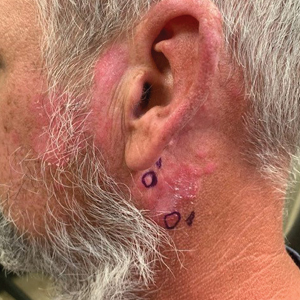User login
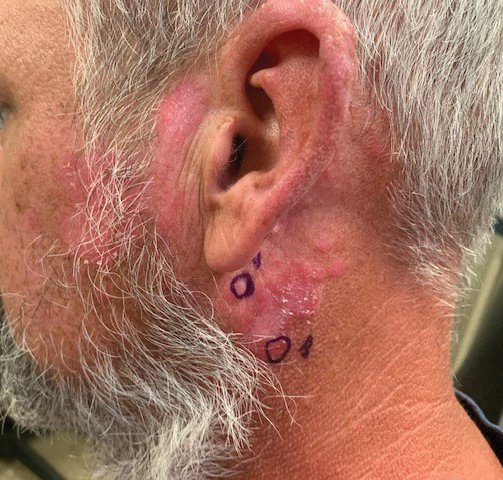
Chronic Erythematous Plaques Around the Ears
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
The Diagnosis: Discoid Lupus Erythematosus
The biopsies demonstrated vacuolar interface changes with superficial and deep perivascular and periadnexal inflammation as well as increased background mucin deposition. The clinical morphology and distributions of the plaques limited to the photoexposed areas of the head suggested a diagnosis of discoid lupus erythematosus (DLE). The interface changes on histopathology supported this clinical impression. Our patient was treated with limited application of triamcinolone ointment 0.1% twice daily around the ears and neck, tacrolimus ointment 0.1% twice daily on the face, and hydroxychloroquine, as well as sun protection instructions. Smoking cessation was strongly advised.
Discoid lupus erythematosus is a disorder with chronic, erythematous, scaly, coin-shaped (discoid) plaques and is the most common form of chronic cutaneous lupus erythematosus.1 Lesions usually present on sun-exposed areas of the face, scalp, neck, ears, lips, or upper torso. They expand slowly with an active peripheral margin and a central scar that can result in induration, pigmentation changes, telangiectases, pruritus, or tenderness. Hair-bearing areas may be involved, causing hair loss due to follicular plugging; irreversible scarring alopecia can result. Facial DLE often spares the nasolabial folds. Ear involvement characteristically includes the conchal bowl and the outer external auditory canal. Discoid lupus erythematosus is considered localized if most of the head and neck region is involved or generalized if lesions also are present below the neck. Risk factors for DLE include genetic and environmental factors such as UV exposure, hormones, or exposure to toxins such as cigarette smoke.1 The disorder most commonly affects females and has a higher prevalence in patients of African descent than in Asian and White patients. Disease can occur at any age but usually occurs between 20 and 40 years of age.2 Discoid lupus erythematosus and other forms of chronic cutaneous lupus can occur independently or in conjunction with systemic lupus erythematosus (SLE), and approximately 15% to 30% of SLE patients develop DLE.1
Discoid lupus erythematosus is clinically diagnosed by the presentation of plaques in the characteristic distribution with confirmation via skin biopsy.1 Elman et al3 created a system for DLE classification that was only clinical and did not involve histopathology. Histologically, DLE often includes basement membrane thickening, follicular keratin plugs, mucin deposition, and vacuolar change with an interface, and a perivascular and periadnexal lymphocytic infiltrate.3,4 Antibodies such as antinuclear antibodies, rheumatoid factor, anti–double-stranded DNA, anti-Smith, and Sjögren syndrome A and B antibodies may be present (albeit with low positive frequency) in cutaneous lupus erythematosus.4 Characteristics of SLE also may be present, helping to confirm the diagnosis. Because there is an association of DLE with SLE, various laboratory tests should be ordered, including complete blood cell count, renal function panel, inflammatory markers, antibodies, and urinalysis for proteinuria.2,4
Treatment of DLE consists of preventative measures, such as sun protection with vitamin D supplementation, avoidance of drug triggers, and smoking cessation, as well as pharmacotherapy. The importance of wearing sun protective hats and garments with sunscreen use cannot be understated.1 Smoking cessation should be advised because smoking reduces the efficacy of antimalarial treatment and potentially increases the likelihood of patients requiring a second antimalarial drug. Quinacrine often is noted in both the dermatology and rheumatology literature to be used for escalating cutaneous lupus erythematosus care when hydroxychloroquine is ineffective or not tolerated, but no US manufacturer produces this medication; thus, compounding is required, which may be financially prohibitive, making this recommendation difficult to translate into clinical practice.5 Firstline therapy for acute flares is high-potency topical corticosteroids. If lesions are primarily on areas other than the face, a medium-potency topical steroid may be used. Topical calcineurin inhibitors or intralesional corticosteroids may be used if minimal improvement is seen after initial topical corticosteroid therapy. Treatment for widespread disease or disease that is resistant to local treatment is systemic therapy with antimalarial agents, followed by antimetabolites, systemic retinoids, thalidomide, or dapsone.1,2 The Cutaneous Lupus Erythematosus Disease Area and Severity Index is a valid tool to gauge the degree of disease and to help with disease progression and treatment response by noting the features of the plaques.1
Patients also should be educated that this disease can last for years, and long-standing DLE plaques infrequently can give rise to squamous cell carcinoma. In addition, isolated DLE can progress to SLE in 5% to 28% of patients.2
The differential diagnosis in our patient included other diseases with violaceous annular lesions and central clearing. Majocchi granuloma usually presents in areas of prior trauma, possibly due to shaving the face in our patient, or in the setting of topical corticosteroid use or immunosuppression. Scaling often is present within lesions, and histology shows fungal elements.6 Cutaneous sarcoidosis usually presents on the face, with scarring alopecia when appearing on the scalp; histology shows noncaseating granulomas, and 70% of patients with cutaneous symptoms will have systemic sarcoidosis.7 Granuloma annulare most commonly presents on the extremities, and histology shows lymphohistiocytic granulomas in a palisaded or interstitial pattern with connective-tissue degeneration and mucinous deposits.8 Annular psoriasis often is scaly and symmetric with parakeratosis, epidermal hyperplasia, dilated dermal capillaries, loss of granular layer, perivascular mononuclear cell infiltrate, and elongation of rete ridges on histology.9 Drug-induced lupus erythematosus always should be considered in patients taking triggering drugs such as antihypertensives, lipid-lowering drugs, antifungals, anti–tumor necrosis factor drugs, and proton pump inhibitors—the latter being a drug our patient was taking.10
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
- Sontheimer CJ, Costner MI, Sontheimer RD. Lupus erythematosus. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:1037-1060.
- Lee KC. Discoid lupus. In: Ferri FF, ed. Ferri’s Clinical Advisor 2021. Elsevier; 2021:477.e15-477.e18.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156:901-906. doi:10.1001/jamadermatol.2020.1698
- Patel P, Werth V. Cutaneous lupus erythematosus: a review. Dermatol Clin. 2002;20:373-385, v. doi:10.1016/s0733-8635(02)00016-5
- Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77:374-377. doi:10.1016/j .jaad.2017.03.027
- Craddock LN, Schieke SM. Superficial fungal infection. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:2925-2951.
- Tan J, Vleugels R. Dermatologic findings in systemic disease. In: McKean S, Dressler D, Ross J, et al, eds. Principles and Practice of Hospital Medicine. 2nd ed. McGraw Hill; 2017:1145-1170.
- Prendiville JS. Granuloma annulare. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:564-571.
- Gudjonsson JE, Elder JT. Psoriasis. In: Kang S, Amagai M, Bruckner A, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019:457-497.
- He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30:490-497. doi:10.1097/BOR.0000000000000522
A 41-year-old man presented to the dermatology clinic with erythematous, pruritic, and painful plaques around the ears of 6 years’ duration. He reported that application of topical steroids, antifungals, and most recently a topical calcineurin inhibitor did not change the appearance or symptoms. His medical history was notable for tobacco smoking and gastroesophageal reflux disease, for which he was taking omeprazole for the last 3 years. He was unsure if the lesions changed with UV exposure. He was an active-duty US military service member, and his job required frequently working outdoors. A review of systems was otherwise unremarkable. Physical examination revealed annular, erythematous, indurated plaques on both the preauricular and postauricular skin on the left ear with associated central atrophy and hypopigmentation. No alopecia was appreciated. The remainder of the skin examination was unremarkable. Ancillary laboratory test results were notable for a negative antinuclear antibody screen but positive (low titer) for Sjögren syndrome A and B antibodies. Two punch biopsies were performed.
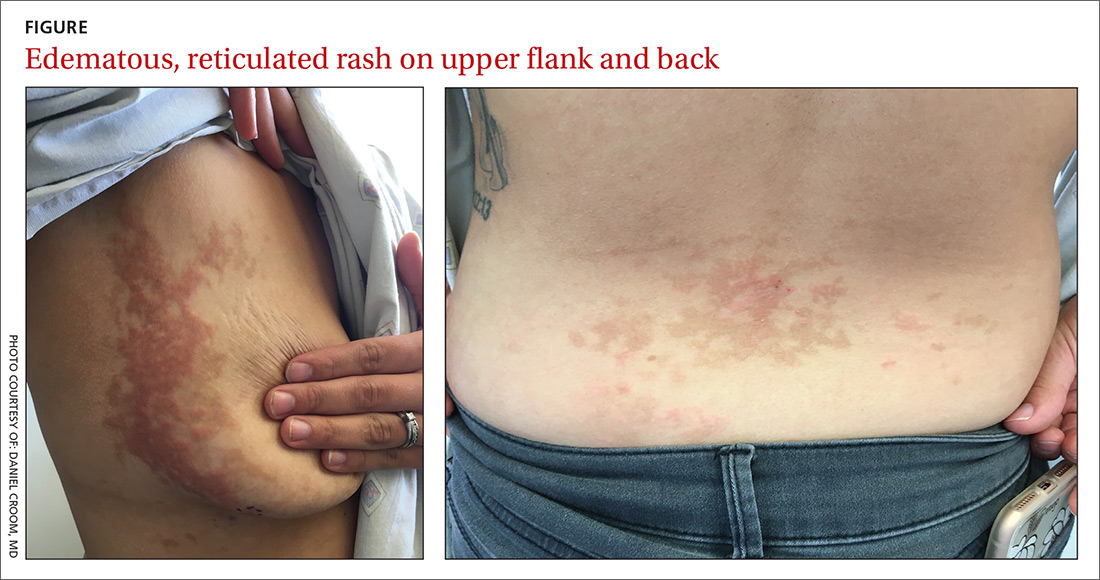
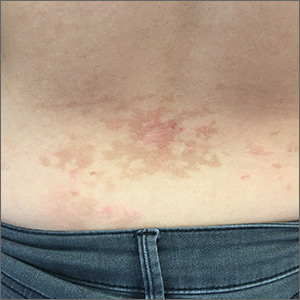
Pruritic rash on chest and back
A 26-year-old woman presented to our clinic with pruritic, hyperpigmented, symmetric edematous plaques on her upper flank, chest, and lower back (FIGURE) 3 weeks after starting a strict ketogenic (high fat/low carbohydrate) diet for postpartum weight loss. The patient was an otherwise healthy stay-at-home mother with an unremarkable medical history.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Prurigo pigmentosa
We recognized that this was a case of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30.1
While the pathophysiology remains unknown, the rash most often is reported in association with ketogenic diets.1 Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
Although prurigo pigmentosa is relatively uncommon (with an unknown incidence), primary care physicians may begin to encounter the characteristic rash more frequently, given the number of articles over the past 5 years in the primary care and nutritional literature highlighting the diet’s health benefits.2 The ketogenic diet is a high-fat, low-carbohydrate diet with preliminary evidence of improved weight loss, cardiovascular health, and glycemic control suggested by a meta-analysis of 13 randomized controlled trials.3 Additionally, the popular press and general public’s rising interest are likely to increase the number of patients on this diet.
A clinical diagnosis
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal. Urinary ketones are present in only 30% to 50% of patients; there is, however, an absence of blood ketones.
Continue to: Other conditions can mimic prurigo pigmentosa
Other conditions can mimic prurigo pigmentosa
Urticaria presents as individual lesions that often have a pale center. The lesions may occur anywhere on the body and generally last less than 24 hours. History may reveal a trigger including drugs, infection, food, or emotional stress in up to 50% of cases.
Irritant contact dermatitis often is associated with a stinging or burning sensation. Irritant and allergic contact dermatitis may have a geometric, or “outside job,” distribution suggestive of external contact, potentially with plants, alkalis, acids, or solvents.5
Confluent and reticulated papillomatosis is a rare asymptomatic dermatosis of unknown etiology that presents as hyperpigmented papules on the upper trunk, neck, and axillae. Most patients lack associated pruritis which is in contrast to prurigo pigmentosa.6
Pityriasis rosea is a viral exanthem that may be associated with constitutional symptoms and often presents initially with a herald patch progressing to a classic “Christmas tree” distribution with a fine collarette of scale. It often is asymptomatic, although some cases may be pruritic.5
Treatment focuses on dietary modification
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade.
Continue to: Pharmaceutical intervention may be necessary
Pharmaceutical intervention may be necessary
If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties.1,4 Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.1,4
Our patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
CORRESPONDENCE
Daniel Croom, MD, 34520 Bob Wilson Dr, Naval Medical Center San Diego, San Diego, CA 92134; daniel.l.croom.mil@mail.mil
1. Kim JK, Chung WK, Chang SE, et al. Prurigo pigmentosa: clinicopathological study and analysis of 50 cases in Korea. J Dermatol. 2012;39:891-897.
2. Abbasi J. Interest in the ketogenic diet grows for weight loss and type 2 diabetes. JAMA. 2018;319:215-217.
3. Bueno NB, de Melo IS, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187.
4. Oh YJ, Lee MH. Prurigo pigmentosa: a clinicopathologic study of 16 cases. J Eur Acad Dermatol Venereol. 2012;26:1149-1153.
5. James WD, Berger T, Elston D. Diseases of the skin appendages. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia, PA: Saunders Elsevier; 2015:747-788.
6. Shevchenko A, Valdes-Rodriguez R, Hsu S, et al. Prurigo pigmentosa: case series and differentiation from confluent and reticulated papillomatosis. JAAD Case Rep. 2018;4:77-80.
A 26-year-old woman presented to our clinic with pruritic, hyperpigmented, symmetric edematous plaques on her upper flank, chest, and lower back (FIGURE) 3 weeks after starting a strict ketogenic (high fat/low carbohydrate) diet for postpartum weight loss. The patient was an otherwise healthy stay-at-home mother with an unremarkable medical history.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Prurigo pigmentosa
We recognized that this was a case of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30.1
While the pathophysiology remains unknown, the rash most often is reported in association with ketogenic diets.1 Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
Although prurigo pigmentosa is relatively uncommon (with an unknown incidence), primary care physicians may begin to encounter the characteristic rash more frequently, given the number of articles over the past 5 years in the primary care and nutritional literature highlighting the diet’s health benefits.2 The ketogenic diet is a high-fat, low-carbohydrate diet with preliminary evidence of improved weight loss, cardiovascular health, and glycemic control suggested by a meta-analysis of 13 randomized controlled trials.3 Additionally, the popular press and general public’s rising interest are likely to increase the number of patients on this diet.
A clinical diagnosis
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal. Urinary ketones are present in only 30% to 50% of patients; there is, however, an absence of blood ketones.
Continue to: Other conditions can mimic prurigo pigmentosa
Other conditions can mimic prurigo pigmentosa
Urticaria presents as individual lesions that often have a pale center. The lesions may occur anywhere on the body and generally last less than 24 hours. History may reveal a trigger including drugs, infection, food, or emotional stress in up to 50% of cases.
Irritant contact dermatitis often is associated with a stinging or burning sensation. Irritant and allergic contact dermatitis may have a geometric, or “outside job,” distribution suggestive of external contact, potentially with plants, alkalis, acids, or solvents.5
Confluent and reticulated papillomatosis is a rare asymptomatic dermatosis of unknown etiology that presents as hyperpigmented papules on the upper trunk, neck, and axillae. Most patients lack associated pruritis which is in contrast to prurigo pigmentosa.6
Pityriasis rosea is a viral exanthem that may be associated with constitutional symptoms and often presents initially with a herald patch progressing to a classic “Christmas tree” distribution with a fine collarette of scale. It often is asymptomatic, although some cases may be pruritic.5
Treatment focuses on dietary modification
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade.
Continue to: Pharmaceutical intervention may be necessary
Pharmaceutical intervention may be necessary
If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties.1,4 Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.1,4
Our patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
CORRESPONDENCE
Daniel Croom, MD, 34520 Bob Wilson Dr, Naval Medical Center San Diego, San Diego, CA 92134; daniel.l.croom.mil@mail.mil
A 26-year-old woman presented to our clinic with pruritic, hyperpigmented, symmetric edematous plaques on her upper flank, chest, and lower back (FIGURE) 3 weeks after starting a strict ketogenic (high fat/low carbohydrate) diet for postpartum weight loss. The patient was an otherwise healthy stay-at-home mother with an unremarkable medical history.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Prurigo pigmentosa
We recognized that this was a case of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30.1
While the pathophysiology remains unknown, the rash most often is reported in association with ketogenic diets.1 Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
Although prurigo pigmentosa is relatively uncommon (with an unknown incidence), primary care physicians may begin to encounter the characteristic rash more frequently, given the number of articles over the past 5 years in the primary care and nutritional literature highlighting the diet’s health benefits.2 The ketogenic diet is a high-fat, low-carbohydrate diet with preliminary evidence of improved weight loss, cardiovascular health, and glycemic control suggested by a meta-analysis of 13 randomized controlled trials.3 Additionally, the popular press and general public’s rising interest are likely to increase the number of patients on this diet.
A clinical diagnosis
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal. Urinary ketones are present in only 30% to 50% of patients; there is, however, an absence of blood ketones.
Continue to: Other conditions can mimic prurigo pigmentosa
Other conditions can mimic prurigo pigmentosa
Urticaria presents as individual lesions that often have a pale center. The lesions may occur anywhere on the body and generally last less than 24 hours. History may reveal a trigger including drugs, infection, food, or emotional stress in up to 50% of cases.
Irritant contact dermatitis often is associated with a stinging or burning sensation. Irritant and allergic contact dermatitis may have a geometric, or “outside job,” distribution suggestive of external contact, potentially with plants, alkalis, acids, or solvents.5
Confluent and reticulated papillomatosis is a rare asymptomatic dermatosis of unknown etiology that presents as hyperpigmented papules on the upper trunk, neck, and axillae. Most patients lack associated pruritis which is in contrast to prurigo pigmentosa.6
Pityriasis rosea is a viral exanthem that may be associated with constitutional symptoms and often presents initially with a herald patch progressing to a classic “Christmas tree” distribution with a fine collarette of scale. It often is asymptomatic, although some cases may be pruritic.5
Treatment focuses on dietary modification
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade.
Continue to: Pharmaceutical intervention may be necessary
Pharmaceutical intervention may be necessary
If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties.1,4 Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.1,4
Our patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
CORRESPONDENCE
Daniel Croom, MD, 34520 Bob Wilson Dr, Naval Medical Center San Diego, San Diego, CA 92134; daniel.l.croom.mil@mail.mil
1. Kim JK, Chung WK, Chang SE, et al. Prurigo pigmentosa: clinicopathological study and analysis of 50 cases in Korea. J Dermatol. 2012;39:891-897.
2. Abbasi J. Interest in the ketogenic diet grows for weight loss and type 2 diabetes. JAMA. 2018;319:215-217.
3. Bueno NB, de Melo IS, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187.
4. Oh YJ, Lee MH. Prurigo pigmentosa: a clinicopathologic study of 16 cases. J Eur Acad Dermatol Venereol. 2012;26:1149-1153.
5. James WD, Berger T, Elston D. Diseases of the skin appendages. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia, PA: Saunders Elsevier; 2015:747-788.
6. Shevchenko A, Valdes-Rodriguez R, Hsu S, et al. Prurigo pigmentosa: case series and differentiation from confluent and reticulated papillomatosis. JAAD Case Rep. 2018;4:77-80.
1. Kim JK, Chung WK, Chang SE, et al. Prurigo pigmentosa: clinicopathological study and analysis of 50 cases in Korea. J Dermatol. 2012;39:891-897.
2. Abbasi J. Interest in the ketogenic diet grows for weight loss and type 2 diabetes. JAMA. 2018;319:215-217.
3. Bueno NB, de Melo IS, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187.
4. Oh YJ, Lee MH. Prurigo pigmentosa: a clinicopathologic study of 16 cases. J Eur Acad Dermatol Venereol. 2012;26:1149-1153.
5. James WD, Berger T, Elston D. Diseases of the skin appendages. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia, PA: Saunders Elsevier; 2015:747-788.
6. Shevchenko A, Valdes-Rodriguez R, Hsu S, et al. Prurigo pigmentosa: case series and differentiation from confluent and reticulated papillomatosis. JAAD Case Rep. 2018;4:77-80.