EDRP). The VA offers research funding such as the Cooperative Studies Programs or program project funding, and rewards in parallel with the National Institute of Health (eg, career development awards, or merit review awards) and other grants. VA researchers have conducted many landmark studies that continue to shape the practice of gastroenterology and hepatology. From the earliest studies to demonstrate the effectiveness of screening colonoscopy, to the largest ongoing clinical trial in US history to assess the effectiveness of fecal immunochemical testing (FIT) vs screening colonoscopy.10-12 The VA has also led the field in the study of gastroesophageal reflux disease, hepatitis C treatment, and liver cancer screening.13-15 VA physicians also benefit from participation in the Federal Employee Retirement System, including its pension system.
These benefits apply to all medical specialties, making the VA a potentially appealing workplace for gastroenterologists. However, recent trends indicate that recruitment and retention of gastroenterologists is increasingly challenging, as the VA gastroenterology workforce grew by 5.0% in fiscal year (FY) 2020 and 1.8% in FY 2021. However, it was on track to end FY 2022 with a loss (-1.1%).5 It must be noted that this trend is not limited to the VA, and the National Center for Health Workforce Analysis predicts that gastroenterology will remain among the highest projected specialty shortages. Driven by increased demand for digestive health care services, more physicians nearing traditional retirement age, and substantially higher rates of burnout after the COVID-19 pandemic.16 All these factors are likely to result in an increasingly competitive market for gastroenterology, highlight the growing differences between VA and non-VA positions, and may augment the impact of differences for the individual gastroenterologist weighing employment options within and outside the VA.
The survey responses from VA gastroenterology section chiefs help identify potential impediments to the successful recruitment and retention in the specialty. Noncompetitive salary was the most significant barrier to the successful recruitment of gastroenterologists, identified by 46 of 55 respondents. According to a 2022 Medical Group Management Association report, the median annual salary for US gastroenterologists was $561,375.7 According to internal VA WMC data, the median 2022 VA gastroenterologist salary ranged between $287,976 and $346,435, depending on facility complexity level, excluding recruitment, retention, or relocation bonuses; performance pay; or cash awards. The current aggregate salary cap of $400,000 indicates that the VHA will likely be increasingly noncompetitive in the coming years unless novel pay authorizations are implemented.
Suboptimal human resources were the second most commonly cited impediment to recruiting gastroenterologists. Many section chiefs expressed frustration with the inefficient and slow administrative process of onboarding new gastroenterologists, which may take many months and not infrequently results in losing candidates to competing entities. While this issue is specific to recruitment, recurring and long-standing vacancies can increase work burdens, complicate logistics for remaining faculty, and may also negatively impact retention. One potential opportunity to improve VHA competitiveness is to streamline the administrative component of recruitment and optimize human resources support. The use of a third-party hiring company also should be considered.
Survey responses also indicated that administrative burden and insufficient support staff were significant retention challenges. Several respondents described a lack of efficient endoscopy workflow and delegation of simple administrative tasks to gastroenterologists as more likely in units without proper task distribution. Importantly, these shortcomings occur at the expense of workload-generating activities and career-enhancing opportunities.
While burnout rates among VA gastroenterologists have not been documented systematically, they likely correlate with workplace frustration and jeopardizegastroenterologist retention. Successful retention of gastroenterologists as highly trained medical professionals is more likely in workplaces that are vertically organized, efficient, and use physicians at the top of their skill level.
Conclusions
The VA offers the opportunity for a rewarding lifelong career in gastroenterology. The fulfillment of serving veterans, teaching future health care leaders, performing impactful research, and having job security is invaluable. Despite the tremendous benefits, this survey supports improving VA recruitment and retention strategies for the high-demand gastroenterology specialty. Improved salary parity is needed for workforce maintenance and recruitment, as is improved administrative and clinical support to maintain the high level of care our veterans deserve.
1. Shin A, Xu H, Imperiale TF. The prevalence, humanistic burden, and health care impact of irritable bowel syndrome among united states veterans. Clin Gastroenterol Hepatol. 2023;21(4):1061-1069.e1. doi:10.1016/j.cgh.2022.08.005.
2. Kent KG. Prevalence of gastrointestinal disease in US military veterans under outpatient care at the veterans health administration. SAGE Open Med. 2021;9:20503121211049112. doi:10.1177/20503121211049112
3. Beste LA, Leipertz SL, Green PK, Dominitz JA, Ross D, Ioannou GN. Trends in burden of cirrhosis and hepatocellular carcinoma by underlying liver disease in US veterans, 2001-2013. Gastroenterology. 2015;149(6):1471-e18. doi:10.1053/j.gastro.2015.07.056
4. Zullig LL, Sims KJ, McNeil R, et al. Cancer incidence among patients of the U.S. veterans affairs health care system: 2010 update. Mil Med. 2017;182(7):e1883-e1891. doi:10.7205/MILMED-D-16-00371
5. VHA Physician Workforce Resources Blueprint. US Dept of Veterans Affairs. https://dvagov.sharepoint.com/sites/WMCPortal/WFP/Documents/Reports/VHA Physician Workforce Resources Blueprint FY 23-27.pdf [Source not verified]
6. AMN Healthcare. 2022 Review of Physician and Advanced Practitioner Recruiting Incentives. Accessed June 12, 2024. https://www1.amnhealthcare.com/l/123142/2022-07-13/q6ywxg/123142/1657737392vyuONaZZ/mha2022incentivesurgraphic.pdf
7. Medical Group Management Association. MGMA DataDive Provider Compensation Data. Accessed June 12, 2024. https://www.mgma.com/datadive/provider-compensation
8. Anderson JC, Bilal M, Burke CA, et al. Burnout among US gastroenterologists and fellows in training: identifying contributing factors and offering solutions. J Clin Gastroenterol. 2023;57(10):1063-1069. doi:10.1097/MCG.0000000000001781
9. Lacy BE, Chan JL. Physician burnout: the hidden health care crisis. Clin Gastroenterol Hepatol. 2018;16(3):311-317. doi:10.1016/j.cgh.2017.06.043
10. Lieberman DA, Weiss DG, Bond JH, Ahnen DJ, Garewal H, Chejfec G. Use of colonoscopy to screen asymptomatic adults for colorectal cancer. Veterans affairs cooperative study group 380. N Engl J Med. 2000;343(3):162-168. doi:10.1056/NEJM200007203430301
11. Lieberman DA, Weiss DG; Veterans Affairs Cooperative Study Group 380. One-time screening for colorectal cancer with combined fecal occult-blood testing and examination of the distal colon. N Engl J Med. 2001;345(8):555-560. doi:10.1056/NEJMoa010328
12. Robertson DJ, Dominitz JA, Beed A, et al. Baseline features and reasons for nonparticipation in the colonoscopy versus fecal immunochemical test in reducing mortality from colorectal cancer (CONFIRM) study, a colorectal cancer screening trial. JAMA Netw Open. 2023;6(7):e2321730. doi:10.1001/jamanetworkopen.2023.21730
13. Spechler SJ, Hunter JG, Jones KM, et al. Randomized trial of medical versus surgical treatment for refractory heartburn. N Engl J Med. 2019;381(16):1513-1523. doi:10.1056/NEJMoa1811424
14. Beste LA, Green PK, Berry K, Kogut MJ, Allison SK, Ioannou GN. Effectiveness of hepatitis C antiviral treatment in a USA cohort of veteran patients with hepatocellular carcinoma. J Hepatol. 2017;67(1):32-39. doi:10.1016/j.jhep.2017.02.027
15. US Department of Veterans Affairs. Veterans affairs cooperative studies program (CSP). CSP #2023. Updated July 2022. Accessed June 12, 2024. https://www.vacsp.research.va.gov/CSP_2023/CSP_2023.asp
16. US Health Resources & Services Administration. Workforce projections. Accessed June 12, 2024. https://data.hrsa.gov/topics/health-workforce/workforce-projections
Veterans have a high burden of digestive diseases, and gastroenterologists are needed for the diagnosis and management of these conditions.1-4 According to the Veterans Health Administration (VHA) Workforce Management and Consulting (WMC) office, the physician specialties with the greatest shortages are psychiatry, primary care, and gastroenterology.5 The VHA estimates it must hire 70 new gastroenterologists annually between fiscal years 2023 and 2027 to provide timely digestive care.5
Filling these positions will be increasingly difficult as competition for gastroenterologists is fierce. A recent Merritt Hawkins review states, “Gastroenterologists were the most in-demand type of provider during the 2022 review period.”6 In 2022, the median annual salary for US gastroenterologists was reported to be $561,375.7 Currently, the US Department of Veterans Affairs (VA) has an aggregate annual pay limit of $400,000 for all federal employees and cannot compete based on salary alone.
Retention of existing VA gastroenterologists also is challenging. The WMC has reported that 21.6% of VA gastroenterologists are eligible to retire, and in 2021, 8.2% left the VA to retire or seek non-VA positions.5 While not specific to the VA, a survey of practicing gastroenterologists conducted by the American College of Gastroenterology found a 49% burnout rate among respondents.8 Factors contributing to burnout at all career stages included administrative nonclinical work and a lack of clinical support staff.8 Burnout is also linked with higher rates of medical errors, interpersonal conflicts, and patient dissatisfaction. Burnout is more common among those with an innate strong sense of purpose and responsibility for their patients, characteristics we have observed in our VA colleagues.9
As members of the Section Chief Subcommittee of the VA Gastroenterology Field Advisory Board (GI FAB), we are passionate about providing outstanding gastroenterology care to US veterans, and we are alarmed at the struggles we are observing with recruiting and retaining a qualified national gastroenterology physician workforce. As such, we set out to survey the VA gastroenterology section chief community to gain insights into recruitment and retention challenges they have faced and identify potential solutions to these problems.
Methods
The GI FAB Section Chief Subcommittee developed a survey on gastroenterologist recruitment and retention using Microsoft Forms (Appendix). A link to the survey, which included 11 questions about facility location, current vacancies, and free text responses on barriers to recruitment and retention and potential solutions, was sent via email to all gastroenterology section chiefs on the National Gastroenterology and Hepatology Program Office’s email list of section chiefs on January 31, 2023. A reminder to complete the survey was sent to all section chiefs on February 8, 2023. Survey responses were aggregated and analyzed by the authors using descriptive statistics.
Results
The VA gastroenterologist recruitment and retention survey was emailed to 131 gastroenterology section chiefs and completed by 55 respondents (42%) (Figure). Of the responding section chiefs, 36 (65%) reported gastroenterologist vacancies at their facilities. Seventeen respondents (47%) reported a single vacancy, 12 (33%) reported 2 vacancies, 4 (11%) reported 3 vacancies, and 3 (8%) reported 4 vacancies. Of the sites with reported vacancies, 32 (89%) reported a need for a general gastroenterologist, 12 (33%) reported a need for a hepatologist, 11 (31%) reported a need for an advanced endoscopist, 9 (25%) reported a need for a gastroenterologist with specialized expertise in inflammatory bowel diseases, and 1 (3%) reported a need for a gastrointestinal motility specialist.
Numerous barriers to the recruitment and retention of gastroenterologists were reported. Given the large number of respondents that reported a unique barrier (ie, being the only respondents to report the barrier), a decision was made to include only barriers to recruitment and retention that were reported by at least 2 sites (Table). While there were some common themes, the reported barriers to retention differed from those to recruitment. The most reported barriers to recruitment were 46 respondents who noted salary, 23 reported human resources-related challenges, and 12 reported location. Respondents also noted various retention barriers, including 32 respondents who reported salary barriers; 13 reported administrative burden barriers, 6 reported medical center leadership, and 4 reported burnout.
Survey respondents provided multiple recommendations on how the VA can best support the recruitment and retention of gastroenterologists. The most frequent recommendations were to increase financial compensation by increasing the current aggregate salary cap to > $400,000, increasing the use of recruitment and retention incentives, and ensuring that gastroenterology is on the national Educational Debt Reduction Program (EDRP) list, which facilitates student loan repayment. It was recommended that a third-party company assist with hiring to overcome perceived issues with human resources. Additionally, there were multiple recommendations for improving administrative and clinical support. These included mandating how many support staff should be assigned to each gastroenterologist and providing best practice recommendations for support staff so that gastroenterologists can focus on physician-level work. Recommendations also included having a dedicated gastroenterology practice manager, nurse care coordinators, a colorectal cancer screening/surveillance coordinator, sufficient medical support assistants, and quality improvement personnel tracking ongoing professional practice evaluation data. Survey respondents also highlighted specific suggestions for recruiting recent graduates. These included offering a 4-day work week, as recent graduates place a premium on work-life balance, and ensuring gastroenterologists have individual offices. One respondent commented that gastroenterology fellows seeing VA gastroenterology attendings in cramped, shared offices, contrasted with private practice gastroenterologists in large private offices, may contribute to choosing private practice over joining the VA.
Discussion
Gastroenterology is currently listed by VHA WMC as 1 of the top 3 medical specialties in the VA with the most physician shortages.5 Working as a physician in the VA has long been recognized to have many benefits. First and foremost, many physicians are motivated by the VA mission to serve veterans, as this offers personal fulfillment and other intangible benefits. In addition, the VA can provide work-life balance, which is often not possible in fee-for-service settings, with patient panels and call volumes typically lower than in comparable private hospital settings. Moreover, VA physicians have outstanding teaching opportunities, as the VA is the largest supporter of medical education, with postgraduate trainees rotating through > 150 VA medical centers. Likewise, the VA offers a variety of student loan repayment programs (eg, the Specialty Education Loan Repayment Program and the EDRP). The VA offers research funding such as the Cooperative Studies Programs or program project funding, and rewards in parallel with the National Institute of Health (eg, career development awards, or merit review awards) and other grants. VA researchers have conducted many landmark studies that continue to shape the practice of gastroenterology and hepatology. From the earliest studies to demonstrate the effectiveness of screening colonoscopy, to the largest ongoing clinical trial in US history to assess the effectiveness of fecal immunochemical testing (FIT) vs screening colonoscopy.10-12 The VA has also led the field in the study of gastroesophageal reflux disease, hepatitis C treatment, and liver cancer screening.13-15 VA physicians also benefit from participation in the Federal Employee Retirement System, including its pension system.
These benefits apply to all medical specialties, making the VA a potentially appealing workplace for gastroenterologists. However, recent trends indicate that recruitment and retention of gastroenterologists is increasingly challenging, as the VA gastroenterology workforce grew by 5.0% in fiscal year (FY) 2020 and 1.8% in FY 2021. However, it was on track to end FY 2022 with a loss (-1.1%).5 It must be noted that this trend is not limited to the VA, and the National Center for Health Workforce Analysis predicts that gastroenterology will remain among the highest projected specialty shortages. Driven by increased demand for digestive health care services, more physicians nearing traditional retirement age, and substantially higher rates of burnout after the COVID-19 pandemic.16 All these factors are likely to result in an increasingly competitive market for gastroenterology, highlight the growing differences between VA and non-VA positions, and may augment the impact of differences for the individual gastroenterologist weighing employment options within and outside the VA.
The survey responses from VA gastroenterology section chiefs help identify potential impediments to the successful recruitment and retention in the specialty. Noncompetitive salary was the most significant barrier to the successful recruitment of gastroenterologists, identified by 46 of 55 respondents. According to a 2022 Medical Group Management Association report, the median annual salary for US gastroenterologists was $561,375.7 According to internal VA WMC data, the median 2022 VA gastroenterologist salary ranged between $287,976 and $346,435, depending on facility complexity level, excluding recruitment, retention, or relocation bonuses; performance pay; or cash awards. The current aggregate salary cap of $400,000 indicates that the VHA will likely be increasingly noncompetitive in the coming years unless novel pay authorizations are implemented.
Suboptimal human resources were the second most commonly cited impediment to recruiting gastroenterologists. Many section chiefs expressed frustration with the inefficient and slow administrative process of onboarding new gastroenterologists, which may take many months and not infrequently results in losing candidates to competing entities. While this issue is specific to recruitment, recurring and long-standing vacancies can increase work burdens, complicate logistics for remaining faculty, and may also negatively impact retention. One potential opportunity to improve VHA competitiveness is to streamline the administrative component of recruitment and optimize human resources support. The use of a third-party hiring company also should be considered.
Survey responses also indicated that administrative burden and insufficient support staff were significant retention challenges. Several respondents described a lack of efficient endoscopy workflow and delegation of simple administrative tasks to gastroenterologists as more likely in units without proper task distribution. Importantly, these shortcomings occur at the expense of workload-generating activities and career-enhancing opportunities.
While burnout rates among VA gastroenterologists have not been documented systematically, they likely correlate with workplace frustration and jeopardizegastroenterologist retention. Successful retention of gastroenterologists as highly trained medical professionals is more likely in workplaces that are vertically organized, efficient, and use physicians at the top of their skill level.
Conclusions
The VA offers the opportunity for a rewarding lifelong career in gastroenterology. The fulfillment of serving veterans, teaching future health care leaders, performing impactful research, and having job security is invaluable. Despite the tremendous benefits, this survey supports improving VA recruitment and retention strategies for the high-demand gastroenterology specialty. Improved salary parity is needed for workforce maintenance and recruitment, as is improved administrative and clinical support to maintain the high level of care our veterans deserve.
Veterans have a high burden of digestive diseases, and gastroenterologists are needed for the diagnosis and management of these conditions.1-4 According to the Veterans Health Administration (VHA) Workforce Management and Consulting (WMC) office, the physician specialties with the greatest shortages are psychiatry, primary care, and gastroenterology.5 The VHA estimates it must hire 70 new gastroenterologists annually between fiscal years 2023 and 2027 to provide timely digestive care.5
Filling these positions will be increasingly difficult as competition for gastroenterologists is fierce. A recent Merritt Hawkins review states, “Gastroenterologists were the most in-demand type of provider during the 2022 review period.”6 In 2022, the median annual salary for US gastroenterologists was reported to be $561,375.7 Currently, the US Department of Veterans Affairs (VA) has an aggregate annual pay limit of $400,000 for all federal employees and cannot compete based on salary alone.
Retention of existing VA gastroenterologists also is challenging. The WMC has reported that 21.6% of VA gastroenterologists are eligible to retire, and in 2021, 8.2% left the VA to retire or seek non-VA positions.5 While not specific to the VA, a survey of practicing gastroenterologists conducted by the American College of Gastroenterology found a 49% burnout rate among respondents.8 Factors contributing to burnout at all career stages included administrative nonclinical work and a lack of clinical support staff.8 Burnout is also linked with higher rates of medical errors, interpersonal conflicts, and patient dissatisfaction. Burnout is more common among those with an innate strong sense of purpose and responsibility for their patients, characteristics we have observed in our VA colleagues.9
As members of the Section Chief Subcommittee of the VA Gastroenterology Field Advisory Board (GI FAB), we are passionate about providing outstanding gastroenterology care to US veterans, and we are alarmed at the struggles we are observing with recruiting and retaining a qualified national gastroenterology physician workforce. As such, we set out to survey the VA gastroenterology section chief community to gain insights into recruitment and retention challenges they have faced and identify potential solutions to these problems.
Methods
The GI FAB Section Chief Subcommittee developed a survey on gastroenterologist recruitment and retention using Microsoft Forms (Appendix). A link to the survey, which included 11 questions about facility location, current vacancies, and free text responses on barriers to recruitment and retention and potential solutions, was sent via email to all gastroenterology section chiefs on the National Gastroenterology and Hepatology Program Office’s email list of section chiefs on January 31, 2023. A reminder to complete the survey was sent to all section chiefs on February 8, 2023. Survey responses were aggregated and analyzed by the authors using descriptive statistics.
Results
The VA gastroenterologist recruitment and retention survey was emailed to 131 gastroenterology section chiefs and completed by 55 respondents (42%) (Figure). Of the responding section chiefs, 36 (65%) reported gastroenterologist vacancies at their facilities. Seventeen respondents (47%) reported a single vacancy, 12 (33%) reported 2 vacancies, 4 (11%) reported 3 vacancies, and 3 (8%) reported 4 vacancies. Of the sites with reported vacancies, 32 (89%) reported a need for a general gastroenterologist, 12 (33%) reported a need for a hepatologist, 11 (31%) reported a need for an advanced endoscopist, 9 (25%) reported a need for a gastroenterologist with specialized expertise in inflammatory bowel diseases, and 1 (3%) reported a need for a gastrointestinal motility specialist.
Numerous barriers to the recruitment and retention of gastroenterologists were reported. Given the large number of respondents that reported a unique barrier (ie, being the only respondents to report the barrier), a decision was made to include only barriers to recruitment and retention that were reported by at least 2 sites (Table). While there were some common themes, the reported barriers to retention differed from those to recruitment. The most reported barriers to recruitment were 46 respondents who noted salary, 23 reported human resources-related challenges, and 12 reported location. Respondents also noted various retention barriers, including 32 respondents who reported salary barriers; 13 reported administrative burden barriers, 6 reported medical center leadership, and 4 reported burnout.
Survey respondents provided multiple recommendations on how the VA can best support the recruitment and retention of gastroenterologists. The most frequent recommendations were to increase financial compensation by increasing the current aggregate salary cap to > $400,000, increasing the use of recruitment and retention incentives, and ensuring that gastroenterology is on the national Educational Debt Reduction Program (EDRP) list, which facilitates student loan repayment. It was recommended that a third-party company assist with hiring to overcome perceived issues with human resources. Additionally, there were multiple recommendations for improving administrative and clinical support. These included mandating how many support staff should be assigned to each gastroenterologist and providing best practice recommendations for support staff so that gastroenterologists can focus on physician-level work. Recommendations also included having a dedicated gastroenterology practice manager, nurse care coordinators, a colorectal cancer screening/surveillance coordinator, sufficient medical support assistants, and quality improvement personnel tracking ongoing professional practice evaluation data. Survey respondents also highlighted specific suggestions for recruiting recent graduates. These included offering a 4-day work week, as recent graduates place a premium on work-life balance, and ensuring gastroenterologists have individual offices. One respondent commented that gastroenterology fellows seeing VA gastroenterology attendings in cramped, shared offices, contrasted with private practice gastroenterologists in large private offices, may contribute to choosing private practice over joining the VA.
Discussion
Gastroenterology is currently listed by VHA WMC as 1 of the top 3 medical specialties in the VA with the most physician shortages.5 Working as a physician in the VA has long been recognized to have many benefits. First and foremost, many physicians are motivated by the VA mission to serve veterans, as this offers personal fulfillment and other intangible benefits. In addition, the VA can provide work-life balance, which is often not possible in fee-for-service settings, with patient panels and call volumes typically lower than in comparable private hospital settings. Moreover, VA physicians have outstanding teaching opportunities, as the VA is the largest supporter of medical education, with postgraduate trainees rotating through > 150 VA medical centers. Likewise, the VA offers a variety of student loan repayment programs (eg, the Specialty Education Loan Repayment Program and the EDRP). The VA offers research funding such as the Cooperative Studies Programs or program project funding, and rewards in parallel with the National Institute of Health (eg, career development awards, or merit review awards) and other grants. VA researchers have conducted many landmark studies that continue to shape the practice of gastroenterology and hepatology. From the earliest studies to demonstrate the effectiveness of screening colonoscopy, to the largest ongoing clinical trial in US history to assess the effectiveness of fecal immunochemical testing (FIT) vs screening colonoscopy.10-12 The VA has also led the field in the study of gastroesophageal reflux disease, hepatitis C treatment, and liver cancer screening.13-15 VA physicians also benefit from participation in the Federal Employee Retirement System, including its pension system.
These benefits apply to all medical specialties, making the VA a potentially appealing workplace for gastroenterologists. However, recent trends indicate that recruitment and retention of gastroenterologists is increasingly challenging, as the VA gastroenterology workforce grew by 5.0% in fiscal year (FY) 2020 and 1.8% in FY 2021. However, it was on track to end FY 2022 with a loss (-1.1%).5 It must be noted that this trend is not limited to the VA, and the National Center for Health Workforce Analysis predicts that gastroenterology will remain among the highest projected specialty shortages. Driven by increased demand for digestive health care services, more physicians nearing traditional retirement age, and substantially higher rates of burnout after the COVID-19 pandemic.16 All these factors are likely to result in an increasingly competitive market for gastroenterology, highlight the growing differences between VA and non-VA positions, and may augment the impact of differences for the individual gastroenterologist weighing employment options within and outside the VA.
The survey responses from VA gastroenterology section chiefs help identify potential impediments to the successful recruitment and retention in the specialty. Noncompetitive salary was the most significant barrier to the successful recruitment of gastroenterologists, identified by 46 of 55 respondents. According to a 2022 Medical Group Management Association report, the median annual salary for US gastroenterologists was $561,375.7 According to internal VA WMC data, the median 2022 VA gastroenterologist salary ranged between $287,976 and $346,435, depending on facility complexity level, excluding recruitment, retention, or relocation bonuses; performance pay; or cash awards. The current aggregate salary cap of $400,000 indicates that the VHA will likely be increasingly noncompetitive in the coming years unless novel pay authorizations are implemented.
Suboptimal human resources were the second most commonly cited impediment to recruiting gastroenterologists. Many section chiefs expressed frustration with the inefficient and slow administrative process of onboarding new gastroenterologists, which may take many months and not infrequently results in losing candidates to competing entities. While this issue is specific to recruitment, recurring and long-standing vacancies can increase work burdens, complicate logistics for remaining faculty, and may also negatively impact retention. One potential opportunity to improve VHA competitiveness is to streamline the administrative component of recruitment and optimize human resources support. The use of a third-party hiring company also should be considered.
Survey responses also indicated that administrative burden and insufficient support staff were significant retention challenges. Several respondents described a lack of efficient endoscopy workflow and delegation of simple administrative tasks to gastroenterologists as more likely in units without proper task distribution. Importantly, these shortcomings occur at the expense of workload-generating activities and career-enhancing opportunities.
While burnout rates among VA gastroenterologists have not been documented systematically, they likely correlate with workplace frustration and jeopardizegastroenterologist retention. Successful retention of gastroenterologists as highly trained medical professionals is more likely in workplaces that are vertically organized, efficient, and use physicians at the top of their skill level.
Conclusions
The VA offers the opportunity for a rewarding lifelong career in gastroenterology. The fulfillment of serving veterans, teaching future health care leaders, performing impactful research, and having job security is invaluable. Despite the tremendous benefits, this survey supports improving VA recruitment and retention strategies for the high-demand gastroenterology specialty. Improved salary parity is needed for workforce maintenance and recruitment, as is improved administrative and clinical support to maintain the high level of care our veterans deserve.
1. Shin A, Xu H, Imperiale TF. The prevalence, humanistic burden, and health care impact of irritable bowel syndrome among united states veterans. Clin Gastroenterol Hepatol. 2023;21(4):1061-1069.e1. doi:10.1016/j.cgh.2022.08.005.
2. Kent KG. Prevalence of gastrointestinal disease in US military veterans under outpatient care at the veterans health administration. SAGE Open Med. 2021;9:20503121211049112. doi:10.1177/20503121211049112
3. Beste LA, Leipertz SL, Green PK, Dominitz JA, Ross D, Ioannou GN. Trends in burden of cirrhosis and hepatocellular carcinoma by underlying liver disease in US veterans, 2001-2013. Gastroenterology. 2015;149(6):1471-e18. doi:10.1053/j.gastro.2015.07.056
4. Zullig LL, Sims KJ, McNeil R, et al. Cancer incidence among patients of the U.S. veterans affairs health care system: 2010 update. Mil Med. 2017;182(7):e1883-e1891. doi:10.7205/MILMED-D-16-00371
5. VHA Physician Workforce Resources Blueprint. US Dept of Veterans Affairs. https://dvagov.sharepoint.com/sites/WMCPortal/WFP/Documents/Reports/VHA Physician Workforce Resources Blueprint FY 23-27.pdf [Source not verified]
6. AMN Healthcare. 2022 Review of Physician and Advanced Practitioner Recruiting Incentives. Accessed June 12, 2024. https://www1.amnhealthcare.com/l/123142/2022-07-13/q6ywxg/123142/1657737392vyuONaZZ/mha2022incentivesurgraphic.pdf
7. Medical Group Management Association. MGMA DataDive Provider Compensation Data. Accessed June 12, 2024. https://www.mgma.com/datadive/provider-compensation
8. Anderson JC, Bilal M, Burke CA, et al. Burnout among US gastroenterologists and fellows in training: identifying contributing factors and offering solutions. J Clin Gastroenterol. 2023;57(10):1063-1069. doi:10.1097/MCG.0000000000001781
9. Lacy BE, Chan JL. Physician burnout: the hidden health care crisis. Clin Gastroenterol Hepatol. 2018;16(3):311-317. doi:10.1016/j.cgh.2017.06.043
10. Lieberman DA, Weiss DG, Bond JH, Ahnen DJ, Garewal H, Chejfec G. Use of colonoscopy to screen asymptomatic adults for colorectal cancer. Veterans affairs cooperative study group 380. N Engl J Med. 2000;343(3):162-168. doi:10.1056/NEJM200007203430301
11. Lieberman DA, Weiss DG; Veterans Affairs Cooperative Study Group 380. One-time screening for colorectal cancer with combined fecal occult-blood testing and examination of the distal colon. N Engl J Med. 2001;345(8):555-560. doi:10.1056/NEJMoa010328
12. Robertson DJ, Dominitz JA, Beed A, et al. Baseline features and reasons for nonparticipation in the colonoscopy versus fecal immunochemical test in reducing mortality from colorectal cancer (CONFIRM) study, a colorectal cancer screening trial. JAMA Netw Open. 2023;6(7):e2321730. doi:10.1001/jamanetworkopen.2023.21730
13. Spechler SJ, Hunter JG, Jones KM, et al. Randomized trial of medical versus surgical treatment for refractory heartburn. N Engl J Med. 2019;381(16):1513-1523. doi:10.1056/NEJMoa1811424
14. Beste LA, Green PK, Berry K, Kogut MJ, Allison SK, Ioannou GN. Effectiveness of hepatitis C antiviral treatment in a USA cohort of veteran patients with hepatocellular carcinoma. J Hepatol. 2017;67(1):32-39. doi:10.1016/j.jhep.2017.02.027
15. US Department of Veterans Affairs. Veterans affairs cooperative studies program (CSP). CSP #2023. Updated July 2022. Accessed June 12, 2024. https://www.vacsp.research.va.gov/CSP_2023/CSP_2023.asp
16. US Health Resources & Services Administration. Workforce projections. Accessed June 12, 2024. https://data.hrsa.gov/topics/health-workforce/workforce-projections
1. Shin A, Xu H, Imperiale TF. The prevalence, humanistic burden, and health care impact of irritable bowel syndrome among united states veterans. Clin Gastroenterol Hepatol. 2023;21(4):1061-1069.e1. doi:10.1016/j.cgh.2022.08.005.
2. Kent KG. Prevalence of gastrointestinal disease in US military veterans under outpatient care at the veterans health administration. SAGE Open Med. 2021;9:20503121211049112. doi:10.1177/20503121211049112
3. Beste LA, Leipertz SL, Green PK, Dominitz JA, Ross D, Ioannou GN. Trends in burden of cirrhosis and hepatocellular carcinoma by underlying liver disease in US veterans, 2001-2013. Gastroenterology. 2015;149(6):1471-e18. doi:10.1053/j.gastro.2015.07.056
4. Zullig LL, Sims KJ, McNeil R, et al. Cancer incidence among patients of the U.S. veterans affairs health care system: 2010 update. Mil Med. 2017;182(7):e1883-e1891. doi:10.7205/MILMED-D-16-00371
5. VHA Physician Workforce Resources Blueprint. US Dept of Veterans Affairs. https://dvagov.sharepoint.com/sites/WMCPortal/WFP/Documents/Reports/VHA Physician Workforce Resources Blueprint FY 23-27.pdf [Source not verified]
6. AMN Healthcare. 2022 Review of Physician and Advanced Practitioner Recruiting Incentives. Accessed June 12, 2024. https://www1.amnhealthcare.com/l/123142/2022-07-13/q6ywxg/123142/1657737392vyuONaZZ/mha2022incentivesurgraphic.pdf
7. Medical Group Management Association. MGMA DataDive Provider Compensation Data. Accessed June 12, 2024. https://www.mgma.com/datadive/provider-compensation
8. Anderson JC, Bilal M, Burke CA, et al. Burnout among US gastroenterologists and fellows in training: identifying contributing factors and offering solutions. J Clin Gastroenterol. 2023;57(10):1063-1069. doi:10.1097/MCG.0000000000001781
9. Lacy BE, Chan JL. Physician burnout: the hidden health care crisis. Clin Gastroenterol Hepatol. 2018;16(3):311-317. doi:10.1016/j.cgh.2017.06.043
10. Lieberman DA, Weiss DG, Bond JH, Ahnen DJ, Garewal H, Chejfec G. Use of colonoscopy to screen asymptomatic adults for colorectal cancer. Veterans affairs cooperative study group 380. N Engl J Med. 2000;343(3):162-168. doi:10.1056/NEJM200007203430301
11. Lieberman DA, Weiss DG; Veterans Affairs Cooperative Study Group 380. One-time screening for colorectal cancer with combined fecal occult-blood testing and examination of the distal colon. N Engl J Med. 2001;345(8):555-560. doi:10.1056/NEJMoa010328
12. Robertson DJ, Dominitz JA, Beed A, et al. Baseline features and reasons for nonparticipation in the colonoscopy versus fecal immunochemical test in reducing mortality from colorectal cancer (CONFIRM) study, a colorectal cancer screening trial. JAMA Netw Open. 2023;6(7):e2321730. doi:10.1001/jamanetworkopen.2023.21730
13. Spechler SJ, Hunter JG, Jones KM, et al. Randomized trial of medical versus surgical treatment for refractory heartburn. N Engl J Med. 2019;381(16):1513-1523. doi:10.1056/NEJMoa1811424
14. Beste LA, Green PK, Berry K, Kogut MJ, Allison SK, Ioannou GN. Effectiveness of hepatitis C antiviral treatment in a USA cohort of veteran patients with hepatocellular carcinoma. J Hepatol. 2017;67(1):32-39. doi:10.1016/j.jhep.2017.02.027
15. US Department of Veterans Affairs. Veterans affairs cooperative studies program (CSP). CSP #2023. Updated July 2022. Accessed June 12, 2024. https://www.vacsp.research.va.gov/CSP_2023/CSP_2023.asp
16. US Health Resources & Services Administration. Workforce projections. Accessed June 12, 2024. https://data.hrsa.gov/topics/health-workforce/workforce-projections
Anti-Tumor Necrosis Factor Treatment for Glomerulopathy: Case Report and Review of Literature
Podocytes are terminally differentiated, highly specialized cells located in juxtaposition to the basement membrane over the abluminal surfaces of endothelial cells within the glomerular tuft. This triad structure is the site of the filtration barrier, which forms highly delicate and tightly regulated architecture to carry out the ultrafiltration function of the kidney.1 The filtration barrier is characterized by foot processes that are connected by specialized junctions called slit diaphragms.
Insults to components of the filtration barrier can initiate cascading events and perpetuate structural alterations that may eventually result in sclerotic changes.2 Common causes among children include minimal change disease (MCD) with the collapse of foot processes resulting in proteinuria, Alport syndrome due to mutation of collagen fibers within the basement membrane leading to hematuria and proteinuria, immune complex mediated nephropathy following common infections or autoimmune diseases, and focal segmental glomerulosclerosis (FSGS) that can show variable histopathology toward eventual glomerular scarring.3,4 These children often clinically have minimal, if any, signs of systemic inflammation.3-5 This has been a limiting factor for the commitment to immunomodulatory treatment, except for steroids for the treatment of MCD.6 Although prolonged steroid treatment may be efficacious, adverse effects are significant in a growing child. Alternative treatments, such as tacrolimus and rituximab have been suggested as second-line steroid-sparing agents.7,8 Not uncommonly, however, these cases are managed by supportive measures only during the progression of the natural course of the disease, which may eventually lead to renal failure, requiring transplant for survival.8,9
This case report highlights a child with a variant of uncertain significance (VUS) in genes involved in Alport syndrome and FSGS who developed an abrupt onset of proteinuria and hematuria after a respiratory illness. To our knowledge, he represents the youngest case demonstrating the benefit of targeted treatment against tumor necrosis factor-α (TNF-α) for glomerulopathy using biologic response modifiers.
Case Description
This is currently a 7-year-old male patient who was born at 39 weeks gestation to gravida 3 para 3 following induced labor due to elevated maternal blood pressure. During the first 2 years of life, his growth and development were normal and his immunizations were up to date. The patient's medical history included upper respiratory tract infections (URIs), respiratory syncytial virus, as well as 3 bouts of pneumonia and multiple otitis media that resulted in 18 rounds of antibiotics. The child was also allergic to nuts and milk protein. The patient’s parents are of Northern European and Native American descent. There is no known family history of eye, ear, or kidney diseases.
Renal concerns were first noted at the age of 2 years and 6 months when he presented to an emergency department in Fall 2019 (week 0) for several weeks of intermittent dark-colored urine. His mother reported that the discoloration recently progressed in intensity to cola-colored, along with the onset of persistent vomiting without any fever or diarrhea. On physical examination, the patient had normal vitals: weight 14.8 kg (68th percentile), height 91 cm (24th percentile), and body surface area 0.6 m2. There was no edema, rash, or lymphadenopathy, but he appeared pale.
The patient’s initial laboratory results included: complete blood count with white blood cells (WBC) 10 x 103/L (reference range, 4.5-13.5 x 103/L); differential lymphocytes 69%; neutrophils 21%; hemoglobin 10 g/dL (reference range, 12-16 g/dL); hematocrit, 30%; (reference range, 37%-45%); platelets 437 103/L (reference range, 150-450 x 103/L); serum creatinine 0.46 mg/dL (reference range, 0.5-0.9 mg/dL); and albumin 3.1 g/dL (reference range, 3.5-5.2 g/dL). Serum electrolyte levels and liver enzymes were normal. A urine analysis revealed 3+ protein and 3+ blood with dysmorphic red blood cells (RBC) and RBC casts without WBC. The patient's spot urine protein-to-creatinine ratio was 4.3 and his renal ultrasound was normal. The patient was referred to Nephrology.
During the next 2 weeks, his protein-to-creatinine ratio progressed to 5.9 and serum albumin fell to 2.7 g/dL. His urine remained red colored, and a microscopic examination with RBC > 500 and WBC up to 10 on a high powered field. His workup was negative for antinuclear antibodies, antineutrophil cytoplasmic antibody, antistreptolysin-O (ASO) and anti-DNase B. Serum C3 was low at 81 mg/dL (reference range, 90-180 mg/dL), C4 was 13.3 mg/dL (reference range, 10-40 mg/dL), and immunoglobulin G was low at 452 mg/dL (reference range 719-1475 mg/dL). A baseline audiology test revealed normal hearing.
Percutaneous renal biopsy yielded about 12 glomeruli, all exhibiting mild mesangial matrix expansion and hypercellularity (Figure 1). One glomerulus had prominent parietal epithelial cells without endocapillary hypercellularity or crescent formation. There was no interstitial fibrosis or tubular atrophy. Immunofluorescence studies showed no evidence of immune complex deposition with negative staining for immunoglobulin heavy and light chains, C3 and C1q. Staining for α 2 and α 5 units of collagen was normal. Electron microscopy showed patchy areas of severe basement membrane thinning with frequent foci of mild to moderate lamina densa splitting and associated visceral epithelial cell foot process effacement (Figure 2).
These were reported as concerning findings for possible Alport syndrome by 3 independent pathology teams. The genetic testing was submitted at a commercial laboratory to screen 17 mutations, including COL4A3, COL4A4, and COL4A5. Results showed the presence of a heterozygous VUS in the COL4A4 gene (c.1055C > T; p.Pro352Leu; dbSNP ID: rs371717486; PolyPhen-2: Probably Damaging; SIFT: Deleterious) as well as the presence of a heterozygous VUS in TRPC6 gene (c2463A>T; p.Lys821Asn; dbSNP ID: rs199948731; PolyPhen-2: Benign; SIFT: Tolerated). Further genetic investigation by whole exome sequencing on approximately 20,000 genes through MNG Laboratories showed a new heterozygous VUS in the OSGEP gene [c.328T>C; p.Cys110Arg]. Additional studies ruled out mitochondrial disease, CoQ10 deficiency, and metabolic disorders upon normal findings for mitochondrial DNA, urine amino acids, plasma acylcarnitine profile, orotic acid, ammonia, and homocysteine levels.
Figure 3 summarizes the patient’s treatment response during 170 weeks of follow-up (Fall 2019 to Summer 2023). The patient was started on enalapril 0.6 mg/kg daily at week 3, which continued throughout treatment. Following a rheumatology consult at week 30, the patient was started on prednisolone 3 mg/mL to assess the role of inflammation through the treatment response. An initial dose of 2 mg/kg daily (9 mL) for 1 month was followed by every other day treatment that was tapered off by week 48. To control mild but noticeably increasing proteinuria in the interim, subcutaneous anakinra 50 mg (3 mg/kg daily) was added as a steroid
DISCUSSION
This case describes a child with rapidly progressive proteinuria and hematuria following a URI who was found to have VUS mutations in 3 different genes associated with chronic kidney disease. Serology tests on the patient were negative for streptococcal antibodies and antinuclear antibodies, ruling out poststreptococcal glomerulonephritis, or systemic lupus erythematosus. His renal biopsy findings were concerning for altered podocytes, mesangial cells, and basement membrane without inflammatory infiltrate, immune complex, complements, immunoglobulin A, or vasculopathy. His blood inflammatory markers, erythrocyte sedimentation rate, C-reactive protein, and ferritin were normal when his care team initiated daily steroids.
Overall, the patient’s clinical presentation and histopathology findings were suggestive of Alport syndrome or thin basement membrane nephropathy with a high potential to progress into FSGS.10-12 Alport syndrome affects 1 in 5000 to 10,000 children annually due to S-linked inheritance of COL4A5, or autosomal recessive inheritance of COL4A3 or COL4A4 genes. It presents with hematuria and hearing loss.10 Our patient had a single copy COL4A4 gene mutation that was classified as VUS. He also had 2 additional VUS affecting the TRPC6 and OSGEP genes. TRPC6 gene mutation can be associated with FSGS through autosomal dominant inheritance. Both COL4A4 and TRPC6 gene mutations were paternally inherited. Although the patient’s father not having renal disease argues against the clinical significance of these findings, there is literature on the potential role of heterozygous COL4A4 variant mimicking thin basement membrane nephropathy that can lead to renal impairment upon copresence of superimposed conditions.13 The patient’s rapidly progressing hematuria and changes in the basement membrane were worrisome for emerging FSGS. Furthermore, VUS of TRPC6 has been reported in late onset autosomal dominant FSGS and can be associated with early onset steroid-resistant nephrotic syndrome (NS) in children.14 This concern was voiced by 3 nephrology consultants during the initial evaluation, leading to the consensus that steroid treatment for podocytopathy would not alter the patient’s long-term outcomes (ie, progression to FSGS).
Immunomodulation
Our rationale for immunomodulatory treatment was based on the abrupt onset of renal concerns following a URI, suggesting the importance of an inflammatory trigger causing altered homeostasis in a genetically susceptible host. Preclinical models show that microbial products such as lipopolysaccharides can lead to podocytopathy by several mechanisms through activation of toll-like receptor signaling. It can directly cause apoptosis by downregulation of the intracellular Akt survival pathway.15 Lipopolysaccharide can also activate the NF-αB pathway and upregulate the production of interleukin-1 (IL-1) and TNF-α in mesangial cells.16,17
Both cytokines can promote mesangial cell proliferation.18 Through autocrine and paracrine mechanisms, proinflammatory cytokines can further perpetuate somatic tissue changes and contribute to the development of podocytopathy. For instance, TNF-α can promote podocyte injury and proteinuria by downregulation of the slit diaphragm protein expression (ie, nephrin, ezrin, or podocin), and disruption of podocyte cytoskeleton.19,20 TNF-α promotes the influx and activation of macrophages and inflammatory cells. It is actively involved in chronic alterations within the glomeruli by the upregulation of matrix metalloproteases by integrins, as well as activation of myofibroblast progenitors and extracellular matrix deposition in crosstalk with transforming growth factor and other key mediators.17,21,22
For the patient described in this case report, initial improvement on steroids encouraged the pursuit of additional treatment to downregulate inflammatory pathways within the glomerular milieu. However, within the COVID-19 environment, escalating the patient’s treatment using traditional immunomodulators (ie, calcineurin inhibitors or mycophenolate mofetil) was not favored due to the risk of infection. Initially, anakinra, a recombinant IL-1 receptor antagonist, was preferred as a steroid-sparing agent for its short life and safety profile during the pandemic. At first, the patient responded well to anakinra and was allowed a steroid wean when the dose was titrated up to 6 mg/kg daily. However, anakinra did not prevent the escalation of proteinuria following a URI. After the treatment was changed to adalimumab, a fully humanized monoclonal antibody to TNF-α, the patient continued to improve and reach full remission despite experiencing a cold and the flu in the following months.
Literature Review
There is a paucity of literature on applications of biological response modifiers for idiopathic NS and FSGS.23,24 Angeletti and colleagues reported that 3 patients with severe long-standing FSGS benefited from anakinra 4 mg/kg daily to reduce proteinuria and improve kidney function. All the patients had positive C3 staining in renal biopsy and treatment response, which supported the role of C3a in inducing podocyte injury through upregulated expression of IL-1 and IL-1R.23 Trachtman and colleagues reported on the phase II FONT trial that included 14 of 21 patients aged < 18 years with advanced FSGS who were treated with adalimumab 24 mg/m2, or ≤ 40 mg every other week.24 Although, during a 6-month period, none of the 7 patients met the endpoint of reduced proteinuria by ≥ 50%, and the authors suggested that careful patient selection may improve the treatment response in future trials.24
A recent study involving transcriptomics on renal tissue samples combined with available pathology (fibrosis), urinary markers, and clinical characteristics on 285 patients with MCD or FSGS from 3 different continents identified 3 distinct clusters. Patients with evidence of activated kidney TNF pathway (n = 72, aged > 18 years) were found to have poor clinical outcomes.25 The study identified 2 urine markers associated with the TNF pathway (ie, tissue inhibitor of metalloproteinases-1 and monocyte chemoattractant protein-1), which aligns with the preclinical findings previously mentioned.25
Conclusions
The patient’s condition in this case illustrates the complex nature of biologically predetermined cascading events in the emergence of glomerular disease upon environmental triggers under the influence of genetic factors.
Chronic kidney disease affects 7.7% of veterans annually, illustrating the need for new therapeutics.26 Based on our experience and literature review, upregulation of TNF-α is a root cause of glomerulopathy; further studies are warranted to evaluate the efficacy of anti-TNF biologic response modifiers for the treatment of these patients. Long-term postmarketing safety profile and steroid-sparing properties of adalimumab should allow inclusion of pediatric cases in future trials. Results may also contribute to identifying new predictive biomarkers related to the basement membrane when combined with precision nephrology to further advance patient selection and targeted treatment.25,27
Acknowledgments
The authors thank the patient’s mother for providing consent to allow publication of this case report.
1. Arif E, Nihalani D. Glomerular filtration barrier assembly: an insight. Postdoc J. 2013;1(4):33-45.
2. Garg PA. Review of podocyte biology. Am J Nephrol. 2018;47(suppl 1):3-13. doi:10.1159/000481633SUPPL
3. Warady BA, Agarwal R, Bangalore S, et al. Alport syndrome classification and management. Kidney Med. 2020;2(5):639-649. doi:10.1016/j.xkme.2020.05.014
4. Angioi A, Pani A. FSGS: from pathogenesis to the histological lesion. J Nephrol. 2016;29(4):517-523. doi:10.1007/s40620-016-0333-2
5. Roca N, Martinez C, Jatem E, Madrid A, Lopez M, Segarra A. Activation of the acute inflammatory phase response in idiopathic nephrotic syndrome: association with clinicopathological phenotypes and with response to corticosteroids. Clin Kidney J. 2021;14(4):1207-1215. doi:10.1093/ckj/sfaa247
6. Vivarelli M, Massella L, Ruggiero B, Emma F. Minimal change disease. Clin J Am Soc Nephrol. 2017;12(2):332-345.
7. Medjeral-Thomas NR, Lawrence C, Condon M, et al. Randomized, controlled trial of tacrolimus and prednisolone monotherapy for adults with De Novo minimal change disease: a multicenter, randomized, controlled trial. Clin J Am Soc Nephrol. 2020;15(2):209-218. doi:10.2215/CJN.06290420
8. Ye Q, Lan B, Liu H, Persson PB, Lai EY, Mao J. A critical role of the podocyte cytoskeleton in the pathogenesis of glomerular proteinuria and autoimmune podocytopathies. Acta Physiol (Oxf). 2022;235(4):e13850. doi:10.1111/apha.13850.
9. Trautmann A, Schnaidt S, Lipska-Ziμtkiewicz BS, et al. Long-term outcome of steroid-resistant nephrotic syndrome in children. J Am Soc Nephrol. 2017;28:3055-3065. doi:10.1681/ASN.2016101121
10. Kashtan CE, Gross O. Clinical practice recommendations for the diagnosis and management of Alport syndrome in children, adolescents, and young adults-an update for 2020. Pediatr Nephrol. 2021;36(3):711-719. doi:10.1007/s00467-020-04819-6
11. Savige J, Rana K, Tonna S, Buzza M, Dagher H, Wang YY. Thin basement membrane nephropathy. Kidney Int. 2003;64(4):1169-78. doi:10.1046/j.1523-1755.2003.00234.x
12. Rosenberg AZ, Kopp JB. Focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2017; 12(3):502-517. doi:10.2215/CJN.05960616
13. Savige J. Should we diagnose autosomal dominant Alport syndrome when there is a pathogenic heterozygous COL4A3 or COL4A4 variant? Kidney Int Rep. 2018;3(6):1239-1241. doi:10.1016/j.ekir.2018.08.002
14. Gigante M, Caridi G, Montemurno E, et al. TRPC6 mutations in children with steroid-resistant nephrotic syndrome and atypical phenotype. Clin J Am Soc Nephrol. 2011;6(7):1626-1634. doi:10.2215/CJN.07830910
15. Saurus P, Kuusela S, Lehtonen E, et al. Podocyte apoptosis is prevented by blocking the toll-like receptor pathway. Cell Death Dis. 2015;6(5):e1752. doi:10.1038/cddis.2015.125.
16. Baud L, Oudinet JP, Bens M, et al. Production of tumor necrosis factor by rat mesangial cells in response to bacterial lipopolysaccharide. Kidney Int. 1989;35(5):1111-1118. doi:10.1038/ki.1989.98
17. White S, Lin L, Hu K. NF-κB and tPA signaling in kidney and other diseases. Cells. 2020;9(6):1348. doi:10.3390/cells9061348.
18. Tesch GH, Lan HY, Atkins RC, Nikolic-Paterson DJ. Role of interleukin-1 in mesangial cell proliferation and matrix deposition in experimental mesangioproliferative nephritis. Am J Pathol. 1997;151(1):141-150.
19. Lai KN, Leung JCK, Chan LYY, et al. Podocyte injury induced by mesangial-derived cytokines in IgA Nephropathy. Nephrol Dial Transplant. 2009;24(1):62-72. doi:10.1093/ndt/gfn441
20. Saleem MA, Kobayashi Y. Cell biology and genetics of minimal change disease. F1000 Res. 2016;5: F1000 Faculty Rev-412. doi:10.12688/f1000research.7300.1
21. Kim KP, Williams CE, Lemmon CA. Cell-matrix interactions in renal fibrosis. Kidney Dial. 2022;2(4):607-624. doi:10.3390/kidneydial2040055
22. Zvaifler NJ. Relevance of the stroma and epithelial-mesenchymal transition (EMT) for the rheumatic diseases. Arthritis Res Ther. 2006;8(3):210. doi:10.1186/ar1963.
23. Angeletti A, Magnasco A, Trivelli A, et al. Refractory minimal change disease and focal segmental glomerular sclerosis treated with Anakinra. Kidney Int Rep. 2021;7(1):121-124. doi:10.1016/j.ekir.2021.10.018
24. Trachtman H, Vento S, Herreshoff E, et al. Efficacy of galactose and adalimumab in patients with resistant focal segmental glomerulosclerosis: report of the font clinical trial group. BMC Nephrol. 2015;16:111. doi:10.1186/s12882-015-0094-5
25. Mariani LH, Eddy S, AlAkwaa FM, et al. Precision nephrology identified tumor necrosis factor activation variability in minimal change disease and focal segmental glomerulosclerosis. Kidney Int. 2023;103(3):565-579. doi:10.1016/j.kint.2022.10.023
26. Korshak L, Washington DL, Powell J, Nylen E, Kokkinos P. Kidney Disease in Veterans. US Dept of Veterans Affairs, Office of Health Equity. Updated May 13, 2020. Accessed June 28, 2024. https://www.va.gov/HEALTHEQUITY/Kidney_Disease_In_Veterans.asp
27. Malone AF, Phelan PJ, Hall G, et al. Rare hereditary COL4A3/COL4A4 variants may be mistaken for familial focal segmental glomerulosclerosis. Kidney Int. 2014;86(6):1253-1259. doi:10.1038/ki.2014.305
Podocytes are terminally differentiated, highly specialized cells located in juxtaposition to the basement membrane over the abluminal surfaces of endothelial cells within the glomerular tuft. This triad structure is the site of the filtration barrier, which forms highly delicate and tightly regulated architecture to carry out the ultrafiltration function of the kidney.1 The filtration barrier is characterized by foot processes that are connected by specialized junctions called slit diaphragms.
Insults to components of the filtration barrier can initiate cascading events and perpetuate structural alterations that may eventually result in sclerotic changes.2 Common causes among children include minimal change disease (MCD) with the collapse of foot processes resulting in proteinuria, Alport syndrome due to mutation of collagen fibers within the basement membrane leading to hematuria and proteinuria, immune complex mediated nephropathy following common infections or autoimmune diseases, and focal segmental glomerulosclerosis (FSGS) that can show variable histopathology toward eventual glomerular scarring.3,4 These children often clinically have minimal, if any, signs of systemic inflammation.3-5 This has been a limiting factor for the commitment to immunomodulatory treatment, except for steroids for the treatment of MCD.6 Although prolonged steroid treatment may be efficacious, adverse effects are significant in a growing child. Alternative treatments, such as tacrolimus and rituximab have been suggested as second-line steroid-sparing agents.7,8 Not uncommonly, however, these cases are managed by supportive measures only during the progression of the natural course of the disease, which may eventually lead to renal failure, requiring transplant for survival.8,9
This case report highlights a child with a variant of uncertain significance (VUS) in genes involved in Alport syndrome and FSGS who developed an abrupt onset of proteinuria and hematuria after a respiratory illness. To our knowledge, he represents the youngest case demonstrating the benefit of targeted treatment against tumor necrosis factor-α (TNF-α) for glomerulopathy using biologic response modifiers.
Case Description
This is currently a 7-year-old male patient who was born at 39 weeks gestation to gravida 3 para 3 following induced labor due to elevated maternal blood pressure. During the first 2 years of life, his growth and development were normal and his immunizations were up to date. The patient's medical history included upper respiratory tract infections (URIs), respiratory syncytial virus, as well as 3 bouts of pneumonia and multiple otitis media that resulted in 18 rounds of antibiotics. The child was also allergic to nuts and milk protein. The patient’s parents are of Northern European and Native American descent. There is no known family history of eye, ear, or kidney diseases.
Renal concerns were first noted at the age of 2 years and 6 months when he presented to an emergency department in Fall 2019 (week 0) for several weeks of intermittent dark-colored urine. His mother reported that the discoloration recently progressed in intensity to cola-colored, along with the onset of persistent vomiting without any fever or diarrhea. On physical examination, the patient had normal vitals: weight 14.8 kg (68th percentile), height 91 cm (24th percentile), and body surface area 0.6 m2. There was no edema, rash, or lymphadenopathy, but he appeared pale.
The patient’s initial laboratory results included: complete blood count with white blood cells (WBC) 10 x 103/L (reference range, 4.5-13.5 x 103/L); differential lymphocytes 69%; neutrophils 21%; hemoglobin 10 g/dL (reference range, 12-16 g/dL); hematocrit, 30%; (reference range, 37%-45%); platelets 437 103/L (reference range, 150-450 x 103/L); serum creatinine 0.46 mg/dL (reference range, 0.5-0.9 mg/dL); and albumin 3.1 g/dL (reference range, 3.5-5.2 g/dL). Serum electrolyte levels and liver enzymes were normal. A urine analysis revealed 3+ protein and 3+ blood with dysmorphic red blood cells (RBC) and RBC casts without WBC. The patient's spot urine protein-to-creatinine ratio was 4.3 and his renal ultrasound was normal. The patient was referred to Nephrology.
During the next 2 weeks, his protein-to-creatinine ratio progressed to 5.9 and serum albumin fell to 2.7 g/dL. His urine remained red colored, and a microscopic examination with RBC > 500 and WBC up to 10 on a high powered field. His workup was negative for antinuclear antibodies, antineutrophil cytoplasmic antibody, antistreptolysin-O (ASO) and anti-DNase B. Serum C3 was low at 81 mg/dL (reference range, 90-180 mg/dL), C4 was 13.3 mg/dL (reference range, 10-40 mg/dL), and immunoglobulin G was low at 452 mg/dL (reference range 719-1475 mg/dL). A baseline audiology test revealed normal hearing.
Percutaneous renal biopsy yielded about 12 glomeruli, all exhibiting mild mesangial matrix expansion and hypercellularity (Figure 1). One glomerulus had prominent parietal epithelial cells without endocapillary hypercellularity or crescent formation. There was no interstitial fibrosis or tubular atrophy. Immunofluorescence studies showed no evidence of immune complex deposition with negative staining for immunoglobulin heavy and light chains, C3 and C1q. Staining for α 2 and α 5 units of collagen was normal. Electron microscopy showed patchy areas of severe basement membrane thinning with frequent foci of mild to moderate lamina densa splitting and associated visceral epithelial cell foot process effacement (Figure 2).
These were reported as concerning findings for possible Alport syndrome by 3 independent pathology teams. The genetic testing was submitted at a commercial laboratory to screen 17 mutations, including COL4A3, COL4A4, and COL4A5. Results showed the presence of a heterozygous VUS in the COL4A4 gene (c.1055C > T; p.Pro352Leu; dbSNP ID: rs371717486; PolyPhen-2: Probably Damaging; SIFT: Deleterious) as well as the presence of a heterozygous VUS in TRPC6 gene (c2463A>T; p.Lys821Asn; dbSNP ID: rs199948731; PolyPhen-2: Benign; SIFT: Tolerated). Further genetic investigation by whole exome sequencing on approximately 20,000 genes through MNG Laboratories showed a new heterozygous VUS in the OSGEP gene [c.328T>C; p.Cys110Arg]. Additional studies ruled out mitochondrial disease, CoQ10 deficiency, and metabolic disorders upon normal findings for mitochondrial DNA, urine amino acids, plasma acylcarnitine profile, orotic acid, ammonia, and homocysteine levels.
Figure 3 summarizes the patient’s treatment response during 170 weeks of follow-up (Fall 2019 to Summer 2023). The patient was started on enalapril 0.6 mg/kg daily at week 3, which continued throughout treatment. Following a rheumatology consult at week 30, the patient was started on prednisolone 3 mg/mL to assess the role of inflammation through the treatment response. An initial dose of 2 mg/kg daily (9 mL) for 1 month was followed by every other day treatment that was tapered off by week 48. To control mild but noticeably increasing proteinuria in the interim, subcutaneous anakinra 50 mg (3 mg/kg daily) was added as a steroid
DISCUSSION
This case describes a child with rapidly progressive proteinuria and hematuria following a URI who was found to have VUS mutations in 3 different genes associated with chronic kidney disease. Serology tests on the patient were negative for streptococcal antibodies and antinuclear antibodies, ruling out poststreptococcal glomerulonephritis, or systemic lupus erythematosus. His renal biopsy findings were concerning for altered podocytes, mesangial cells, and basement membrane without inflammatory infiltrate, immune complex, complements, immunoglobulin A, or vasculopathy. His blood inflammatory markers, erythrocyte sedimentation rate, C-reactive protein, and ferritin were normal when his care team initiated daily steroids.
Overall, the patient’s clinical presentation and histopathology findings were suggestive of Alport syndrome or thin basement membrane nephropathy with a high potential to progress into FSGS.10-12 Alport syndrome affects 1 in 5000 to 10,000 children annually due to S-linked inheritance of COL4A5, or autosomal recessive inheritance of COL4A3 or COL4A4 genes. It presents with hematuria and hearing loss.10 Our patient had a single copy COL4A4 gene mutation that was classified as VUS. He also had 2 additional VUS affecting the TRPC6 and OSGEP genes. TRPC6 gene mutation can be associated with FSGS through autosomal dominant inheritance. Both COL4A4 and TRPC6 gene mutations were paternally inherited. Although the patient’s father not having renal disease argues against the clinical significance of these findings, there is literature on the potential role of heterozygous COL4A4 variant mimicking thin basement membrane nephropathy that can lead to renal impairment upon copresence of superimposed conditions.13 The patient’s rapidly progressing hematuria and changes in the basement membrane were worrisome for emerging FSGS. Furthermore, VUS of TRPC6 has been reported in late onset autosomal dominant FSGS and can be associated with early onset steroid-resistant nephrotic syndrome (NS) in children.14 This concern was voiced by 3 nephrology consultants during the initial evaluation, leading to the consensus that steroid treatment for podocytopathy would not alter the patient’s long-term outcomes (ie, progression to FSGS).
Immunomodulation
Our rationale for immunomodulatory treatment was based on the abrupt onset of renal concerns following a URI, suggesting the importance of an inflammatory trigger causing altered homeostasis in a genetically susceptible host. Preclinical models show that microbial products such as lipopolysaccharides can lead to podocytopathy by several mechanisms through activation of toll-like receptor signaling. It can directly cause apoptosis by downregulation of the intracellular Akt survival pathway.15 Lipopolysaccharide can also activate the NF-αB pathway and upregulate the production of interleukin-1 (IL-1) and TNF-α in mesangial cells.16,17
Both cytokines can promote mesangial cell proliferation.18 Through autocrine and paracrine mechanisms, proinflammatory cytokines can further perpetuate somatic tissue changes and contribute to the development of podocytopathy. For instance, TNF-α can promote podocyte injury and proteinuria by downregulation of the slit diaphragm protein expression (ie, nephrin, ezrin, or podocin), and disruption of podocyte cytoskeleton.19,20 TNF-α promotes the influx and activation of macrophages and inflammatory cells. It is actively involved in chronic alterations within the glomeruli by the upregulation of matrix metalloproteases by integrins, as well as activation of myofibroblast progenitors and extracellular matrix deposition in crosstalk with transforming growth factor and other key mediators.17,21,22
For the patient described in this case report, initial improvement on steroids encouraged the pursuit of additional treatment to downregulate inflammatory pathways within the glomerular milieu. However, within the COVID-19 environment, escalating the patient’s treatment using traditional immunomodulators (ie, calcineurin inhibitors or mycophenolate mofetil) was not favored due to the risk of infection. Initially, anakinra, a recombinant IL-1 receptor antagonist, was preferred as a steroid-sparing agent for its short life and safety profile during the pandemic. At first, the patient responded well to anakinra and was allowed a steroid wean when the dose was titrated up to 6 mg/kg daily. However, anakinra did not prevent the escalation of proteinuria following a URI. After the treatment was changed to adalimumab, a fully humanized monoclonal antibody to TNF-α, the patient continued to improve and reach full remission despite experiencing a cold and the flu in the following months.
Literature Review
There is a paucity of literature on applications of biological response modifiers for idiopathic NS and FSGS.23,24 Angeletti and colleagues reported that 3 patients with severe long-standing FSGS benefited from anakinra 4 mg/kg daily to reduce proteinuria and improve kidney function. All the patients had positive C3 staining in renal biopsy and treatment response, which supported the role of C3a in inducing podocyte injury through upregulated expression of IL-1 and IL-1R.23 Trachtman and colleagues reported on the phase II FONT trial that included 14 of 21 patients aged < 18 years with advanced FSGS who were treated with adalimumab 24 mg/m2, or ≤ 40 mg every other week.24 Although, during a 6-month period, none of the 7 patients met the endpoint of reduced proteinuria by ≥ 50%, and the authors suggested that careful patient selection may improve the treatment response in future trials.24
A recent study involving transcriptomics on renal tissue samples combined with available pathology (fibrosis), urinary markers, and clinical characteristics on 285 patients with MCD or FSGS from 3 different continents identified 3 distinct clusters. Patients with evidence of activated kidney TNF pathway (n = 72, aged > 18 years) were found to have poor clinical outcomes.25 The study identified 2 urine markers associated with the TNF pathway (ie, tissue inhibitor of metalloproteinases-1 and monocyte chemoattractant protein-1), which aligns with the preclinical findings previously mentioned.25
Conclusions
The patient’s condition in this case illustrates the complex nature of biologically predetermined cascading events in the emergence of glomerular disease upon environmental triggers under the influence of genetic factors.
Chronic kidney disease affects 7.7% of veterans annually, illustrating the need for new therapeutics.26 Based on our experience and literature review, upregulation of TNF-α is a root cause of glomerulopathy; further studies are warranted to evaluate the efficacy of anti-TNF biologic response modifiers for the treatment of these patients. Long-term postmarketing safety profile and steroid-sparing properties of adalimumab should allow inclusion of pediatric cases in future trials. Results may also contribute to identifying new predictive biomarkers related to the basement membrane when combined with precision nephrology to further advance patient selection and targeted treatment.25,27
Acknowledgments
The authors thank the patient’s mother for providing consent to allow publication of this case report.
Podocytes are terminally differentiated, highly specialized cells located in juxtaposition to the basement membrane over the abluminal surfaces of endothelial cells within the glomerular tuft. This triad structure is the site of the filtration barrier, which forms highly delicate and tightly regulated architecture to carry out the ultrafiltration function of the kidney.1 The filtration barrier is characterized by foot processes that are connected by specialized junctions called slit diaphragms.
Insults to components of the filtration barrier can initiate cascading events and perpetuate structural alterations that may eventually result in sclerotic changes.2 Common causes among children include minimal change disease (MCD) with the collapse of foot processes resulting in proteinuria, Alport syndrome due to mutation of collagen fibers within the basement membrane leading to hematuria and proteinuria, immune complex mediated nephropathy following common infections or autoimmune diseases, and focal segmental glomerulosclerosis (FSGS) that can show variable histopathology toward eventual glomerular scarring.3,4 These children often clinically have minimal, if any, signs of systemic inflammation.3-5 This has been a limiting factor for the commitment to immunomodulatory treatment, except for steroids for the treatment of MCD.6 Although prolonged steroid treatment may be efficacious, adverse effects are significant in a growing child. Alternative treatments, such as tacrolimus and rituximab have been suggested as second-line steroid-sparing agents.7,8 Not uncommonly, however, these cases are managed by supportive measures only during the progression of the natural course of the disease, which may eventually lead to renal failure, requiring transplant for survival.8,9
This case report highlights a child with a variant of uncertain significance (VUS) in genes involved in Alport syndrome and FSGS who developed an abrupt onset of proteinuria and hematuria after a respiratory illness. To our knowledge, he represents the youngest case demonstrating the benefit of targeted treatment against tumor necrosis factor-α (TNF-α) for glomerulopathy using biologic response modifiers.
Case Description
This is currently a 7-year-old male patient who was born at 39 weeks gestation to gravida 3 para 3 following induced labor due to elevated maternal blood pressure. During the first 2 years of life, his growth and development were normal and his immunizations were up to date. The patient's medical history included upper respiratory tract infections (URIs), respiratory syncytial virus, as well as 3 bouts of pneumonia and multiple otitis media that resulted in 18 rounds of antibiotics. The child was also allergic to nuts and milk protein. The patient’s parents are of Northern European and Native American descent. There is no known family history of eye, ear, or kidney diseases.
Renal concerns were first noted at the age of 2 years and 6 months when he presented to an emergency department in Fall 2019 (week 0) for several weeks of intermittent dark-colored urine. His mother reported that the discoloration recently progressed in intensity to cola-colored, along with the onset of persistent vomiting without any fever or diarrhea. On physical examination, the patient had normal vitals: weight 14.8 kg (68th percentile), height 91 cm (24th percentile), and body surface area 0.6 m2. There was no edema, rash, or lymphadenopathy, but he appeared pale.
The patient’s initial laboratory results included: complete blood count with white blood cells (WBC) 10 x 103/L (reference range, 4.5-13.5 x 103/L); differential lymphocytes 69%; neutrophils 21%; hemoglobin 10 g/dL (reference range, 12-16 g/dL); hematocrit, 30%; (reference range, 37%-45%); platelets 437 103/L (reference range, 150-450 x 103/L); serum creatinine 0.46 mg/dL (reference range, 0.5-0.9 mg/dL); and albumin 3.1 g/dL (reference range, 3.5-5.2 g/dL). Serum electrolyte levels and liver enzymes were normal. A urine analysis revealed 3+ protein and 3+ blood with dysmorphic red blood cells (RBC) and RBC casts without WBC. The patient's spot urine protein-to-creatinine ratio was 4.3 and his renal ultrasound was normal. The patient was referred to Nephrology.
During the next 2 weeks, his protein-to-creatinine ratio progressed to 5.9 and serum albumin fell to 2.7 g/dL. His urine remained red colored, and a microscopic examination with RBC > 500 and WBC up to 10 on a high powered field. His workup was negative for antinuclear antibodies, antineutrophil cytoplasmic antibody, antistreptolysin-O (ASO) and anti-DNase B. Serum C3 was low at 81 mg/dL (reference range, 90-180 mg/dL), C4 was 13.3 mg/dL (reference range, 10-40 mg/dL), and immunoglobulin G was low at 452 mg/dL (reference range 719-1475 mg/dL). A baseline audiology test revealed normal hearing.
Percutaneous renal biopsy yielded about 12 glomeruli, all exhibiting mild mesangial matrix expansion and hypercellularity (Figure 1). One glomerulus had prominent parietal epithelial cells without endocapillary hypercellularity or crescent formation. There was no interstitial fibrosis or tubular atrophy. Immunofluorescence studies showed no evidence of immune complex deposition with negative staining for immunoglobulin heavy and light chains, C3 and C1q. Staining for α 2 and α 5 units of collagen was normal. Electron microscopy showed patchy areas of severe basement membrane thinning with frequent foci of mild to moderate lamina densa splitting and associated visceral epithelial cell foot process effacement (Figure 2).
These were reported as concerning findings for possible Alport syndrome by 3 independent pathology teams. The genetic testing was submitted at a commercial laboratory to screen 17 mutations, including COL4A3, COL4A4, and COL4A5. Results showed the presence of a heterozygous VUS in the COL4A4 gene (c.1055C > T; p.Pro352Leu; dbSNP ID: rs371717486; PolyPhen-2: Probably Damaging; SIFT: Deleterious) as well as the presence of a heterozygous VUS in TRPC6 gene (c2463A>T; p.Lys821Asn; dbSNP ID: rs199948731; PolyPhen-2: Benign; SIFT: Tolerated). Further genetic investigation by whole exome sequencing on approximately 20,000 genes through MNG Laboratories showed a new heterozygous VUS in the OSGEP gene [c.328T>C; p.Cys110Arg]. Additional studies ruled out mitochondrial disease, CoQ10 deficiency, and metabolic disorders upon normal findings for mitochondrial DNA, urine amino acids, plasma acylcarnitine profile, orotic acid, ammonia, and homocysteine levels.
Figure 3 summarizes the patient’s treatment response during 170 weeks of follow-up (Fall 2019 to Summer 2023). The patient was started on enalapril 0.6 mg/kg daily at week 3, which continued throughout treatment. Following a rheumatology consult at week 30, the patient was started on prednisolone 3 mg/mL to assess the role of inflammation through the treatment response. An initial dose of 2 mg/kg daily (9 mL) for 1 month was followed by every other day treatment that was tapered off by week 48. To control mild but noticeably increasing proteinuria in the interim, subcutaneous anakinra 50 mg (3 mg/kg daily) was added as a steroid
DISCUSSION
This case describes a child with rapidly progressive proteinuria and hematuria following a URI who was found to have VUS mutations in 3 different genes associated with chronic kidney disease. Serology tests on the patient were negative for streptococcal antibodies and antinuclear antibodies, ruling out poststreptococcal glomerulonephritis, or systemic lupus erythematosus. His renal biopsy findings were concerning for altered podocytes, mesangial cells, and basement membrane without inflammatory infiltrate, immune complex, complements, immunoglobulin A, or vasculopathy. His blood inflammatory markers, erythrocyte sedimentation rate, C-reactive protein, and ferritin were normal when his care team initiated daily steroids.
Overall, the patient’s clinical presentation and histopathology findings were suggestive of Alport syndrome or thin basement membrane nephropathy with a high potential to progress into FSGS.10-12 Alport syndrome affects 1 in 5000 to 10,000 children annually due to S-linked inheritance of COL4A5, or autosomal recessive inheritance of COL4A3 or COL4A4 genes. It presents with hematuria and hearing loss.10 Our patient had a single copy COL4A4 gene mutation that was classified as VUS. He also had 2 additional VUS affecting the TRPC6 and OSGEP genes. TRPC6 gene mutation can be associated with FSGS through autosomal dominant inheritance. Both COL4A4 and TRPC6 gene mutations were paternally inherited. Although the patient’s father not having renal disease argues against the clinical significance of these findings, there is literature on the potential role of heterozygous COL4A4 variant mimicking thin basement membrane nephropathy that can lead to renal impairment upon copresence of superimposed conditions.13 The patient’s rapidly progressing hematuria and changes in the basement membrane were worrisome for emerging FSGS. Furthermore, VUS of TRPC6 has been reported in late onset autosomal dominant FSGS and can be associated with early onset steroid-resistant nephrotic syndrome (NS) in children.14 This concern was voiced by 3 nephrology consultants during the initial evaluation, leading to the consensus that steroid treatment for podocytopathy would not alter the patient’s long-term outcomes (ie, progression to FSGS).
Immunomodulation
Our rationale for immunomodulatory treatment was based on the abrupt onset of renal concerns following a URI, suggesting the importance of an inflammatory trigger causing altered homeostasis in a genetically susceptible host. Preclinical models show that microbial products such as lipopolysaccharides can lead to podocytopathy by several mechanisms through activation of toll-like receptor signaling. It can directly cause apoptosis by downregulation of the intracellular Akt survival pathway.15 Lipopolysaccharide can also activate the NF-αB pathway and upregulate the production of interleukin-1 (IL-1) and TNF-α in mesangial cells.16,17
Both cytokines can promote mesangial cell proliferation.18 Through autocrine and paracrine mechanisms, proinflammatory cytokines can further perpetuate somatic tissue changes and contribute to the development of podocytopathy. For instance, TNF-α can promote podocyte injury and proteinuria by downregulation of the slit diaphragm protein expression (ie, nephrin, ezrin, or podocin), and disruption of podocyte cytoskeleton.19,20 TNF-α promotes the influx and activation of macrophages and inflammatory cells. It is actively involved in chronic alterations within the glomeruli by the upregulation of matrix metalloproteases by integrins, as well as activation of myofibroblast progenitors and extracellular matrix deposition in crosstalk with transforming growth factor and other key mediators.17,21,22
For the patient described in this case report, initial improvement on steroids encouraged the pursuit of additional treatment to downregulate inflammatory pathways within the glomerular milieu. However, within the COVID-19 environment, escalating the patient’s treatment using traditional immunomodulators (ie, calcineurin inhibitors or mycophenolate mofetil) was not favored due to the risk of infection. Initially, anakinra, a recombinant IL-1 receptor antagonist, was preferred as a steroid-sparing agent for its short life and safety profile during the pandemic. At first, the patient responded well to anakinra and was allowed a steroid wean when the dose was titrated up to 6 mg/kg daily. However, anakinra did not prevent the escalation of proteinuria following a URI. After the treatment was changed to adalimumab, a fully humanized monoclonal antibody to TNF-α, the patient continued to improve and reach full remission despite experiencing a cold and the flu in the following months.
Literature Review
There is a paucity of literature on applications of biological response modifiers for idiopathic NS and FSGS.23,24 Angeletti and colleagues reported that 3 patients with severe long-standing FSGS benefited from anakinra 4 mg/kg daily to reduce proteinuria and improve kidney function. All the patients had positive C3 staining in renal biopsy and treatment response, which supported the role of C3a in inducing podocyte injury through upregulated expression of IL-1 and IL-1R.23 Trachtman and colleagues reported on the phase II FONT trial that included 14 of 21 patients aged < 18 years with advanced FSGS who were treated with adalimumab 24 mg/m2, or ≤ 40 mg every other week.24 Although, during a 6-month period, none of the 7 patients met the endpoint of reduced proteinuria by ≥ 50%, and the authors suggested that careful patient selection may improve the treatment response in future trials.24
A recent study involving transcriptomics on renal tissue samples combined with available pathology (fibrosis), urinary markers, and clinical characteristics on 285 patients with MCD or FSGS from 3 different continents identified 3 distinct clusters. Patients with evidence of activated kidney TNF pathway (n = 72, aged > 18 years) were found to have poor clinical outcomes.25 The study identified 2 urine markers associated with the TNF pathway (ie, tissue inhibitor of metalloproteinases-1 and monocyte chemoattractant protein-1), which aligns with the preclinical findings previously mentioned.25
Conclusions
The patient’s condition in this case illustrates the complex nature of biologically predetermined cascading events in the emergence of glomerular disease upon environmental triggers under the influence of genetic factors.
Chronic kidney disease affects 7.7% of veterans annually, illustrating the need for new therapeutics.26 Based on our experience and literature review, upregulation of TNF-α is a root cause of glomerulopathy; further studies are warranted to evaluate the efficacy of anti-TNF biologic response modifiers for the treatment of these patients. Long-term postmarketing safety profile and steroid-sparing properties of adalimumab should allow inclusion of pediatric cases in future trials. Results may also contribute to identifying new predictive biomarkers related to the basement membrane when combined with precision nephrology to further advance patient selection and targeted treatment.25,27
Acknowledgments
The authors thank the patient’s mother for providing consent to allow publication of this case report.
1. Arif E, Nihalani D. Glomerular filtration barrier assembly: an insight. Postdoc J. 2013;1(4):33-45.
2. Garg PA. Review of podocyte biology. Am J Nephrol. 2018;47(suppl 1):3-13. doi:10.1159/000481633SUPPL
3. Warady BA, Agarwal R, Bangalore S, et al. Alport syndrome classification and management. Kidney Med. 2020;2(5):639-649. doi:10.1016/j.xkme.2020.05.014
4. Angioi A, Pani A. FSGS: from pathogenesis to the histological lesion. J Nephrol. 2016;29(4):517-523. doi:10.1007/s40620-016-0333-2
5. Roca N, Martinez C, Jatem E, Madrid A, Lopez M, Segarra A. Activation of the acute inflammatory phase response in idiopathic nephrotic syndrome: association with clinicopathological phenotypes and with response to corticosteroids. Clin Kidney J. 2021;14(4):1207-1215. doi:10.1093/ckj/sfaa247
6. Vivarelli M, Massella L, Ruggiero B, Emma F. Minimal change disease. Clin J Am Soc Nephrol. 2017;12(2):332-345.
7. Medjeral-Thomas NR, Lawrence C, Condon M, et al. Randomized, controlled trial of tacrolimus and prednisolone monotherapy for adults with De Novo minimal change disease: a multicenter, randomized, controlled trial. Clin J Am Soc Nephrol. 2020;15(2):209-218. doi:10.2215/CJN.06290420
8. Ye Q, Lan B, Liu H, Persson PB, Lai EY, Mao J. A critical role of the podocyte cytoskeleton in the pathogenesis of glomerular proteinuria and autoimmune podocytopathies. Acta Physiol (Oxf). 2022;235(4):e13850. doi:10.1111/apha.13850.
9. Trautmann A, Schnaidt S, Lipska-Ziμtkiewicz BS, et al. Long-term outcome of steroid-resistant nephrotic syndrome in children. J Am Soc Nephrol. 2017;28:3055-3065. doi:10.1681/ASN.2016101121
10. Kashtan CE, Gross O. Clinical practice recommendations for the diagnosis and management of Alport syndrome in children, adolescents, and young adults-an update for 2020. Pediatr Nephrol. 2021;36(3):711-719. doi:10.1007/s00467-020-04819-6
11. Savige J, Rana K, Tonna S, Buzza M, Dagher H, Wang YY. Thin basement membrane nephropathy. Kidney Int. 2003;64(4):1169-78. doi:10.1046/j.1523-1755.2003.00234.x
12. Rosenberg AZ, Kopp JB. Focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2017; 12(3):502-517. doi:10.2215/CJN.05960616
13. Savige J. Should we diagnose autosomal dominant Alport syndrome when there is a pathogenic heterozygous COL4A3 or COL4A4 variant? Kidney Int Rep. 2018;3(6):1239-1241. doi:10.1016/j.ekir.2018.08.002
14. Gigante M, Caridi G, Montemurno E, et al. TRPC6 mutations in children with steroid-resistant nephrotic syndrome and atypical phenotype. Clin J Am Soc Nephrol. 2011;6(7):1626-1634. doi:10.2215/CJN.07830910
15. Saurus P, Kuusela S, Lehtonen E, et al. Podocyte apoptosis is prevented by blocking the toll-like receptor pathway. Cell Death Dis. 2015;6(5):e1752. doi:10.1038/cddis.2015.125.
16. Baud L, Oudinet JP, Bens M, et al. Production of tumor necrosis factor by rat mesangial cells in response to bacterial lipopolysaccharide. Kidney Int. 1989;35(5):1111-1118. doi:10.1038/ki.1989.98
17. White S, Lin L, Hu K. NF-κB and tPA signaling in kidney and other diseases. Cells. 2020;9(6):1348. doi:10.3390/cells9061348.
18. Tesch GH, Lan HY, Atkins RC, Nikolic-Paterson DJ. Role of interleukin-1 in mesangial cell proliferation and matrix deposition in experimental mesangioproliferative nephritis. Am J Pathol. 1997;151(1):141-150.
19. Lai KN, Leung JCK, Chan LYY, et al. Podocyte injury induced by mesangial-derived cytokines in IgA Nephropathy. Nephrol Dial Transplant. 2009;24(1):62-72. doi:10.1093/ndt/gfn441
20. Saleem MA, Kobayashi Y. Cell biology and genetics of minimal change disease. F1000 Res. 2016;5: F1000 Faculty Rev-412. doi:10.12688/f1000research.7300.1
21. Kim KP, Williams CE, Lemmon CA. Cell-matrix interactions in renal fibrosis. Kidney Dial. 2022;2(4):607-624. doi:10.3390/kidneydial2040055
22. Zvaifler NJ. Relevance of the stroma and epithelial-mesenchymal transition (EMT) for the rheumatic diseases. Arthritis Res Ther. 2006;8(3):210. doi:10.1186/ar1963.
23. Angeletti A, Magnasco A, Trivelli A, et al. Refractory minimal change disease and focal segmental glomerular sclerosis treated with Anakinra. Kidney Int Rep. 2021;7(1):121-124. doi:10.1016/j.ekir.2021.10.018
24. Trachtman H, Vento S, Herreshoff E, et al. Efficacy of galactose and adalimumab in patients with resistant focal segmental glomerulosclerosis: report of the font clinical trial group. BMC Nephrol. 2015;16:111. doi:10.1186/s12882-015-0094-5
25. Mariani LH, Eddy S, AlAkwaa FM, et al. Precision nephrology identified tumor necrosis factor activation variability in minimal change disease and focal segmental glomerulosclerosis. Kidney Int. 2023;103(3):565-579. doi:10.1016/j.kint.2022.10.023
26. Korshak L, Washington DL, Powell J, Nylen E, Kokkinos P. Kidney Disease in Veterans. US Dept of Veterans Affairs, Office of Health Equity. Updated May 13, 2020. Accessed June 28, 2024. https://www.va.gov/HEALTHEQUITY/Kidney_Disease_In_Veterans.asp
27. Malone AF, Phelan PJ, Hall G, et al. Rare hereditary COL4A3/COL4A4 variants may be mistaken for familial focal segmental glomerulosclerosis. Kidney Int. 2014;86(6):1253-1259. doi:10.1038/ki.2014.305
1. Arif E, Nihalani D. Glomerular filtration barrier assembly: an insight. Postdoc J. 2013;1(4):33-45.
2. Garg PA. Review of podocyte biology. Am J Nephrol. 2018;47(suppl 1):3-13. doi:10.1159/000481633SUPPL
3. Warady BA, Agarwal R, Bangalore S, et al. Alport syndrome classification and management. Kidney Med. 2020;2(5):639-649. doi:10.1016/j.xkme.2020.05.014
4. Angioi A, Pani A. FSGS: from pathogenesis to the histological lesion. J Nephrol. 2016;29(4):517-523. doi:10.1007/s40620-016-0333-2
5. Roca N, Martinez C, Jatem E, Madrid A, Lopez M, Segarra A. Activation of the acute inflammatory phase response in idiopathic nephrotic syndrome: association with clinicopathological phenotypes and with response to corticosteroids. Clin Kidney J. 2021;14(4):1207-1215. doi:10.1093/ckj/sfaa247
6. Vivarelli M, Massella L, Ruggiero B, Emma F. Minimal change disease. Clin J Am Soc Nephrol. 2017;12(2):332-345.
7. Medjeral-Thomas NR, Lawrence C, Condon M, et al. Randomized, controlled trial of tacrolimus and prednisolone monotherapy for adults with De Novo minimal change disease: a multicenter, randomized, controlled trial. Clin J Am Soc Nephrol. 2020;15(2):209-218. doi:10.2215/CJN.06290420
8. Ye Q, Lan B, Liu H, Persson PB, Lai EY, Mao J. A critical role of the podocyte cytoskeleton in the pathogenesis of glomerular proteinuria and autoimmune podocytopathies. Acta Physiol (Oxf). 2022;235(4):e13850. doi:10.1111/apha.13850.
9. Trautmann A, Schnaidt S, Lipska-Ziμtkiewicz BS, et al. Long-term outcome of steroid-resistant nephrotic syndrome in children. J Am Soc Nephrol. 2017;28:3055-3065. doi:10.1681/ASN.2016101121
10. Kashtan CE, Gross O. Clinical practice recommendations for the diagnosis and management of Alport syndrome in children, adolescents, and young adults-an update for 2020. Pediatr Nephrol. 2021;36(3):711-719. doi:10.1007/s00467-020-04819-6
11. Savige J, Rana K, Tonna S, Buzza M, Dagher H, Wang YY. Thin basement membrane nephropathy. Kidney Int. 2003;64(4):1169-78. doi:10.1046/j.1523-1755.2003.00234.x
12. Rosenberg AZ, Kopp JB. Focal segmental glomerulosclerosis. Clin J Am Soc Nephrol. 2017; 12(3):502-517. doi:10.2215/CJN.05960616
13. Savige J. Should we diagnose autosomal dominant Alport syndrome when there is a pathogenic heterozygous COL4A3 or COL4A4 variant? Kidney Int Rep. 2018;3(6):1239-1241. doi:10.1016/j.ekir.2018.08.002
14. Gigante M, Caridi G, Montemurno E, et al. TRPC6 mutations in children with steroid-resistant nephrotic syndrome and atypical phenotype. Clin J Am Soc Nephrol. 2011;6(7):1626-1634. doi:10.2215/CJN.07830910
15. Saurus P, Kuusela S, Lehtonen E, et al. Podocyte apoptosis is prevented by blocking the toll-like receptor pathway. Cell Death Dis. 2015;6(5):e1752. doi:10.1038/cddis.2015.125.
16. Baud L, Oudinet JP, Bens M, et al. Production of tumor necrosis factor by rat mesangial cells in response to bacterial lipopolysaccharide. Kidney Int. 1989;35(5):1111-1118. doi:10.1038/ki.1989.98
17. White S, Lin L, Hu K. NF-κB and tPA signaling in kidney and other diseases. Cells. 2020;9(6):1348. doi:10.3390/cells9061348.
18. Tesch GH, Lan HY, Atkins RC, Nikolic-Paterson DJ. Role of interleukin-1 in mesangial cell proliferation and matrix deposition in experimental mesangioproliferative nephritis. Am J Pathol. 1997;151(1):141-150.
19. Lai KN, Leung JCK, Chan LYY, et al. Podocyte injury induced by mesangial-derived cytokines in IgA Nephropathy. Nephrol Dial Transplant. 2009;24(1):62-72. doi:10.1093/ndt/gfn441
20. Saleem MA, Kobayashi Y. Cell biology and genetics of minimal change disease. F1000 Res. 2016;5: F1000 Faculty Rev-412. doi:10.12688/f1000research.7300.1
21. Kim KP, Williams CE, Lemmon CA. Cell-matrix interactions in renal fibrosis. Kidney Dial. 2022;2(4):607-624. doi:10.3390/kidneydial2040055
22. Zvaifler NJ. Relevance of the stroma and epithelial-mesenchymal transition (EMT) for the rheumatic diseases. Arthritis Res Ther. 2006;8(3):210. doi:10.1186/ar1963.
23. Angeletti A, Magnasco A, Trivelli A, et al. Refractory minimal change disease and focal segmental glomerular sclerosis treated with Anakinra. Kidney Int Rep. 2021;7(1):121-124. doi:10.1016/j.ekir.2021.10.018
24. Trachtman H, Vento S, Herreshoff E, et al. Efficacy of galactose and adalimumab in patients with resistant focal segmental glomerulosclerosis: report of the font clinical trial group. BMC Nephrol. 2015;16:111. doi:10.1186/s12882-015-0094-5
25. Mariani LH, Eddy S, AlAkwaa FM, et al. Precision nephrology identified tumor necrosis factor activation variability in minimal change disease and focal segmental glomerulosclerosis. Kidney Int. 2023;103(3):565-579. doi:10.1016/j.kint.2022.10.023
26. Korshak L, Washington DL, Powell J, Nylen E, Kokkinos P. Kidney Disease in Veterans. US Dept of Veterans Affairs, Office of Health Equity. Updated May 13, 2020. Accessed June 28, 2024. https://www.va.gov/HEALTHEQUITY/Kidney_Disease_In_Veterans.asp
27. Malone AF, Phelan PJ, Hall G, et al. Rare hereditary COL4A3/COL4A4 variants may be mistaken for familial focal segmental glomerulosclerosis. Kidney Int. 2014;86(6):1253-1259. doi:10.1038/ki.2014.305