User login
Late ERCP After Cholecystectomy Linked with Worse Outcomes
, according to investigators.
These findings suggest a need for more careful patient selection with ERCP, and greater reliance upon noninvasive imaging prior to considering the procedure, reported lead author Nikhil R. Thiruvengadam, MD, of Loma Linda University Health, Loma Linda, California, and colleagues.
“It is assumed that cholecystectomy is a definitive procedure for symptomatic gallstone disease in patients without concomitant choledocholithiasis,” the investigators wrote in Clinical Gastroenterology and Hepatology. “This is because the development of primary choledocholithiasis is rare. Despite this, many patients have persistent or new gastrointestinal symptoms post cholecystectomy.”
Symptoms such as a dilated bile duct or abnormal liver function tests may suggest choledocholithiasis or sphincter of Oddi disorders (SOD), they noted, but recent data supporting ERCP for SOD show no significant benefit for patients with normal-sized ducts.
“Guidelines advocate for confirming the presence of choledocholithiasis using magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasound (EUS) given the substantial risks associated with ERCP,” Dr. Thiruvengadam and colleagues wrote.
Real-world implementation of this and associated strategies, however, remain unclear, prompting the present study.
The dataset, drawn from the Optum Clinformatics Data Mart, included 583,712 adults who had undergone cholecystectomy from 2004 to 2019, focusing on 4274 individuals who had their first ERCP more than one year post surgery. The investigators assessed the incidence, characteristics, and outcomes of these late ERCP procedures, exploring their association with patient comorbidities and the use of biliary imaging techniques such as MRCP and EUS.
From 2004 to 2021, use of noninvasive biliary imaging approximately doubled from 35.9% to 65.5% (P < .001). Yet incidence of first-time ERCP more than 1 year after cholecystectomy increased much more — by eightfold — from 0.5 to 4.2 per 1000 person-years (P < .001). Less than half (44%) of these late ERCP procedures involved gallstone removal.
Patients undergoing late ERCP were more likely to have higher baseline comorbidities, including disorders of gut-brain interaction (DGBI) and metabolic dysfunction-associated steatotic liver disease. They were also more likely to be taking an antispasmodic, anxiolytic, or chronic opioid medication.
“Late ERCP is more common and associated with worse outcomes, presumably because of higher baseline comorbidities that overlap with DGBI and mimickers of choledocholithiasis,” the investigators noted. “These highly symptomatic individuals are more likely to undergo noninvasive biliary imaging, which seems to be prompting more late ERCP.”
In turn, late ERCP is incurring more adverse events, including post-ERCP pancreatitis (7.1%), hospitalization (13.1%), and new chronic opioid use (9.7%).
“Given the known risks of ERCP, especially in this context, there remains a need to be more restrictive with offering ERCP in this setting,” Dr. Thiruvengadam and colleagues concluded. “ERCP should be used sparingly for patients who do not have confirmed choledocholithiasis until future studies ... can define which patients with a remote history of cholecystectomy respond to ERCP interventions.”
The investigators disclosed relationships with Olympus, Medtronic, ACI, and others.
, according to investigators.
These findings suggest a need for more careful patient selection with ERCP, and greater reliance upon noninvasive imaging prior to considering the procedure, reported lead author Nikhil R. Thiruvengadam, MD, of Loma Linda University Health, Loma Linda, California, and colleagues.
“It is assumed that cholecystectomy is a definitive procedure for symptomatic gallstone disease in patients without concomitant choledocholithiasis,” the investigators wrote in Clinical Gastroenterology and Hepatology. “This is because the development of primary choledocholithiasis is rare. Despite this, many patients have persistent or new gastrointestinal symptoms post cholecystectomy.”
Symptoms such as a dilated bile duct or abnormal liver function tests may suggest choledocholithiasis or sphincter of Oddi disorders (SOD), they noted, but recent data supporting ERCP for SOD show no significant benefit for patients with normal-sized ducts.
“Guidelines advocate for confirming the presence of choledocholithiasis using magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasound (EUS) given the substantial risks associated with ERCP,” Dr. Thiruvengadam and colleagues wrote.
Real-world implementation of this and associated strategies, however, remain unclear, prompting the present study.
The dataset, drawn from the Optum Clinformatics Data Mart, included 583,712 adults who had undergone cholecystectomy from 2004 to 2019, focusing on 4274 individuals who had their first ERCP more than one year post surgery. The investigators assessed the incidence, characteristics, and outcomes of these late ERCP procedures, exploring their association with patient comorbidities and the use of biliary imaging techniques such as MRCP and EUS.
From 2004 to 2021, use of noninvasive biliary imaging approximately doubled from 35.9% to 65.5% (P < .001). Yet incidence of first-time ERCP more than 1 year after cholecystectomy increased much more — by eightfold — from 0.5 to 4.2 per 1000 person-years (P < .001). Less than half (44%) of these late ERCP procedures involved gallstone removal.
Patients undergoing late ERCP were more likely to have higher baseline comorbidities, including disorders of gut-brain interaction (DGBI) and metabolic dysfunction-associated steatotic liver disease. They were also more likely to be taking an antispasmodic, anxiolytic, or chronic opioid medication.
“Late ERCP is more common and associated with worse outcomes, presumably because of higher baseline comorbidities that overlap with DGBI and mimickers of choledocholithiasis,” the investigators noted. “These highly symptomatic individuals are more likely to undergo noninvasive biliary imaging, which seems to be prompting more late ERCP.”
In turn, late ERCP is incurring more adverse events, including post-ERCP pancreatitis (7.1%), hospitalization (13.1%), and new chronic opioid use (9.7%).
“Given the known risks of ERCP, especially in this context, there remains a need to be more restrictive with offering ERCP in this setting,” Dr. Thiruvengadam and colleagues concluded. “ERCP should be used sparingly for patients who do not have confirmed choledocholithiasis until future studies ... can define which patients with a remote history of cholecystectomy respond to ERCP interventions.”
The investigators disclosed relationships with Olympus, Medtronic, ACI, and others.
, according to investigators.
These findings suggest a need for more careful patient selection with ERCP, and greater reliance upon noninvasive imaging prior to considering the procedure, reported lead author Nikhil R. Thiruvengadam, MD, of Loma Linda University Health, Loma Linda, California, and colleagues.
“It is assumed that cholecystectomy is a definitive procedure for symptomatic gallstone disease in patients without concomitant choledocholithiasis,” the investigators wrote in Clinical Gastroenterology and Hepatology. “This is because the development of primary choledocholithiasis is rare. Despite this, many patients have persistent or new gastrointestinal symptoms post cholecystectomy.”
Symptoms such as a dilated bile duct or abnormal liver function tests may suggest choledocholithiasis or sphincter of Oddi disorders (SOD), they noted, but recent data supporting ERCP for SOD show no significant benefit for patients with normal-sized ducts.
“Guidelines advocate for confirming the presence of choledocholithiasis using magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasound (EUS) given the substantial risks associated with ERCP,” Dr. Thiruvengadam and colleagues wrote.
Real-world implementation of this and associated strategies, however, remain unclear, prompting the present study.
The dataset, drawn from the Optum Clinformatics Data Mart, included 583,712 adults who had undergone cholecystectomy from 2004 to 2019, focusing on 4274 individuals who had their first ERCP more than one year post surgery. The investigators assessed the incidence, characteristics, and outcomes of these late ERCP procedures, exploring their association with patient comorbidities and the use of biliary imaging techniques such as MRCP and EUS.
From 2004 to 2021, use of noninvasive biliary imaging approximately doubled from 35.9% to 65.5% (P < .001). Yet incidence of first-time ERCP more than 1 year after cholecystectomy increased much more — by eightfold — from 0.5 to 4.2 per 1000 person-years (P < .001). Less than half (44%) of these late ERCP procedures involved gallstone removal.
Patients undergoing late ERCP were more likely to have higher baseline comorbidities, including disorders of gut-brain interaction (DGBI) and metabolic dysfunction-associated steatotic liver disease. They were also more likely to be taking an antispasmodic, anxiolytic, or chronic opioid medication.
“Late ERCP is more common and associated with worse outcomes, presumably because of higher baseline comorbidities that overlap with DGBI and mimickers of choledocholithiasis,” the investigators noted. “These highly symptomatic individuals are more likely to undergo noninvasive biliary imaging, which seems to be prompting more late ERCP.”
In turn, late ERCP is incurring more adverse events, including post-ERCP pancreatitis (7.1%), hospitalization (13.1%), and new chronic opioid use (9.7%).
“Given the known risks of ERCP, especially in this context, there remains a need to be more restrictive with offering ERCP in this setting,” Dr. Thiruvengadam and colleagues concluded. “ERCP should be used sparingly for patients who do not have confirmed choledocholithiasis until future studies ... can define which patients with a remote history of cholecystectomy respond to ERCP interventions.”
The investigators disclosed relationships with Olympus, Medtronic, ACI, and others.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
August 2024 – ICYMI
Gastroenterology
April 2024
Shah I, et al. Disparities in Colorectal Cancer Screening Among Asian American Populations and Strategies to Address These Disparities. Gastroenterology. 2024 Apr;166(4):549-552. doi: 10.1053/j.gastro.2024.02.009. PMID: 38521575.
Shiha MG, et al. Accuracy of the No-Biopsy Approach for the Diagnosis of Celiac Disease in Adults: A Systematic Review and Meta-Analysis. Gastroenterology. 2024 Apr;166(4):620-630. doi: 10.1053/j.gastro.2023.12.023. Epub 2024 Jan 2. PMID: 38176661.
Goltstein LCMJ, et al. Standard of Care Versus Octreotide in Angiodysplasia-Related Bleeding (the OCEAN Study): A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Apr;166(4):690-703. doi: 10.1053/j.gastro.2023.12.020. Epub 2023 Dec 28. PMID: 38158089.
May 2024
Robertson DJ, et al. Colonoscopy vs the Fecal Immunochemical Test: Which is Best? Gastroenterology. 2024 May;166(5):758-771. doi: 10.1053/j.gastro.2023.12.027. Epub 2024 Feb 9. PMID: 38342196.
Mårild K, et al. Histologic Remission in Inflammatory Bowel Disease and Female Fertility: A Nationwide Study. Gastroenterology. 2024 May;166(5):802-814.e18. doi: 10.1053/j.gastro.2024.01.018. Epub 2024 Feb 6. PMID: 38331202.
June 2024
Trivedi PJ, et al. Immunopathogenesis of Primary Biliary Cholangitis, Primary Sclerosing Cholangitis and Autoimmune Hepatitis: Themes and Concepts. Gastroenterology. 2024 Jun;166(6):995-1019. doi: 10.1053/j.gastro.2024.01.049. Epub 2024 Feb 10. PMID: 38342195.
Rubenstein JH, et al. AGA Clinical Practice Guideline on Endoscopic Eradication Therapy of Barrett’s Esophagus and Related Neoplasia. Gastroenterology. 2024 Jun;166(6):1020-1055. doi: 10.1053/j.gastro.2024.03.019. PMID: 38763697.
Ridtitid W, et al. Endoscopic Gallbladder Stenting to Prevent Recurrent Cholecystitis in Deferred Cholecystectomy: A Randomized Trial. Gastroenterology. 2024 Jun;166(6):1145-1155. doi: 10.1053/j.gastro.2024.02.007. Epub 2024 Feb 14. PMID: 38360274.
Clinical Gastroenterology and Hepatology
April 2024
Berwald G, et al. The Diagnostic Performance of Fecal Immunochemical Tests for Detecting Advanced Neoplasia at Surveillance Colonoscopy. Clin Gastroenterol Hepatol. 2024 Apr;22(4):878-885.e2. doi: 10.1016/j.cgh.2023.09.016. Epub 2023 Sep 22. PMID: 37743036.
Hashash JG, et al. AGA Rapid Clinical Practice Update on the Management of Patients Taking GLP-1 Receptor Agonists Prior to Endoscopy: Communication. Clin Gastroenterol Hepatol. 2024 Apr;22(4):705-707. doi: 10.1016/j.cgh.2023.11.002. Epub 2023 Nov 7. PMID: 37944573.
Sharma R, et al. Statins Are Associated With a Decreased Risk of Severe Liver Disease in Individuals With Noncirrhotic Chronic Liver Disease. Clin Gastroenterol Hepatol. 2024 Apr;22(4):749-759.e19. doi: 10.1016/j.cgh.2023.04.017. Epub 2023 Apr 28. PMID: 37121528.
May 2024
Overbeek KA, et al; PrescrAIP Study Group. Type 1 Autoimmune Pancreatitis in Europe: Clinical Profile and Response to Treatment. Clin Gastroenterol Hepatol. 2024 May;22(5):994-1004.e10. doi: 10.1016/j.cgh.2023.12.010. Epub 2024 Jan 5. Erratum in: Clin Gastroenterol Hepatol. 2024 Jun 1:S1542-3565(24)00446-4. doi: 10.1016/j.cgh.2024.05.005. PMID: 38184096.
Jairath V, et al. ENTERPRET: A Randomized Controlled Trial of Vedolizumab Dose Optimization in Patients With Ulcerative Colitis Who Have Early Nonresponse. Clin Gastroenterol Hepatol. 2024 May;22(5):1077-1086.e13. doi: 10.1016/j.cgh.2023.10.029. Epub 2023 Nov 10. PMID: 37951560.
Gunby SA, et al. Smoking and Alcohol Consumption and Risk of Incident Diverticulitis in Women. Clin Gastroenterol Hepatol. 2024 May;22(5):1108-1116. doi: 10.1016/j.cgh.2023.11.036. Epub 2023 Dec 19. PMID: 38122959; PMCID: PMC11045313.
June 2024
Krause AJ, et al. Validated Clinical Score to Predict Gastroesophageal Reflux in Patients With Chronic Laryngeal Symptoms: COuGH RefluX. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1200-1209.e1. doi: 10.1016/j.cgh.2024.01.021. Epub 2024 Feb 2. PMID: 38309491; PMCID: PMC11128352.
Peng X, et al. Efficacy and Safety of Vonoprazan-Amoxicillin Dual Regimen With Varying Dose and Duration for Helicobacter pylori Eradication: A Multicenter, Prospective, Randomized Study. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1210-1216. doi: 10.1016/j.cgh.2024.01.022. Epub 2024 Feb 1. PMID: 38309492.
Kedia S, et al. Coconut Water Induces Clinical Remission in Mild to Moderate Ulcerative Colitis: Double-blind Placebo-controlled Trial. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1295-1306.e7. doi: 10.1016/j.cgh.2024.01.013. Epub 2024 Jan 24. PMID: 38278200.
Techniques and Innovations in Gastrointestinal Endoscopy
Ogura T, et al. Step-Up Strategy for Endoscopic Hemostasis Using PuraStat After Endoscopic Sphincterotomy Bleeding (STOP Trial). Tech Innov Gastrointest Endosc. 2024 March 16. doi: 10.1016/j.tige.2024.03.005.
Nakai Y, et al. Cyst Detection Rate: A Quality Indicator in the Era of Pancreatic Screening Endoscopic Ultrasonography. Tech Innov Gastrointest Endosc. 2024 May. doi: 10.1016/j.tige.2024.04.001.
Gastro Hep Advances
Kimura Y, et al. Early Sonographic Improvement Predicts Clinical Remission and Mucosal Healing With Molecular-Targeted Drugs in Ulcerative Colitis. Gastro Hep Adv. 2024 April 22. doi: 10.1016/j.gastha.2024.04.007.
Hunaut T, et al. Long-Term Neoplastic Risk Associated With Colorectal Strictures in Crohn’s Disease: A Multicenter Study. Gastro Hep Adv. 2024 May 15. doi: 10.1016/j.gastha.2024.05.003.
Gastroenterology
April 2024
Shah I, et al. Disparities in Colorectal Cancer Screening Among Asian American Populations and Strategies to Address These Disparities. Gastroenterology. 2024 Apr;166(4):549-552. doi: 10.1053/j.gastro.2024.02.009. PMID: 38521575.
Shiha MG, et al. Accuracy of the No-Biopsy Approach for the Diagnosis of Celiac Disease in Adults: A Systematic Review and Meta-Analysis. Gastroenterology. 2024 Apr;166(4):620-630. doi: 10.1053/j.gastro.2023.12.023. Epub 2024 Jan 2. PMID: 38176661.
Goltstein LCMJ, et al. Standard of Care Versus Octreotide in Angiodysplasia-Related Bleeding (the OCEAN Study): A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Apr;166(4):690-703. doi: 10.1053/j.gastro.2023.12.020. Epub 2023 Dec 28. PMID: 38158089.
May 2024
Robertson DJ, et al. Colonoscopy vs the Fecal Immunochemical Test: Which is Best? Gastroenterology. 2024 May;166(5):758-771. doi: 10.1053/j.gastro.2023.12.027. Epub 2024 Feb 9. PMID: 38342196.
Mårild K, et al. Histologic Remission in Inflammatory Bowel Disease and Female Fertility: A Nationwide Study. Gastroenterology. 2024 May;166(5):802-814.e18. doi: 10.1053/j.gastro.2024.01.018. Epub 2024 Feb 6. PMID: 38331202.
June 2024
Trivedi PJ, et al. Immunopathogenesis of Primary Biliary Cholangitis, Primary Sclerosing Cholangitis and Autoimmune Hepatitis: Themes and Concepts. Gastroenterology. 2024 Jun;166(6):995-1019. doi: 10.1053/j.gastro.2024.01.049. Epub 2024 Feb 10. PMID: 38342195.
Rubenstein JH, et al. AGA Clinical Practice Guideline on Endoscopic Eradication Therapy of Barrett’s Esophagus and Related Neoplasia. Gastroenterology. 2024 Jun;166(6):1020-1055. doi: 10.1053/j.gastro.2024.03.019. PMID: 38763697.
Ridtitid W, et al. Endoscopic Gallbladder Stenting to Prevent Recurrent Cholecystitis in Deferred Cholecystectomy: A Randomized Trial. Gastroenterology. 2024 Jun;166(6):1145-1155. doi: 10.1053/j.gastro.2024.02.007. Epub 2024 Feb 14. PMID: 38360274.
Clinical Gastroenterology and Hepatology
April 2024
Berwald G, et al. The Diagnostic Performance of Fecal Immunochemical Tests for Detecting Advanced Neoplasia at Surveillance Colonoscopy. Clin Gastroenterol Hepatol. 2024 Apr;22(4):878-885.e2. doi: 10.1016/j.cgh.2023.09.016. Epub 2023 Sep 22. PMID: 37743036.
Hashash JG, et al. AGA Rapid Clinical Practice Update on the Management of Patients Taking GLP-1 Receptor Agonists Prior to Endoscopy: Communication. Clin Gastroenterol Hepatol. 2024 Apr;22(4):705-707. doi: 10.1016/j.cgh.2023.11.002. Epub 2023 Nov 7. PMID: 37944573.
Sharma R, et al. Statins Are Associated With a Decreased Risk of Severe Liver Disease in Individuals With Noncirrhotic Chronic Liver Disease. Clin Gastroenterol Hepatol. 2024 Apr;22(4):749-759.e19. doi: 10.1016/j.cgh.2023.04.017. Epub 2023 Apr 28. PMID: 37121528.
May 2024
Overbeek KA, et al; PrescrAIP Study Group. Type 1 Autoimmune Pancreatitis in Europe: Clinical Profile and Response to Treatment. Clin Gastroenterol Hepatol. 2024 May;22(5):994-1004.e10. doi: 10.1016/j.cgh.2023.12.010. Epub 2024 Jan 5. Erratum in: Clin Gastroenterol Hepatol. 2024 Jun 1:S1542-3565(24)00446-4. doi: 10.1016/j.cgh.2024.05.005. PMID: 38184096.
Jairath V, et al. ENTERPRET: A Randomized Controlled Trial of Vedolizumab Dose Optimization in Patients With Ulcerative Colitis Who Have Early Nonresponse. Clin Gastroenterol Hepatol. 2024 May;22(5):1077-1086.e13. doi: 10.1016/j.cgh.2023.10.029. Epub 2023 Nov 10. PMID: 37951560.
Gunby SA, et al. Smoking and Alcohol Consumption and Risk of Incident Diverticulitis in Women. Clin Gastroenterol Hepatol. 2024 May;22(5):1108-1116. doi: 10.1016/j.cgh.2023.11.036. Epub 2023 Dec 19. PMID: 38122959; PMCID: PMC11045313.
June 2024
Krause AJ, et al. Validated Clinical Score to Predict Gastroesophageal Reflux in Patients With Chronic Laryngeal Symptoms: COuGH RefluX. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1200-1209.e1. doi: 10.1016/j.cgh.2024.01.021. Epub 2024 Feb 2. PMID: 38309491; PMCID: PMC11128352.
Peng X, et al. Efficacy and Safety of Vonoprazan-Amoxicillin Dual Regimen With Varying Dose and Duration for Helicobacter pylori Eradication: A Multicenter, Prospective, Randomized Study. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1210-1216. doi: 10.1016/j.cgh.2024.01.022. Epub 2024 Feb 1. PMID: 38309492.
Kedia S, et al. Coconut Water Induces Clinical Remission in Mild to Moderate Ulcerative Colitis: Double-blind Placebo-controlled Trial. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1295-1306.e7. doi: 10.1016/j.cgh.2024.01.013. Epub 2024 Jan 24. PMID: 38278200.
Techniques and Innovations in Gastrointestinal Endoscopy
Ogura T, et al. Step-Up Strategy for Endoscopic Hemostasis Using PuraStat After Endoscopic Sphincterotomy Bleeding (STOP Trial). Tech Innov Gastrointest Endosc. 2024 March 16. doi: 10.1016/j.tige.2024.03.005.
Nakai Y, et al. Cyst Detection Rate: A Quality Indicator in the Era of Pancreatic Screening Endoscopic Ultrasonography. Tech Innov Gastrointest Endosc. 2024 May. doi: 10.1016/j.tige.2024.04.001.
Gastro Hep Advances
Kimura Y, et al. Early Sonographic Improvement Predicts Clinical Remission and Mucosal Healing With Molecular-Targeted Drugs in Ulcerative Colitis. Gastro Hep Adv. 2024 April 22. doi: 10.1016/j.gastha.2024.04.007.
Hunaut T, et al. Long-Term Neoplastic Risk Associated With Colorectal Strictures in Crohn’s Disease: A Multicenter Study. Gastro Hep Adv. 2024 May 15. doi: 10.1016/j.gastha.2024.05.003.
Gastroenterology
April 2024
Shah I, et al. Disparities in Colorectal Cancer Screening Among Asian American Populations and Strategies to Address These Disparities. Gastroenterology. 2024 Apr;166(4):549-552. doi: 10.1053/j.gastro.2024.02.009. PMID: 38521575.
Shiha MG, et al. Accuracy of the No-Biopsy Approach for the Diagnosis of Celiac Disease in Adults: A Systematic Review and Meta-Analysis. Gastroenterology. 2024 Apr;166(4):620-630. doi: 10.1053/j.gastro.2023.12.023. Epub 2024 Jan 2. PMID: 38176661.
Goltstein LCMJ, et al. Standard of Care Versus Octreotide in Angiodysplasia-Related Bleeding (the OCEAN Study): A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Apr;166(4):690-703. doi: 10.1053/j.gastro.2023.12.020. Epub 2023 Dec 28. PMID: 38158089.
May 2024
Robertson DJ, et al. Colonoscopy vs the Fecal Immunochemical Test: Which is Best? Gastroenterology. 2024 May;166(5):758-771. doi: 10.1053/j.gastro.2023.12.027. Epub 2024 Feb 9. PMID: 38342196.
Mårild K, et al. Histologic Remission in Inflammatory Bowel Disease and Female Fertility: A Nationwide Study. Gastroenterology. 2024 May;166(5):802-814.e18. doi: 10.1053/j.gastro.2024.01.018. Epub 2024 Feb 6. PMID: 38331202.
June 2024
Trivedi PJ, et al. Immunopathogenesis of Primary Biliary Cholangitis, Primary Sclerosing Cholangitis and Autoimmune Hepatitis: Themes and Concepts. Gastroenterology. 2024 Jun;166(6):995-1019. doi: 10.1053/j.gastro.2024.01.049. Epub 2024 Feb 10. PMID: 38342195.
Rubenstein JH, et al. AGA Clinical Practice Guideline on Endoscopic Eradication Therapy of Barrett’s Esophagus and Related Neoplasia. Gastroenterology. 2024 Jun;166(6):1020-1055. doi: 10.1053/j.gastro.2024.03.019. PMID: 38763697.
Ridtitid W, et al. Endoscopic Gallbladder Stenting to Prevent Recurrent Cholecystitis in Deferred Cholecystectomy: A Randomized Trial. Gastroenterology. 2024 Jun;166(6):1145-1155. doi: 10.1053/j.gastro.2024.02.007. Epub 2024 Feb 14. PMID: 38360274.
Clinical Gastroenterology and Hepatology
April 2024
Berwald G, et al. The Diagnostic Performance of Fecal Immunochemical Tests for Detecting Advanced Neoplasia at Surveillance Colonoscopy. Clin Gastroenterol Hepatol. 2024 Apr;22(4):878-885.e2. doi: 10.1016/j.cgh.2023.09.016. Epub 2023 Sep 22. PMID: 37743036.
Hashash JG, et al. AGA Rapid Clinical Practice Update on the Management of Patients Taking GLP-1 Receptor Agonists Prior to Endoscopy: Communication. Clin Gastroenterol Hepatol. 2024 Apr;22(4):705-707. doi: 10.1016/j.cgh.2023.11.002. Epub 2023 Nov 7. PMID: 37944573.
Sharma R, et al. Statins Are Associated With a Decreased Risk of Severe Liver Disease in Individuals With Noncirrhotic Chronic Liver Disease. Clin Gastroenterol Hepatol. 2024 Apr;22(4):749-759.e19. doi: 10.1016/j.cgh.2023.04.017. Epub 2023 Apr 28. PMID: 37121528.
May 2024
Overbeek KA, et al; PrescrAIP Study Group. Type 1 Autoimmune Pancreatitis in Europe: Clinical Profile and Response to Treatment. Clin Gastroenterol Hepatol. 2024 May;22(5):994-1004.e10. doi: 10.1016/j.cgh.2023.12.010. Epub 2024 Jan 5. Erratum in: Clin Gastroenterol Hepatol. 2024 Jun 1:S1542-3565(24)00446-4. doi: 10.1016/j.cgh.2024.05.005. PMID: 38184096.
Jairath V, et al. ENTERPRET: A Randomized Controlled Trial of Vedolizumab Dose Optimization in Patients With Ulcerative Colitis Who Have Early Nonresponse. Clin Gastroenterol Hepatol. 2024 May;22(5):1077-1086.e13. doi: 10.1016/j.cgh.2023.10.029. Epub 2023 Nov 10. PMID: 37951560.
Gunby SA, et al. Smoking and Alcohol Consumption and Risk of Incident Diverticulitis in Women. Clin Gastroenterol Hepatol. 2024 May;22(5):1108-1116. doi: 10.1016/j.cgh.2023.11.036. Epub 2023 Dec 19. PMID: 38122959; PMCID: PMC11045313.
June 2024
Krause AJ, et al. Validated Clinical Score to Predict Gastroesophageal Reflux in Patients With Chronic Laryngeal Symptoms: COuGH RefluX. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1200-1209.e1. doi: 10.1016/j.cgh.2024.01.021. Epub 2024 Feb 2. PMID: 38309491; PMCID: PMC11128352.
Peng X, et al. Efficacy and Safety of Vonoprazan-Amoxicillin Dual Regimen With Varying Dose and Duration for Helicobacter pylori Eradication: A Multicenter, Prospective, Randomized Study. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1210-1216. doi: 10.1016/j.cgh.2024.01.022. Epub 2024 Feb 1. PMID: 38309492.
Kedia S, et al. Coconut Water Induces Clinical Remission in Mild to Moderate Ulcerative Colitis: Double-blind Placebo-controlled Trial. Clin Gastroenterol Hepatol. 2024 Jun;22(6):1295-1306.e7. doi: 10.1016/j.cgh.2024.01.013. Epub 2024 Jan 24. PMID: 38278200.
Techniques and Innovations in Gastrointestinal Endoscopy
Ogura T, et al. Step-Up Strategy for Endoscopic Hemostasis Using PuraStat After Endoscopic Sphincterotomy Bleeding (STOP Trial). Tech Innov Gastrointest Endosc. 2024 March 16. doi: 10.1016/j.tige.2024.03.005.
Nakai Y, et al. Cyst Detection Rate: A Quality Indicator in the Era of Pancreatic Screening Endoscopic Ultrasonography. Tech Innov Gastrointest Endosc. 2024 May. doi: 10.1016/j.tige.2024.04.001.
Gastro Hep Advances
Kimura Y, et al. Early Sonographic Improvement Predicts Clinical Remission and Mucosal Healing With Molecular-Targeted Drugs in Ulcerative Colitis. Gastro Hep Adv. 2024 April 22. doi: 10.1016/j.gastha.2024.04.007.
Hunaut T, et al. Long-Term Neoplastic Risk Associated With Colorectal Strictures in Crohn’s Disease: A Multicenter Study. Gastro Hep Adv. 2024 May 15. doi: 10.1016/j.gastha.2024.05.003.
FDA OKs Iqirvo, First-in-Class PPAR Treatment for Primary Biliary Cholangitis
in combination with ursodeoxycholic acid (UDCA) in adults who do not respond adequately to UDCA or as monotherapy in patients unable to tolerate UDCA.
PBC is a rare, chronic cholestatic liver disease that destroys interlobular bile ducts and leads to cholestasis and liver fibrosis. Left untreated, the disease can worsen over time, leading to cirrhosis and liver transplant and, in some cases, premature death. PBC also harms quality of life, with patients often experiencing severe fatigue and pruritus.
Iqirvo, an oral dual peroxisome proliferator–activated receptor (PPAR) alpha and delta agonist, is the first new drug approved in nearly a decade for treatment of PBC.
Accelerated approval of Iqirvo for PBC was based on data from the phase 3 ELATIVE trial published last year in The New England Journal of Medicine.
The trial randomly assigned patients with PBC who had an inadequate response to or unacceptable side effects with UDCA to receive either once-daily elafibranor (80 mg) or placebo.
The primary endpoint was a biochemical response, defined as an alkaline phosphatase (ALP) level < 1.67 times the upper limit of the normal range, with a reduction ≥ 15% from baseline, as well as normal total bilirubin levels.
Among 161 patients, a biochemical response was seen in 55 of 108 (51%) who received elafibranor vs 2 of 53 (4%) who received placebo.
At week 52, the ALP level normalized in 15% of patients in the elafibranor group and none of the patients in the placebo group.
In a news release announcing approval of Iqirvo, the company notes that improvement in survival and prevention of liver decompensation events have not been demonstrated and that continued approval for PBC may be contingent upon verification and description of clinical benefit in confirmatory trials.
The most common adverse effects with Iqirvo, reported in ≥ 10% of study participants, were weight gain, abdominal pain, diarrhea, nausea, and vomiting. Iqirvo is not recommended for people who have or develop decompensated cirrhosis. Full prescribing information is available online.
The data show that Iqirvo is “an effective second-line treatment for patients with PBC with favorable benefit and risk data,” Kris Kowdley, MD, AGAF, director of the Liver Institute Northwest in Seattle, Washington, and a primary investigator on the ELATIVE study, said in the news release.
The approval of Iqirvo “will allow healthcare providers in the US to address an unmet need with the potential to significantly reduce ALP levels for our patients with PBC,” Dr. Kowdley said.
A version of this article appeared on Medscape.com.
in combination with ursodeoxycholic acid (UDCA) in adults who do not respond adequately to UDCA or as monotherapy in patients unable to tolerate UDCA.
PBC is a rare, chronic cholestatic liver disease that destroys interlobular bile ducts and leads to cholestasis and liver fibrosis. Left untreated, the disease can worsen over time, leading to cirrhosis and liver transplant and, in some cases, premature death. PBC also harms quality of life, with patients often experiencing severe fatigue and pruritus.
Iqirvo, an oral dual peroxisome proliferator–activated receptor (PPAR) alpha and delta agonist, is the first new drug approved in nearly a decade for treatment of PBC.
Accelerated approval of Iqirvo for PBC was based on data from the phase 3 ELATIVE trial published last year in The New England Journal of Medicine.
The trial randomly assigned patients with PBC who had an inadequate response to or unacceptable side effects with UDCA to receive either once-daily elafibranor (80 mg) or placebo.
The primary endpoint was a biochemical response, defined as an alkaline phosphatase (ALP) level < 1.67 times the upper limit of the normal range, with a reduction ≥ 15% from baseline, as well as normal total bilirubin levels.
Among 161 patients, a biochemical response was seen in 55 of 108 (51%) who received elafibranor vs 2 of 53 (4%) who received placebo.
At week 52, the ALP level normalized in 15% of patients in the elafibranor group and none of the patients in the placebo group.
In a news release announcing approval of Iqirvo, the company notes that improvement in survival and prevention of liver decompensation events have not been demonstrated and that continued approval for PBC may be contingent upon verification and description of clinical benefit in confirmatory trials.
The most common adverse effects with Iqirvo, reported in ≥ 10% of study participants, were weight gain, abdominal pain, diarrhea, nausea, and vomiting. Iqirvo is not recommended for people who have or develop decompensated cirrhosis. Full prescribing information is available online.
The data show that Iqirvo is “an effective second-line treatment for patients with PBC with favorable benefit and risk data,” Kris Kowdley, MD, AGAF, director of the Liver Institute Northwest in Seattle, Washington, and a primary investigator on the ELATIVE study, said in the news release.
The approval of Iqirvo “will allow healthcare providers in the US to address an unmet need with the potential to significantly reduce ALP levels for our patients with PBC,” Dr. Kowdley said.
A version of this article appeared on Medscape.com.
in combination with ursodeoxycholic acid (UDCA) in adults who do not respond adequately to UDCA or as monotherapy in patients unable to tolerate UDCA.
PBC is a rare, chronic cholestatic liver disease that destroys interlobular bile ducts and leads to cholestasis and liver fibrosis. Left untreated, the disease can worsen over time, leading to cirrhosis and liver transplant and, in some cases, premature death. PBC also harms quality of life, with patients often experiencing severe fatigue and pruritus.
Iqirvo, an oral dual peroxisome proliferator–activated receptor (PPAR) alpha and delta agonist, is the first new drug approved in nearly a decade for treatment of PBC.
Accelerated approval of Iqirvo for PBC was based on data from the phase 3 ELATIVE trial published last year in The New England Journal of Medicine.
The trial randomly assigned patients with PBC who had an inadequate response to or unacceptable side effects with UDCA to receive either once-daily elafibranor (80 mg) or placebo.
The primary endpoint was a biochemical response, defined as an alkaline phosphatase (ALP) level < 1.67 times the upper limit of the normal range, with a reduction ≥ 15% from baseline, as well as normal total bilirubin levels.
Among 161 patients, a biochemical response was seen in 55 of 108 (51%) who received elafibranor vs 2 of 53 (4%) who received placebo.
At week 52, the ALP level normalized in 15% of patients in the elafibranor group and none of the patients in the placebo group.
In a news release announcing approval of Iqirvo, the company notes that improvement in survival and prevention of liver decompensation events have not been demonstrated and that continued approval for PBC may be contingent upon verification and description of clinical benefit in confirmatory trials.
The most common adverse effects with Iqirvo, reported in ≥ 10% of study participants, were weight gain, abdominal pain, diarrhea, nausea, and vomiting. Iqirvo is not recommended for people who have or develop decompensated cirrhosis. Full prescribing information is available online.
The data show that Iqirvo is “an effective second-line treatment for patients with PBC with favorable benefit and risk data,” Kris Kowdley, MD, AGAF, director of the Liver Institute Northwest in Seattle, Washington, and a primary investigator on the ELATIVE study, said in the news release.
The approval of Iqirvo “will allow healthcare providers in the US to address an unmet need with the potential to significantly reduce ALP levels for our patients with PBC,” Dr. Kowdley said.
A version of this article appeared on Medscape.com.
Fluid Management in Acute Pancreatitis
Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis [published correction appears in Am J Gastroenterol. 2014;109(2):302]. Am J Gastroenterol. 2013;108(9):1400-1415. doi:10.1038/ajg.2013.218
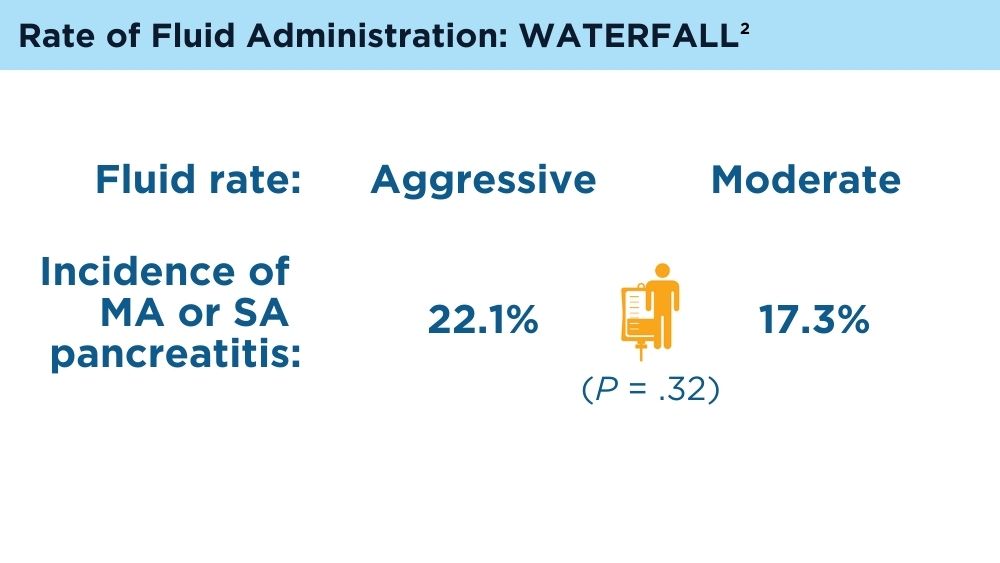
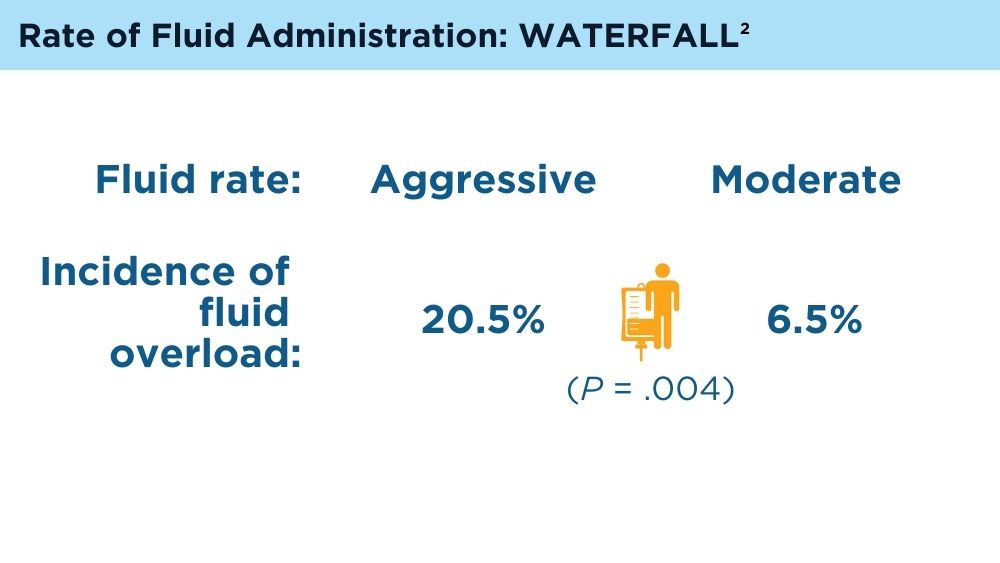
de-Madaria E, Buxbaum JL, Maisonneuve P, et al. Aggressive or moderate fluid resuscitation in acute pancreatitis. N Engl J Med. 2022;387(11):989-1000. doi:10.1056/NEJMoa2202884
Zhao G, Zhang JG, Wu HS, et al. Effects of different resuscitation fluid on severe acute pancreatitis. World J Gastroenterol. 2013;19(13):2044-2052. doi:10.3748/wjg.v19.i13.2044
Guzmán-Calderón E, Diaz-Arocutipa C, Monge E. Lactate Ringer's versus normal saline in the management of acute pancreatitis: a systematic review and meta-analysis of randomized controlled trials. Dig Dis Sci. 2022;67(8):4131-4139. doi:10.1007/s10620-021-07269-8
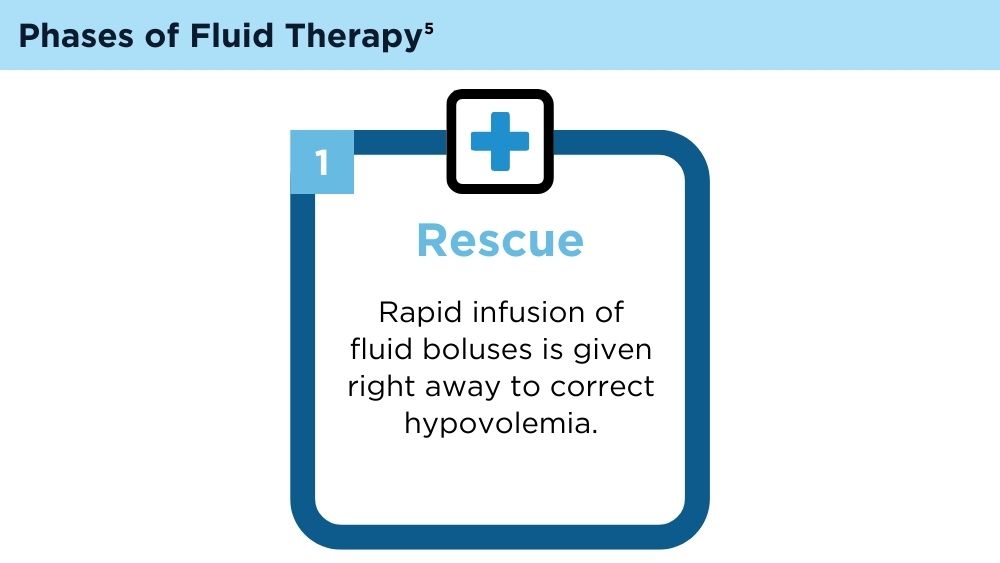
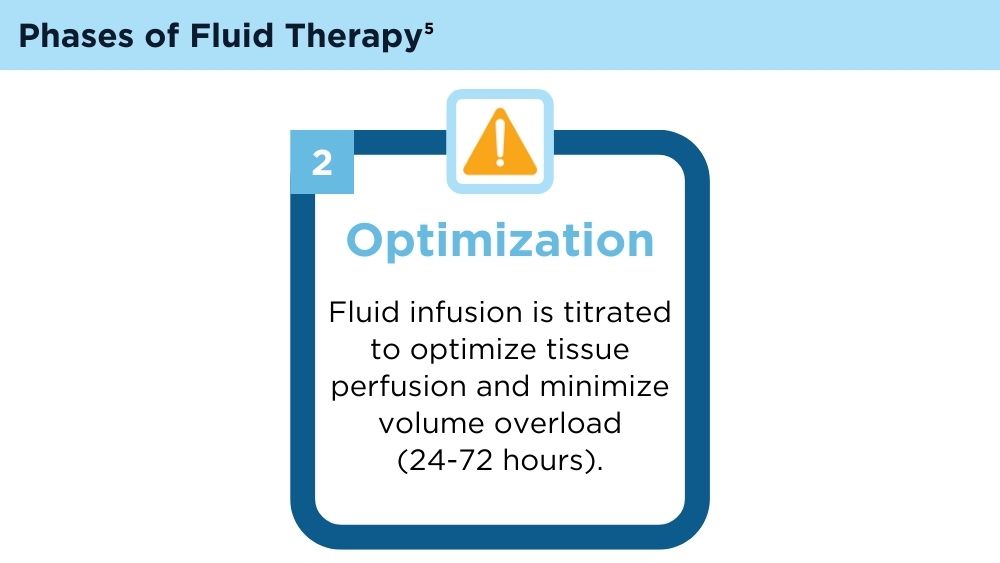
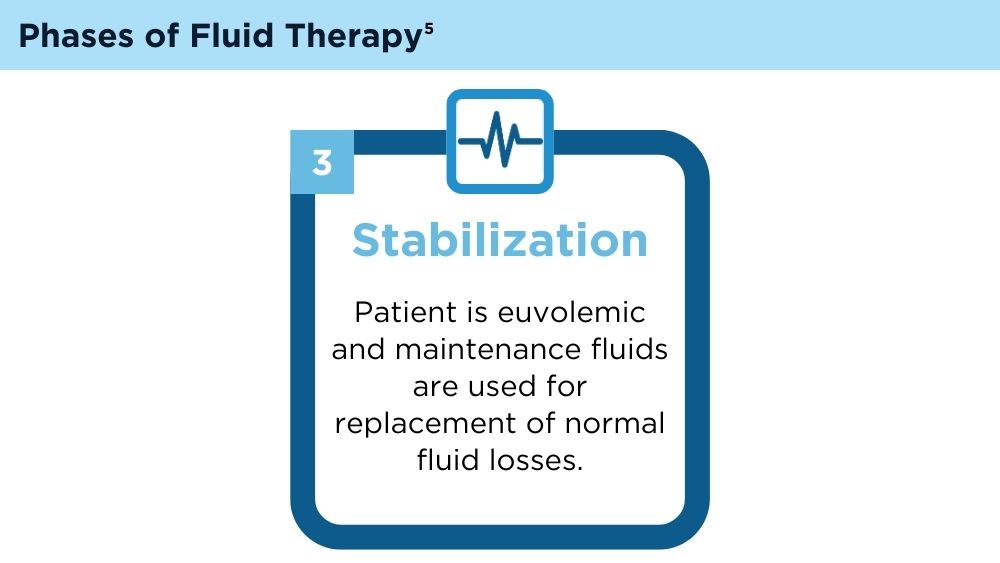
Hoste EA, Maitland K, Brudney CS, et al; ADQI XII Investigators Group. Four phases of intravenous fluid therapy: a conceptual model. Br J Anaesth. 2014;113(5):740-747. doi:10.1093/bja/aeu300
Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 suppl 2):e1-e15. doi:10.1016/j.pan.2013.07.063
Machicado JD, Papachristou GI. Pharmacologic management and prevention of acute pancreatitis. Curr Opin Gastroenterol. 2019;35(5):460-467. doi:10.1097/MOG.0000000000000563
Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis [published correction appears in Am J Gastroenterol. 2014;109(2):302]. Am J Gastroenterol. 2013;108(9):1400-1415. doi:10.1038/ajg.2013.218
de-Madaria E, Buxbaum JL, Maisonneuve P, et al. Aggressive or moderate fluid resuscitation in acute pancreatitis. N Engl J Med. 2022;387(11):989-1000. doi:10.1056/NEJMoa2202884
Zhao G, Zhang JG, Wu HS, et al. Effects of different resuscitation fluid on severe acute pancreatitis. World J Gastroenterol. 2013;19(13):2044-2052. doi:10.3748/wjg.v19.i13.2044
Guzmán-Calderón E, Diaz-Arocutipa C, Monge E. Lactate Ringer's versus normal saline in the management of acute pancreatitis: a systematic review and meta-analysis of randomized controlled trials. Dig Dis Sci. 2022;67(8):4131-4139. doi:10.1007/s10620-021-07269-8
Hoste EA, Maitland K, Brudney CS, et al; ADQI XII Investigators Group. Four phases of intravenous fluid therapy: a conceptual model. Br J Anaesth. 2014;113(5):740-747. doi:10.1093/bja/aeu300
Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 suppl 2):e1-e15. doi:10.1016/j.pan.2013.07.063
Machicado JD, Papachristou GI. Pharmacologic management and prevention of acute pancreatitis. Curr Opin Gastroenterol. 2019;35(5):460-467. doi:10.1097/MOG.0000000000000563
Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis [published correction appears in Am J Gastroenterol. 2014;109(2):302]. Am J Gastroenterol. 2013;108(9):1400-1415. doi:10.1038/ajg.2013.218
de-Madaria E, Buxbaum JL, Maisonneuve P, et al. Aggressive or moderate fluid resuscitation in acute pancreatitis. N Engl J Med. 2022;387(11):989-1000. doi:10.1056/NEJMoa2202884
Zhao G, Zhang JG, Wu HS, et al. Effects of different resuscitation fluid on severe acute pancreatitis. World J Gastroenterol. 2013;19(13):2044-2052. doi:10.3748/wjg.v19.i13.2044
Guzmán-Calderón E, Diaz-Arocutipa C, Monge E. Lactate Ringer's versus normal saline in the management of acute pancreatitis: a systematic review and meta-analysis of randomized controlled trials. Dig Dis Sci. 2022;67(8):4131-4139. doi:10.1007/s10620-021-07269-8
Hoste EA, Maitland K, Brudney CS, et al; ADQI XII Investigators Group. Four phases of intravenous fluid therapy: a conceptual model. Br J Anaesth. 2014;113(5):740-747. doi:10.1093/bja/aeu300
Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13(4 suppl 2):e1-e15. doi:10.1016/j.pan.2013.07.063
Machicado JD, Papachristou GI. Pharmacologic management and prevention of acute pancreatitis. Curr Opin Gastroenterol. 2019;35(5):460-467. doi:10.1097/MOG.0000000000000563
In IBD Patients, Statin Use Associated with Lower Risk of Developing PSC
WASHINGTON — , according to a study presented at Digestive Disease Week® (DDW) 2024.
Statin use was associated with an 86% risk reduction, and only .09% of IBD patients who took statins developed PSC.
“We all take care of patients with liver disease, and we know what a significant burden PSC is. These patients have a significantly elevated risk of enhanced fibrosis and cirrhosis, multiple cancers, and cholangitis and sepsis,” said lead author Chiraag Kulkarni, MD, a gastroenterology fellow at Stanford (California) University Medical School.
“Despite this, we have to date no proven effective medical care for PSC,” he said. “However, over the last decade, there is growing evidence that statins may be beneficial in liver disease, and we see this evidence base stretching from basic science to clinical data.”
Dr. Kulkarni pointed to numerous studies that indicate statins may slow disease progression in steatotic liver disease, viral hepatitis, and cirrhosis. But could statins prevent the onset of PSC?
Because PSC incidence is low, Dr. Kulkarni and colleagues focused on a patient population with higher prevalence — those with IBD, who have an overall lifetime risk of 2% to 7%. The research team followed patients from the date of IBD diagnosis.
Among 33,813 patients with IBD in a national dataset from 2018 onward, 8813 used statins. Statin users tended to be older than non–statin users.
Overall, 181 patients developed new onset PSC during a median follow-up of about 45 months after initial IBD diagnosis. Only eight statin users (.09%) developed PSC, compared with 173 patients (.69%) in the control group.
In a propensity score-matched analysis, statin therapy was associated with a significantly lower risk of developing PSC (HR .14, P < .001). The associated E-value was 5.5, which suggested a robust finding and unlikely to be due to non-visible confounding.
The findings were consistent across secondary and sensitivity analyses, including by age, duration of statin use, and type of statin. For instance, for patients under age 50 where PSC is more likely to occur, statins were associated with a 90% reduction in PSC risk.
“We take away two things from this. First, it’s suggested that a protective effect occurs at ages where PSC is most likely to occur,” Dr. Kulkarni said. “Second, in combination with our propensity score-matched analysis, the results we are observing are not due to a survival bias, where the patients who survive to an age where statins are prescribed simply have a biologically different predilection for developing PSC.”
Statins also protected against PSC in both ulcerative colitis (HR .21) and Crohn’s disease (HR .15), as well as both women (HR .16) and men (HR .22).
Given the uncertainty about the optimal duration of statin therapy for a protective effect, Dr. Kulkarni and colleagues looked at a lag time of 12 months. They found statins were associated with an 84% risk reduction (HR .16), which was similar to the primary analysis.
The study was limited by the inability to capture dosage data or medication adherence. The findings raised several questions, Dr. Kulkarni said, such as the underlying mechanisms and clinical implications. For instance, the underlying mechanisms appear to be related to the pleiotropic effect of statins, modulation of gut inflammation, and alterations in bile acid profiles.
“This is really fascinating and interesting. I wonder about this as a primary prevention strategy in those who have normal cholesterol. Could this work or not?” said Gyongyi Szabo, MD, AGAF, chief academic officer at Beth Israel Deaconess Medical Center, Boston, who was a moderator for the Liver & Biliary Section Distinguished Abstract Plenary Session.
Dr. Kulkarni noted that these findings wouldn’t change clinical practice alone, but based on existing literature around statin hesitancy among patients with cardiovascular disease, the risk reduction for PSC could provide another reason to encourage patients to take them.
“To move this to a place where you can actually think about primary prevention, I think the biological mechanisms need to be teased out a little bit more,” Dr. Kulkarni said. “Then I think you probably still need to identify a higher-risk group than IBD alone.”
Dr. Kulkarni declared no disclosures.
WASHINGTON — , according to a study presented at Digestive Disease Week® (DDW) 2024.
Statin use was associated with an 86% risk reduction, and only .09% of IBD patients who took statins developed PSC.
“We all take care of patients with liver disease, and we know what a significant burden PSC is. These patients have a significantly elevated risk of enhanced fibrosis and cirrhosis, multiple cancers, and cholangitis and sepsis,” said lead author Chiraag Kulkarni, MD, a gastroenterology fellow at Stanford (California) University Medical School.
“Despite this, we have to date no proven effective medical care for PSC,” he said. “However, over the last decade, there is growing evidence that statins may be beneficial in liver disease, and we see this evidence base stretching from basic science to clinical data.”
Dr. Kulkarni pointed to numerous studies that indicate statins may slow disease progression in steatotic liver disease, viral hepatitis, and cirrhosis. But could statins prevent the onset of PSC?
Because PSC incidence is low, Dr. Kulkarni and colleagues focused on a patient population with higher prevalence — those with IBD, who have an overall lifetime risk of 2% to 7%. The research team followed patients from the date of IBD diagnosis.
Among 33,813 patients with IBD in a national dataset from 2018 onward, 8813 used statins. Statin users tended to be older than non–statin users.
Overall, 181 patients developed new onset PSC during a median follow-up of about 45 months after initial IBD diagnosis. Only eight statin users (.09%) developed PSC, compared with 173 patients (.69%) in the control group.
In a propensity score-matched analysis, statin therapy was associated with a significantly lower risk of developing PSC (HR .14, P < .001). The associated E-value was 5.5, which suggested a robust finding and unlikely to be due to non-visible confounding.
The findings were consistent across secondary and sensitivity analyses, including by age, duration of statin use, and type of statin. For instance, for patients under age 50 where PSC is more likely to occur, statins were associated with a 90% reduction in PSC risk.
“We take away two things from this. First, it’s suggested that a protective effect occurs at ages where PSC is most likely to occur,” Dr. Kulkarni said. “Second, in combination with our propensity score-matched analysis, the results we are observing are not due to a survival bias, where the patients who survive to an age where statins are prescribed simply have a biologically different predilection for developing PSC.”
Statins also protected against PSC in both ulcerative colitis (HR .21) and Crohn’s disease (HR .15), as well as both women (HR .16) and men (HR .22).
Given the uncertainty about the optimal duration of statin therapy for a protective effect, Dr. Kulkarni and colleagues looked at a lag time of 12 months. They found statins were associated with an 84% risk reduction (HR .16), which was similar to the primary analysis.
The study was limited by the inability to capture dosage data or medication adherence. The findings raised several questions, Dr. Kulkarni said, such as the underlying mechanisms and clinical implications. For instance, the underlying mechanisms appear to be related to the pleiotropic effect of statins, modulation of gut inflammation, and alterations in bile acid profiles.
“This is really fascinating and interesting. I wonder about this as a primary prevention strategy in those who have normal cholesterol. Could this work or not?” said Gyongyi Szabo, MD, AGAF, chief academic officer at Beth Israel Deaconess Medical Center, Boston, who was a moderator for the Liver & Biliary Section Distinguished Abstract Plenary Session.
Dr. Kulkarni noted that these findings wouldn’t change clinical practice alone, but based on existing literature around statin hesitancy among patients with cardiovascular disease, the risk reduction for PSC could provide another reason to encourage patients to take them.
“To move this to a place where you can actually think about primary prevention, I think the biological mechanisms need to be teased out a little bit more,” Dr. Kulkarni said. “Then I think you probably still need to identify a higher-risk group than IBD alone.”
Dr. Kulkarni declared no disclosures.
WASHINGTON — , according to a study presented at Digestive Disease Week® (DDW) 2024.
Statin use was associated with an 86% risk reduction, and only .09% of IBD patients who took statins developed PSC.
“We all take care of patients with liver disease, and we know what a significant burden PSC is. These patients have a significantly elevated risk of enhanced fibrosis and cirrhosis, multiple cancers, and cholangitis and sepsis,” said lead author Chiraag Kulkarni, MD, a gastroenterology fellow at Stanford (California) University Medical School.
“Despite this, we have to date no proven effective medical care for PSC,” he said. “However, over the last decade, there is growing evidence that statins may be beneficial in liver disease, and we see this evidence base stretching from basic science to clinical data.”
Dr. Kulkarni pointed to numerous studies that indicate statins may slow disease progression in steatotic liver disease, viral hepatitis, and cirrhosis. But could statins prevent the onset of PSC?
Because PSC incidence is low, Dr. Kulkarni and colleagues focused on a patient population with higher prevalence — those with IBD, who have an overall lifetime risk of 2% to 7%. The research team followed patients from the date of IBD diagnosis.
Among 33,813 patients with IBD in a national dataset from 2018 onward, 8813 used statins. Statin users tended to be older than non–statin users.
Overall, 181 patients developed new onset PSC during a median follow-up of about 45 months after initial IBD diagnosis. Only eight statin users (.09%) developed PSC, compared with 173 patients (.69%) in the control group.
In a propensity score-matched analysis, statin therapy was associated with a significantly lower risk of developing PSC (HR .14, P < .001). The associated E-value was 5.5, which suggested a robust finding and unlikely to be due to non-visible confounding.
The findings were consistent across secondary and sensitivity analyses, including by age, duration of statin use, and type of statin. For instance, for patients under age 50 where PSC is more likely to occur, statins were associated with a 90% reduction in PSC risk.
“We take away two things from this. First, it’s suggested that a protective effect occurs at ages where PSC is most likely to occur,” Dr. Kulkarni said. “Second, in combination with our propensity score-matched analysis, the results we are observing are not due to a survival bias, where the patients who survive to an age where statins are prescribed simply have a biologically different predilection for developing PSC.”
Statins also protected against PSC in both ulcerative colitis (HR .21) and Crohn’s disease (HR .15), as well as both women (HR .16) and men (HR .22).
Given the uncertainty about the optimal duration of statin therapy for a protective effect, Dr. Kulkarni and colleagues looked at a lag time of 12 months. They found statins were associated with an 84% risk reduction (HR .16), which was similar to the primary analysis.
The study was limited by the inability to capture dosage data or medication adherence. The findings raised several questions, Dr. Kulkarni said, such as the underlying mechanisms and clinical implications. For instance, the underlying mechanisms appear to be related to the pleiotropic effect of statins, modulation of gut inflammation, and alterations in bile acid profiles.
“This is really fascinating and interesting. I wonder about this as a primary prevention strategy in those who have normal cholesterol. Could this work or not?” said Gyongyi Szabo, MD, AGAF, chief academic officer at Beth Israel Deaconess Medical Center, Boston, who was a moderator for the Liver & Biliary Section Distinguished Abstract Plenary Session.
Dr. Kulkarni noted that these findings wouldn’t change clinical practice alone, but based on existing literature around statin hesitancy among patients with cardiovascular disease, the risk reduction for PSC could provide another reason to encourage patients to take them.
“To move this to a place where you can actually think about primary prevention, I think the biological mechanisms need to be teased out a little bit more,” Dr. Kulkarni said. “Then I think you probably still need to identify a higher-risk group than IBD alone.”
Dr. Kulkarni declared no disclosures.
FROM DDW 2024
May 2024 – ICYMI
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
Liquid Biopsy Has Near-Perfect Accuracy for Early Pancreatic Cancer
the most common type of pancreatic cancer.
It is quite encouraging to know we have a blood test that could potentially find this disease early, said Ajay Goel, PhD, a molecular diagnostics specialist at City of Hope in Duarte, California, who presented the findings at the annual meeting of the American Association for Cancer Research (AACR).
Dr. Goel and colleagues developed a signature for pancreatic cancer based on microRNAs identified in the exomes shed from pancreatic cancers and cell-free DNA markers found in the blood of patients with the disease.
Their initial assay tested blood samples for this signature in a training cohort of 252 people in Japan, approximately 60% of whom had pancreatic cancer. The rest were healthy controls. The assay was then tested in validation cohorts of 400 subjects, half with pancreatic cancer and half controls, in China and South Korea.
In both the initial and validation tests, the microRNA assay had an accuracy of about 90% for stage I/II pancreatic cancer, already far better than commercially available assays.
In an additional validation cohort in the United States with 139 patients with pancreatic cancer and 193 controls at six centers across the country, the researchers found that adding carbohydrate antigen 19-9 — a well-known marker of pancreatic cancer — to the assay boosted the test’s accuracy to 97%.
The test performed the same whether the tumor was in the head or tail of the pancreas.
“We are very excited about this data,” said Dr. Goel.
The technology was recently licensed to Pharus Diagnostics for commercial development, which will likely include a prospective screening trial, he told this news organization.
Because pancreatic cancer is fairly uncommon, Dr. Goel did not anticipate the test being used for general screening but rather for screening high-risk patients such as those with newly diagnosed type 2 diabetes, a family history of pancreatic cancer, or predisposing genetic mutations.
“It should be a very inexpensive test; it doesn’t cost us much to do in the lab,” he added.
Study moderator Ryan Corcoran, MD, PhD, a gastrointestinal (GI) oncologist at Massachusetts General Hospital, Boston, saw the potential.
“As a GI oncologist, I know how lethal and hard to treat pancreatic cancer is,” he said. A test that could reliably detect pancreatic cancer early, with an acceptable false-positive rate, would be extremely useful.
“The cure rate is many, many times higher,” if we detect it before it has a chance to spread, he explained.
In the meantime, Dr. Goel said there’s more work to be done.
Almost 4,000 subjects have been enrolled in ongoing validation efforts, and efforts are underway to use the test to screen thousands of banked blood samples from the PLCO, a prospective cancer screening trial in healthy subjects.
The researchers also want to see if the test can distinguish benign pancreatic cysts from ones that turn cancerous.
The idea is to find the earliest possible signs of this disease to see if we can find it not “at the moment of clinical diagnosis, but possibly 6 months, 1 year, 2 years earlier” than with radiologic imaging, Dr. Goel said.
The work was funded by the National Cancer Institute and others. Dr. Goel is a consultant for Pharus Diagnostics and Cellomics. Dr. Corcoran is a consultant for, has grants from, and/or holds stock in numerous companies, including Pfizer, Novartis, Eli Lilly, and Revolution Medicines.
A version of this article appeared on Medscape.com.
the most common type of pancreatic cancer.
It is quite encouraging to know we have a blood test that could potentially find this disease early, said Ajay Goel, PhD, a molecular diagnostics specialist at City of Hope in Duarte, California, who presented the findings at the annual meeting of the American Association for Cancer Research (AACR).
Dr. Goel and colleagues developed a signature for pancreatic cancer based on microRNAs identified in the exomes shed from pancreatic cancers and cell-free DNA markers found in the blood of patients with the disease.
Their initial assay tested blood samples for this signature in a training cohort of 252 people in Japan, approximately 60% of whom had pancreatic cancer. The rest were healthy controls. The assay was then tested in validation cohorts of 400 subjects, half with pancreatic cancer and half controls, in China and South Korea.
In both the initial and validation tests, the microRNA assay had an accuracy of about 90% for stage I/II pancreatic cancer, already far better than commercially available assays.
In an additional validation cohort in the United States with 139 patients with pancreatic cancer and 193 controls at six centers across the country, the researchers found that adding carbohydrate antigen 19-9 — a well-known marker of pancreatic cancer — to the assay boosted the test’s accuracy to 97%.
The test performed the same whether the tumor was in the head or tail of the pancreas.
“We are very excited about this data,” said Dr. Goel.
The technology was recently licensed to Pharus Diagnostics for commercial development, which will likely include a prospective screening trial, he told this news organization.
Because pancreatic cancer is fairly uncommon, Dr. Goel did not anticipate the test being used for general screening but rather for screening high-risk patients such as those with newly diagnosed type 2 diabetes, a family history of pancreatic cancer, or predisposing genetic mutations.
“It should be a very inexpensive test; it doesn’t cost us much to do in the lab,” he added.
Study moderator Ryan Corcoran, MD, PhD, a gastrointestinal (GI) oncologist at Massachusetts General Hospital, Boston, saw the potential.
“As a GI oncologist, I know how lethal and hard to treat pancreatic cancer is,” he said. A test that could reliably detect pancreatic cancer early, with an acceptable false-positive rate, would be extremely useful.
“The cure rate is many, many times higher,” if we detect it before it has a chance to spread, he explained.
In the meantime, Dr. Goel said there’s more work to be done.
Almost 4,000 subjects have been enrolled in ongoing validation efforts, and efforts are underway to use the test to screen thousands of banked blood samples from the PLCO, a prospective cancer screening trial in healthy subjects.
The researchers also want to see if the test can distinguish benign pancreatic cysts from ones that turn cancerous.
The idea is to find the earliest possible signs of this disease to see if we can find it not “at the moment of clinical diagnosis, but possibly 6 months, 1 year, 2 years earlier” than with radiologic imaging, Dr. Goel said.
The work was funded by the National Cancer Institute and others. Dr. Goel is a consultant for Pharus Diagnostics and Cellomics. Dr. Corcoran is a consultant for, has grants from, and/or holds stock in numerous companies, including Pfizer, Novartis, Eli Lilly, and Revolution Medicines.
A version of this article appeared on Medscape.com.
the most common type of pancreatic cancer.
It is quite encouraging to know we have a blood test that could potentially find this disease early, said Ajay Goel, PhD, a molecular diagnostics specialist at City of Hope in Duarte, California, who presented the findings at the annual meeting of the American Association for Cancer Research (AACR).
Dr. Goel and colleagues developed a signature for pancreatic cancer based on microRNAs identified in the exomes shed from pancreatic cancers and cell-free DNA markers found in the blood of patients with the disease.
Their initial assay tested blood samples for this signature in a training cohort of 252 people in Japan, approximately 60% of whom had pancreatic cancer. The rest were healthy controls. The assay was then tested in validation cohorts of 400 subjects, half with pancreatic cancer and half controls, in China and South Korea.
In both the initial and validation tests, the microRNA assay had an accuracy of about 90% for stage I/II pancreatic cancer, already far better than commercially available assays.
In an additional validation cohort in the United States with 139 patients with pancreatic cancer and 193 controls at six centers across the country, the researchers found that adding carbohydrate antigen 19-9 — a well-known marker of pancreatic cancer — to the assay boosted the test’s accuracy to 97%.
The test performed the same whether the tumor was in the head or tail of the pancreas.
“We are very excited about this data,” said Dr. Goel.
The technology was recently licensed to Pharus Diagnostics for commercial development, which will likely include a prospective screening trial, he told this news organization.
Because pancreatic cancer is fairly uncommon, Dr. Goel did not anticipate the test being used for general screening but rather for screening high-risk patients such as those with newly diagnosed type 2 diabetes, a family history of pancreatic cancer, or predisposing genetic mutations.
“It should be a very inexpensive test; it doesn’t cost us much to do in the lab,” he added.
Study moderator Ryan Corcoran, MD, PhD, a gastrointestinal (GI) oncologist at Massachusetts General Hospital, Boston, saw the potential.
“As a GI oncologist, I know how lethal and hard to treat pancreatic cancer is,” he said. A test that could reliably detect pancreatic cancer early, with an acceptable false-positive rate, would be extremely useful.
“The cure rate is many, many times higher,” if we detect it before it has a chance to spread, he explained.
In the meantime, Dr. Goel said there’s more work to be done.
Almost 4,000 subjects have been enrolled in ongoing validation efforts, and efforts are underway to use the test to screen thousands of banked blood samples from the PLCO, a prospective cancer screening trial in healthy subjects.
The researchers also want to see if the test can distinguish benign pancreatic cysts from ones that turn cancerous.
The idea is to find the earliest possible signs of this disease to see if we can find it not “at the moment of clinical diagnosis, but possibly 6 months, 1 year, 2 years earlier” than with radiologic imaging, Dr. Goel said.
The work was funded by the National Cancer Institute and others. Dr. Goel is a consultant for Pharus Diagnostics and Cellomics. Dr. Corcoran is a consultant for, has grants from, and/or holds stock in numerous companies, including Pfizer, Novartis, Eli Lilly, and Revolution Medicines.
A version of this article appeared on Medscape.com.
FROM AACR 2024
TRAIL-targeting Therapies Still Hold Promise in Cholangiocarcinoma
primarily due to the adaptive resistance and unexpected immune modulation, according to investigators.
Those prior studies evaluated a combination of immunotherapy and TRAIL agonism, but selective TRAIL antagonism shows greater potential via dual ligand/receptor (TRAIL/TRAIL-R) targeting to block immunosuppression, reported lead author Emilien J. Loeuillard, PhD, of Mayo Clinic, Rochester, Minnesota, and colleagues.
“The TRAIL/TRAIL-R system has garnered considerable interest in cancer biology, especially as a potential anticancer therapy,” the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology. “However, TRAIL-R agonists have had very limited anticancer activity in human beings, challenging this concept of TRAIL as an anticancer agent.”
This may be because they were working in the wrong direction, Dr. Loeuillard and colleagues suggested, citing recent work linking TRAIL with tumor proliferation and invasion, possibly via modification of the tumor immune microenvironment.
Exact mechanisms of modification, however, remain unclear. While TRAIL has been associated with tumor-promoting effects like induction of a promyeloid secretome in adenocarcinoma, it has also been linked with anticancer effects like activation of natural killer cells and cytotoxic T lymphocytes.
“Thus, the potency and hierarchy of TRAIL anticancer vs procancer processes in cancer biology has yet to be defined,” the investigators wrote.
While TRAIL ligation of cognate receptors has been previously investigated and shown to trigger proapoptotic signaling pathways, noncanonical TRAIL-mediated signaling remains largely unexplored, particularly in CCA.
The present study evaluated TRAIL biology in CCA using immunocompetent mouse models.
These experiments showed that noncanonical TRAIL signaling immunosuppresses the tumor microenvironment by increasing quantity and activity of myeloid-derived suppressor cells (MDSCs). Blocking noncanonical TRAIL signaling by selective deletion of TRAIL-R in immune cells had significantly reduced tumor volumes alongside fewer MDSCs, driven by FLICE inhibitory protein (cFLIP)-dependent nuclear factor kappa-B activation (NF-kappa-B) in MDSCs, which has antiapoptotic activity. While MDSCs present one possible target in this chain of immunosuppression, “therapeutic strategies for targeting MDSCs are limited,” the investigators wrote, noting that available myeloid modulators have fallen short in clinical trials.
Instead, cFLIP may be a convincing option, they suggested, as targeting cFLIP can sensitize cancer cells to proapoptotic TRAIL signaling. What’s more, cFLIP appears to protect MDSCs from TRAIL-mediated apoptosis, so taking out this barrier could render MDSCs susceptible to therapy.
“Our studies suggest that switching prosurvival/proliferation TRAIL signaling to canonical proapoptotic TRAIL signaling will promote MDSC apoptosis, which in turn has therapeutic implications for CCA suppression,” the investigators wrote.
Hope therefore remains for targeting TRAIL in patients with CCA, but with selective antagonism instead of agonism, as previously attempted.
“In summary, our findings support the role of selective therapeutic targeting of TRAIL-positive cancer cells in an effort to block TRAIL/TRAIL-R–mediated tumor immunosuppression,” Dr. Loeuillard and colleagues concluded.
This study was funded by the Cholangiocarcinoma Foundation and the Mayo Clinic Eagles 5th District Cancer Telethon Funds for Research Fellowship Program, the CTSA/National Center for Advancing Translational Science, the National Institutes of Health/National Cancer Institute, and others. The investigators disclosed no conflicts of interest.
The dismal response of cholangiocarcinoma to immune checkpoint inhibitors (ICI) is particularly concerning, as it impedes the adoption of combination regimens, now standard in most solid tumors. Strategies modulating selective genes involved in the tumor inflammatory environment and tumor cell viability, including those within the tumor necrosis factor superfamily, parallel the mechanism of action of ICI and present a double-edged sword due to the context-dependent pro- and/or anticancer effects of their canonical and/or phantom roles.
Recent investigations suggest that selectively antagonizing TRAIL via (TRAIL/TRAIL-R) targeting may be more effective than agonism. Dr. Ilyas’ group from Mayo Clinic delved into the potential of TRAIL in cancer biology, particularly in CCA, shedding light on the complexities of TRAIL’s role in cancer, where both procancer and anticancer effects are observed.
Further investigation is warranted to explore how TRAIL/TRAIL-R therapy can be effectively combined with other broad-spectrum and/or targeted therapies to maximize selective toxicity to CCA cells, sparing the nonmalignant tissue, thereby extending the lifespan of CCA patients as well as assessing its preventive potential in predisposed premalignant stages, including cholestasis patients.
Sungjin Ko, DVM, PhD, is assistant professor in the Division of Experimental Pathology at the University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania. He is also a member of the Pittsburgh Liver Research Center. He reported no conflicts of interest.
The dismal response of cholangiocarcinoma to immune checkpoint inhibitors (ICI) is particularly concerning, as it impedes the adoption of combination regimens, now standard in most solid tumors. Strategies modulating selective genes involved in the tumor inflammatory environment and tumor cell viability, including those within the tumor necrosis factor superfamily, parallel the mechanism of action of ICI and present a double-edged sword due to the context-dependent pro- and/or anticancer effects of their canonical and/or phantom roles.
Recent investigations suggest that selectively antagonizing TRAIL via (TRAIL/TRAIL-R) targeting may be more effective than agonism. Dr. Ilyas’ group from Mayo Clinic delved into the potential of TRAIL in cancer biology, particularly in CCA, shedding light on the complexities of TRAIL’s role in cancer, where both procancer and anticancer effects are observed.
Further investigation is warranted to explore how TRAIL/TRAIL-R therapy can be effectively combined with other broad-spectrum and/or targeted therapies to maximize selective toxicity to CCA cells, sparing the nonmalignant tissue, thereby extending the lifespan of CCA patients as well as assessing its preventive potential in predisposed premalignant stages, including cholestasis patients.
Sungjin Ko, DVM, PhD, is assistant professor in the Division of Experimental Pathology at the University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania. He is also a member of the Pittsburgh Liver Research Center. He reported no conflicts of interest.
The dismal response of cholangiocarcinoma to immune checkpoint inhibitors (ICI) is particularly concerning, as it impedes the adoption of combination regimens, now standard in most solid tumors. Strategies modulating selective genes involved in the tumor inflammatory environment and tumor cell viability, including those within the tumor necrosis factor superfamily, parallel the mechanism of action of ICI and present a double-edged sword due to the context-dependent pro- and/or anticancer effects of their canonical and/or phantom roles.
Recent investigations suggest that selectively antagonizing TRAIL via (TRAIL/TRAIL-R) targeting may be more effective than agonism. Dr. Ilyas’ group from Mayo Clinic delved into the potential of TRAIL in cancer biology, particularly in CCA, shedding light on the complexities of TRAIL’s role in cancer, where both procancer and anticancer effects are observed.
Further investigation is warranted to explore how TRAIL/TRAIL-R therapy can be effectively combined with other broad-spectrum and/or targeted therapies to maximize selective toxicity to CCA cells, sparing the nonmalignant tissue, thereby extending the lifespan of CCA patients as well as assessing its preventive potential in predisposed premalignant stages, including cholestasis patients.
Sungjin Ko, DVM, PhD, is assistant professor in the Division of Experimental Pathology at the University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania. He is also a member of the Pittsburgh Liver Research Center. He reported no conflicts of interest.
primarily due to the adaptive resistance and unexpected immune modulation, according to investigators.
Those prior studies evaluated a combination of immunotherapy and TRAIL agonism, but selective TRAIL antagonism shows greater potential via dual ligand/receptor (TRAIL/TRAIL-R) targeting to block immunosuppression, reported lead author Emilien J. Loeuillard, PhD, of Mayo Clinic, Rochester, Minnesota, and colleagues.
“The TRAIL/TRAIL-R system has garnered considerable interest in cancer biology, especially as a potential anticancer therapy,” the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology. “However, TRAIL-R agonists have had very limited anticancer activity in human beings, challenging this concept of TRAIL as an anticancer agent.”
This may be because they were working in the wrong direction, Dr. Loeuillard and colleagues suggested, citing recent work linking TRAIL with tumor proliferation and invasion, possibly via modification of the tumor immune microenvironment.
Exact mechanisms of modification, however, remain unclear. While TRAIL has been associated with tumor-promoting effects like induction of a promyeloid secretome in adenocarcinoma, it has also been linked with anticancer effects like activation of natural killer cells and cytotoxic T lymphocytes.
“Thus, the potency and hierarchy of TRAIL anticancer vs procancer processes in cancer biology has yet to be defined,” the investigators wrote.
While TRAIL ligation of cognate receptors has been previously investigated and shown to trigger proapoptotic signaling pathways, noncanonical TRAIL-mediated signaling remains largely unexplored, particularly in CCA.
The present study evaluated TRAIL biology in CCA using immunocompetent mouse models.
These experiments showed that noncanonical TRAIL signaling immunosuppresses the tumor microenvironment by increasing quantity and activity of myeloid-derived suppressor cells (MDSCs). Blocking noncanonical TRAIL signaling by selective deletion of TRAIL-R in immune cells had significantly reduced tumor volumes alongside fewer MDSCs, driven by FLICE inhibitory protein (cFLIP)-dependent nuclear factor kappa-B activation (NF-kappa-B) in MDSCs, which has antiapoptotic activity. While MDSCs present one possible target in this chain of immunosuppression, “therapeutic strategies for targeting MDSCs are limited,” the investigators wrote, noting that available myeloid modulators have fallen short in clinical trials.
Instead, cFLIP may be a convincing option, they suggested, as targeting cFLIP can sensitize cancer cells to proapoptotic TRAIL signaling. What’s more, cFLIP appears to protect MDSCs from TRAIL-mediated apoptosis, so taking out this barrier could render MDSCs susceptible to therapy.
“Our studies suggest that switching prosurvival/proliferation TRAIL signaling to canonical proapoptotic TRAIL signaling will promote MDSC apoptosis, which in turn has therapeutic implications for CCA suppression,” the investigators wrote.
Hope therefore remains for targeting TRAIL in patients with CCA, but with selective antagonism instead of agonism, as previously attempted.
“In summary, our findings support the role of selective therapeutic targeting of TRAIL-positive cancer cells in an effort to block TRAIL/TRAIL-R–mediated tumor immunosuppression,” Dr. Loeuillard and colleagues concluded.
This study was funded by the Cholangiocarcinoma Foundation and the Mayo Clinic Eagles 5th District Cancer Telethon Funds for Research Fellowship Program, the CTSA/National Center for Advancing Translational Science, the National Institutes of Health/National Cancer Institute, and others. The investigators disclosed no conflicts of interest.
primarily due to the adaptive resistance and unexpected immune modulation, according to investigators.
Those prior studies evaluated a combination of immunotherapy and TRAIL agonism, but selective TRAIL antagonism shows greater potential via dual ligand/receptor (TRAIL/TRAIL-R) targeting to block immunosuppression, reported lead author Emilien J. Loeuillard, PhD, of Mayo Clinic, Rochester, Minnesota, and colleagues.
“The TRAIL/TRAIL-R system has garnered considerable interest in cancer biology, especially as a potential anticancer therapy,” the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology. “However, TRAIL-R agonists have had very limited anticancer activity in human beings, challenging this concept of TRAIL as an anticancer agent.”
This may be because they were working in the wrong direction, Dr. Loeuillard and colleagues suggested, citing recent work linking TRAIL with tumor proliferation and invasion, possibly via modification of the tumor immune microenvironment.
Exact mechanisms of modification, however, remain unclear. While TRAIL has been associated with tumor-promoting effects like induction of a promyeloid secretome in adenocarcinoma, it has also been linked with anticancer effects like activation of natural killer cells and cytotoxic T lymphocytes.
“Thus, the potency and hierarchy of TRAIL anticancer vs procancer processes in cancer biology has yet to be defined,” the investigators wrote.
While TRAIL ligation of cognate receptors has been previously investigated and shown to trigger proapoptotic signaling pathways, noncanonical TRAIL-mediated signaling remains largely unexplored, particularly in CCA.
The present study evaluated TRAIL biology in CCA using immunocompetent mouse models.
These experiments showed that noncanonical TRAIL signaling immunosuppresses the tumor microenvironment by increasing quantity and activity of myeloid-derived suppressor cells (MDSCs). Blocking noncanonical TRAIL signaling by selective deletion of TRAIL-R in immune cells had significantly reduced tumor volumes alongside fewer MDSCs, driven by FLICE inhibitory protein (cFLIP)-dependent nuclear factor kappa-B activation (NF-kappa-B) in MDSCs, which has antiapoptotic activity. While MDSCs present one possible target in this chain of immunosuppression, “therapeutic strategies for targeting MDSCs are limited,” the investigators wrote, noting that available myeloid modulators have fallen short in clinical trials.
Instead, cFLIP may be a convincing option, they suggested, as targeting cFLIP can sensitize cancer cells to proapoptotic TRAIL signaling. What’s more, cFLIP appears to protect MDSCs from TRAIL-mediated apoptosis, so taking out this barrier could render MDSCs susceptible to therapy.
“Our studies suggest that switching prosurvival/proliferation TRAIL signaling to canonical proapoptotic TRAIL signaling will promote MDSC apoptosis, which in turn has therapeutic implications for CCA suppression,” the investigators wrote.
Hope therefore remains for targeting TRAIL in patients with CCA, but with selective antagonism instead of agonism, as previously attempted.
“In summary, our findings support the role of selective therapeutic targeting of TRAIL-positive cancer cells in an effort to block TRAIL/TRAIL-R–mediated tumor immunosuppression,” Dr. Loeuillard and colleagues concluded.
This study was funded by the Cholangiocarcinoma Foundation and the Mayo Clinic Eagles 5th District Cancer Telethon Funds for Research Fellowship Program, the CTSA/National Center for Advancing Translational Science, the National Institutes of Health/National Cancer Institute, and others. The investigators disclosed no conflicts of interest.
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY
February 2024 – ICYMI
Gastroenterology
October 2023
El-Salhy M et al. Efficacy of Fecal Microbiota Transplantation for Patients With Irritable Bowel Syndrome at 3 Years After Transplantation. Gastroenterology. 2022 Oct;163(4):982-994.e14. doi: 10.1053/j.gastro.2022.06.020. Epub 2022 Jun 14. PMID: 35709830.
Bajaj JS and Nagy LE. Natural History of Alcohol-Associated Liver Disease: Understanding the Changing Landscape of Pathophysiology and Patient Care. Gastroenterology. 2022 Oct;163(4):840-851. doi: 10.1053/j.gastro.2022.05.031. Epub 2022 May 19. PMID: 35598629; PMCID: PMC9509416.
Lo CH et al. Association of Proton Pump Inhibitor Use With All-Cause and Cause-Specific Mortality. Gastroenterology. 2022 Oct;163(4):852-861.e2. doi: 10.1053/j.gastro.2022.06.067. Epub 2022 Jul 1. PMID: 35788344; PMCID: PMC9509450.
November 2023
Khoshiwal AM et al. The Tissue Systems Pathology Test Outperforms Pathology Review in Risk Stratifying Patients With Low-Grade Dysplasia. Gastroenterology. 2023 Nov;165(5):1168-1179.e6. doi: 10.1053/j.gastro.2023.07.029. Epub 2023 Aug 30. PMID: 37657759.
Chen YI et al. Endoscopic Ultrasound-Guided Biliary Drainage of First Intent With a Lumen-Apposing Metal Stent vs Endoscopic Retrograde Cholangiopancreatography in Malignant Distal Biliary Obstruction: A Multicenter Randomized Controlled Study (ELEMENT Trial). Gastroenterology. 2023 Nov;165(5):1249-1261.e5. doi: 10.1053/j.gastro.2023.07.024. Epub 2023 Aug 6. PMID: 37549753.
December 2023
Almario CV et al. Prevalence and Burden of Illness of Rome IV Irritable Bowel Syndrome in the United States: Results From a Nationwide Cross-Sectional Study. Gastroenterology. 2023 Dec;165(6):1475-1487. doi: 10.1053/j.gastro.2023.08.010. Epub 2023 Aug 16. PMID: 37595647.
Koopmann BDM et al. The Natural Disease Course of Pancreatic Cyst-Associated Neoplasia, Dysplasia, and Ductal Adenocarcinoma: Results of a Microsimulation Model. Gastroenterology. 2023 Dec;165(6):1522-1532. doi: 10.1053/j.gastro.2023.08.027. Epub 2023 Aug 24. PMID: 37633497.
Clinical Gastroenterology and Hepatology
October 2023
Jung DH et al. Comparison of a Polysaccharide Hemostatic Powder and Conventional Therapy for Peptic Ulcer Bleeding. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2844-2253.e5. doi: 10.1016/j.cgh.2023.02.031. Epub 2023 Mar 10. PMID: 36906081.
Liang PS et al. Blood Test Increases Colorectal Cancer Screening in Persons Who Declined Colonoscopy and Fecal Immunochemical Test: A Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2951-2957.e2. doi: 10.1016/j.cgh.2023.03.036. Epub 2023 Apr 8. PMID: 37037262; PMCID: PMC10523873.
November 2023
Li YK et al. Risk of Postcolonoscopy Thromboembolic Events: A Real-World Cohort Study. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3051-3059.e4. doi: 10.1016/j.cgh.2022.09.021. Epub 2022 Sep 24. PMID: 36167228.
Tome J et al. Bile Acid Sequestrants in Microscopic Colitis: Clinical Outcomes and Utility of Bile Acid Testing. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3125-3131.e2. doi: 10.1016/j.cgh.2023.04.031. Epub 2023 May 10. PMID: 37172800.
Berry SK et al. A Randomized Parallel-group Study of Digital Gut-directed Hypnotherapy vs Muscle Relaxation for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3152-3159.e2. doi: 10.1016/j.cgh.2023.06.015. Epub 2023 Jun 28. PMID: 37391055.
December 2023
Kanwal F et al. Risk Stratification Model for Hepatocellular Cancer in Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3296-3304.e3. doi: 10.1016/j.cgh.2023.04.019. Epub 2023 Apr 30. PMID: 37390101; PMCID: PMC10661677.
Forss A et al. Patients With Microscopic Colitis Are at Higher Risk of Major Adverse Cardiovascular Events: A Matched Cohort Study. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3356-3364.e9. doi: 10.1016/j.cgh.2023.05.014. Epub 2023 May 26. PMID: 37245713.
Zheng T et al. A Randomized, Controlled Trial of Efficacy and Safety of Cannabidiol in Idiopathic and Diabetic Gastroparesis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3405-3414.e4. doi: 10.1016/j.cgh.2023.07.008. Epub 2023 Jul 22. PMID: 37482172.
Techniques and Innovations in Gastrointestinal Endoscopy
Rengarajan A and Aadam A. Peroral Endoscopic Myotomy (POEM) and Its Use in Esophageal Dysmotility. Tech Innov Gastrointest Endosc. 2023 Dec 16. doi: 10.1016/j.tige.2023.12.004.
Wang D et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2023 Nov 7. doi: 10.1016/j.tige.2023.10.003
Gastro Hep Advances
Gregory MH et al. Short Bowel Syndrome: Transition of Pediatric Patients to Adult Gastroenterology Care. Gastro Hep Advances. 2023 Sep 8. doi: 10.1016/j.gastha.2023.09.006.
Viser AC et al. Inflammatory Bowel Disease Patients in the Ambulatory Setting Commonly Screen Positive for Malnutrition. Gastro Hep Advances. 2023 Nov 16. doi: 10.1016/j.gastha.2023.11.007.
Gastroenterology
October 2023
El-Salhy M et al. Efficacy of Fecal Microbiota Transplantation for Patients With Irritable Bowel Syndrome at 3 Years After Transplantation. Gastroenterology. 2022 Oct;163(4):982-994.e14. doi: 10.1053/j.gastro.2022.06.020. Epub 2022 Jun 14. PMID: 35709830.
Bajaj JS and Nagy LE. Natural History of Alcohol-Associated Liver Disease: Understanding the Changing Landscape of Pathophysiology and Patient Care. Gastroenterology. 2022 Oct;163(4):840-851. doi: 10.1053/j.gastro.2022.05.031. Epub 2022 May 19. PMID: 35598629; PMCID: PMC9509416.
Lo CH et al. Association of Proton Pump Inhibitor Use With All-Cause and Cause-Specific Mortality. Gastroenterology. 2022 Oct;163(4):852-861.e2. doi: 10.1053/j.gastro.2022.06.067. Epub 2022 Jul 1. PMID: 35788344; PMCID: PMC9509450.
November 2023
Khoshiwal AM et al. The Tissue Systems Pathology Test Outperforms Pathology Review in Risk Stratifying Patients With Low-Grade Dysplasia. Gastroenterology. 2023 Nov;165(5):1168-1179.e6. doi: 10.1053/j.gastro.2023.07.029. Epub 2023 Aug 30. PMID: 37657759.
Chen YI et al. Endoscopic Ultrasound-Guided Biliary Drainage of First Intent With a Lumen-Apposing Metal Stent vs Endoscopic Retrograde Cholangiopancreatography in Malignant Distal Biliary Obstruction: A Multicenter Randomized Controlled Study (ELEMENT Trial). Gastroenterology. 2023 Nov;165(5):1249-1261.e5. doi: 10.1053/j.gastro.2023.07.024. Epub 2023 Aug 6. PMID: 37549753.
December 2023
Almario CV et al. Prevalence and Burden of Illness of Rome IV Irritable Bowel Syndrome in the United States: Results From a Nationwide Cross-Sectional Study. Gastroenterology. 2023 Dec;165(6):1475-1487. doi: 10.1053/j.gastro.2023.08.010. Epub 2023 Aug 16. PMID: 37595647.
Koopmann BDM et al. The Natural Disease Course of Pancreatic Cyst-Associated Neoplasia, Dysplasia, and Ductal Adenocarcinoma: Results of a Microsimulation Model. Gastroenterology. 2023 Dec;165(6):1522-1532. doi: 10.1053/j.gastro.2023.08.027. Epub 2023 Aug 24. PMID: 37633497.
Clinical Gastroenterology and Hepatology
October 2023
Jung DH et al. Comparison of a Polysaccharide Hemostatic Powder and Conventional Therapy for Peptic Ulcer Bleeding. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2844-2253.e5. doi: 10.1016/j.cgh.2023.02.031. Epub 2023 Mar 10. PMID: 36906081.
Liang PS et al. Blood Test Increases Colorectal Cancer Screening in Persons Who Declined Colonoscopy and Fecal Immunochemical Test: A Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2951-2957.e2. doi: 10.1016/j.cgh.2023.03.036. Epub 2023 Apr 8. PMID: 37037262; PMCID: PMC10523873.
November 2023
Li YK et al. Risk of Postcolonoscopy Thromboembolic Events: A Real-World Cohort Study. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3051-3059.e4. doi: 10.1016/j.cgh.2022.09.021. Epub 2022 Sep 24. PMID: 36167228.
Tome J et al. Bile Acid Sequestrants in Microscopic Colitis: Clinical Outcomes and Utility of Bile Acid Testing. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3125-3131.e2. doi: 10.1016/j.cgh.2023.04.031. Epub 2023 May 10. PMID: 37172800.
Berry SK et al. A Randomized Parallel-group Study of Digital Gut-directed Hypnotherapy vs Muscle Relaxation for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3152-3159.e2. doi: 10.1016/j.cgh.2023.06.015. Epub 2023 Jun 28. PMID: 37391055.
December 2023
Kanwal F et al. Risk Stratification Model for Hepatocellular Cancer in Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3296-3304.e3. doi: 10.1016/j.cgh.2023.04.019. Epub 2023 Apr 30. PMID: 37390101; PMCID: PMC10661677.
Forss A et al. Patients With Microscopic Colitis Are at Higher Risk of Major Adverse Cardiovascular Events: A Matched Cohort Study. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3356-3364.e9. doi: 10.1016/j.cgh.2023.05.014. Epub 2023 May 26. PMID: 37245713.
Zheng T et al. A Randomized, Controlled Trial of Efficacy and Safety of Cannabidiol in Idiopathic and Diabetic Gastroparesis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3405-3414.e4. doi: 10.1016/j.cgh.2023.07.008. Epub 2023 Jul 22. PMID: 37482172.
Techniques and Innovations in Gastrointestinal Endoscopy
Rengarajan A and Aadam A. Peroral Endoscopic Myotomy (POEM) and Its Use in Esophageal Dysmotility. Tech Innov Gastrointest Endosc. 2023 Dec 16. doi: 10.1016/j.tige.2023.12.004.
Wang D et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2023 Nov 7. doi: 10.1016/j.tige.2023.10.003
Gastro Hep Advances
Gregory MH et al. Short Bowel Syndrome: Transition of Pediatric Patients to Adult Gastroenterology Care. Gastro Hep Advances. 2023 Sep 8. doi: 10.1016/j.gastha.2023.09.006.
Viser AC et al. Inflammatory Bowel Disease Patients in the Ambulatory Setting Commonly Screen Positive for Malnutrition. Gastro Hep Advances. 2023 Nov 16. doi: 10.1016/j.gastha.2023.11.007.
Gastroenterology
October 2023
El-Salhy M et al. Efficacy of Fecal Microbiota Transplantation for Patients With Irritable Bowel Syndrome at 3 Years After Transplantation. Gastroenterology. 2022 Oct;163(4):982-994.e14. doi: 10.1053/j.gastro.2022.06.020. Epub 2022 Jun 14. PMID: 35709830.
Bajaj JS and Nagy LE. Natural History of Alcohol-Associated Liver Disease: Understanding the Changing Landscape of Pathophysiology and Patient Care. Gastroenterology. 2022 Oct;163(4):840-851. doi: 10.1053/j.gastro.2022.05.031. Epub 2022 May 19. PMID: 35598629; PMCID: PMC9509416.
Lo CH et al. Association of Proton Pump Inhibitor Use With All-Cause and Cause-Specific Mortality. Gastroenterology. 2022 Oct;163(4):852-861.e2. doi: 10.1053/j.gastro.2022.06.067. Epub 2022 Jul 1. PMID: 35788344; PMCID: PMC9509450.
November 2023
Khoshiwal AM et al. The Tissue Systems Pathology Test Outperforms Pathology Review in Risk Stratifying Patients With Low-Grade Dysplasia. Gastroenterology. 2023 Nov;165(5):1168-1179.e6. doi: 10.1053/j.gastro.2023.07.029. Epub 2023 Aug 30. PMID: 37657759.
Chen YI et al. Endoscopic Ultrasound-Guided Biliary Drainage of First Intent With a Lumen-Apposing Metal Stent vs Endoscopic Retrograde Cholangiopancreatography in Malignant Distal Biliary Obstruction: A Multicenter Randomized Controlled Study (ELEMENT Trial). Gastroenterology. 2023 Nov;165(5):1249-1261.e5. doi: 10.1053/j.gastro.2023.07.024. Epub 2023 Aug 6. PMID: 37549753.
December 2023
Almario CV et al. Prevalence and Burden of Illness of Rome IV Irritable Bowel Syndrome in the United States: Results From a Nationwide Cross-Sectional Study. Gastroenterology. 2023 Dec;165(6):1475-1487. doi: 10.1053/j.gastro.2023.08.010. Epub 2023 Aug 16. PMID: 37595647.
Koopmann BDM et al. The Natural Disease Course of Pancreatic Cyst-Associated Neoplasia, Dysplasia, and Ductal Adenocarcinoma: Results of a Microsimulation Model. Gastroenterology. 2023 Dec;165(6):1522-1532. doi: 10.1053/j.gastro.2023.08.027. Epub 2023 Aug 24. PMID: 37633497.
Clinical Gastroenterology and Hepatology
October 2023
Jung DH et al. Comparison of a Polysaccharide Hemostatic Powder and Conventional Therapy for Peptic Ulcer Bleeding. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2844-2253.e5. doi: 10.1016/j.cgh.2023.02.031. Epub 2023 Mar 10. PMID: 36906081.
Liang PS et al. Blood Test Increases Colorectal Cancer Screening in Persons Who Declined Colonoscopy and Fecal Immunochemical Test: A Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2023 Oct;21(11):2951-2957.e2. doi: 10.1016/j.cgh.2023.03.036. Epub 2023 Apr 8. PMID: 37037262; PMCID: PMC10523873.
November 2023
Li YK et al. Risk of Postcolonoscopy Thromboembolic Events: A Real-World Cohort Study. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3051-3059.e4. doi: 10.1016/j.cgh.2022.09.021. Epub 2022 Sep 24. PMID: 36167228.
Tome J et al. Bile Acid Sequestrants in Microscopic Colitis: Clinical Outcomes and Utility of Bile Acid Testing. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3125-3131.e2. doi: 10.1016/j.cgh.2023.04.031. Epub 2023 May 10. PMID: 37172800.
Berry SK et al. A Randomized Parallel-group Study of Digital Gut-directed Hypnotherapy vs Muscle Relaxation for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2023 Nov;21(12):3152-3159.e2. doi: 10.1016/j.cgh.2023.06.015. Epub 2023 Jun 28. PMID: 37391055.
December 2023
Kanwal F et al. Risk Stratification Model for Hepatocellular Cancer in Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3296-3304.e3. doi: 10.1016/j.cgh.2023.04.019. Epub 2023 Apr 30. PMID: 37390101; PMCID: PMC10661677.
Forss A et al. Patients With Microscopic Colitis Are at Higher Risk of Major Adverse Cardiovascular Events: A Matched Cohort Study. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3356-3364.e9. doi: 10.1016/j.cgh.2023.05.014. Epub 2023 May 26. PMID: 37245713.
Zheng T et al. A Randomized, Controlled Trial of Efficacy and Safety of Cannabidiol in Idiopathic and Diabetic Gastroparesis. Clin Gastroenterol Hepatol. 2023 Dec;21(13):3405-3414.e4. doi: 10.1016/j.cgh.2023.07.008. Epub 2023 Jul 22. PMID: 37482172.
Techniques and Innovations in Gastrointestinal Endoscopy
Rengarajan A and Aadam A. Peroral Endoscopic Myotomy (POEM) and Its Use in Esophageal Dysmotility. Tech Innov Gastrointest Endosc. 2023 Dec 16. doi: 10.1016/j.tige.2023.12.004.
Wang D et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2023 Nov 7. doi: 10.1016/j.tige.2023.10.003
Gastro Hep Advances
Gregory MH et al. Short Bowel Syndrome: Transition of Pediatric Patients to Adult Gastroenterology Care. Gastro Hep Advances. 2023 Sep 8. doi: 10.1016/j.gastha.2023.09.006.
Viser AC et al. Inflammatory Bowel Disease Patients in the Ambulatory Setting Commonly Screen Positive for Malnutrition. Gastro Hep Advances. 2023 Nov 16. doi: 10.1016/j.gastha.2023.11.007.
What's your diagnosis?
The diagnosis
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.
References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
The diagnosis
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.

References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
The diagnosis
Based on the clinical and imaging findings, a diagnosis of gallbladder adenomyomatosis was made. GA is a benign and usually asymptomatic condition that occurs mainly beyond the age of 50-60 years and is very rare in childhood.1 Symptomatic gallbladder adenomyomatosis indicates cholecystectomy, considering the presence of inflammation or gallbladder stones.2 Therefore, a laparoscopic cholecystectomy was performed on our patient. Rokitansky-Aschoff sinuses were seen in the entire thickened gallbladder wall on gross pathologic examination (Figure D). Histopathologic examination confirmed the diagnosis of GA with cholecystitis. The patient was eventually diagnosed with diffuse GA. She was successfully discharged from the hospital 4 days after surgery, and 3 months of follow-up were uneventful.

References
Eroglu N et al. Diffuse adenomyomatosis of the gallbladder in a child. J Pediatr Hematol Oncol. 2016;38:e307-9.
Bonatti M. et al. Gallbladder adenomyomatosis: imaging findings, tricks and pitfalls. Insights Imaging. 2017;8:243-53.
Hammad AY et al. A literature review of radiological findings to guide the diagnosis of gallbladder adenomyomatosis. HPB (Oxford). 2016;18:129-35.
A 15-year-old girl presented with an 18-month history of intermittent right upper quadrant pain that appeared after meals and was relieved after rest. She denied any nausea, vomiting, chills, diarrhea, or constipation. The patient reported no trauma. At admission, physical examination showed tenderness in the right upper abdomen without rebound or guarding. Murphy's sign was also present. The laboratory tests were unremarkable.
Ultrasound examination indicated gallbladder wall thickening. Furthermore, a contrast-enhanced computed tomographic scan showed marked gallbladder wall thickening with an annular unenhanced proliferative muscularis layer surrounding enhanced proliferative mucosal epithelium (Figure A), and magnetic resonance imaging showed multiple cyst-like spaces in the gallbladder wall (Figures B and C).
What is the diagnosis, and how should it be managed?
Previously published in Gastroenterology