User login
Health Canada approves two tests for CML patients
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Markers associated with efficacy of malaria vaccine
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
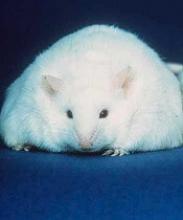
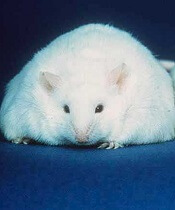
Diet change can improve survival in obese mice with ALL
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
FDA clears instrument for blood typing, screening
The U.S. Food and Drug Administration has granted marketing clearance for the immunohematology instrument NEO Iris™.
NEO Iris is a fully automated blood bank instrument designed for the mid- to high-volume laboratory, according to Immucor, Inc., the company marketing the device.
Immucor says NEO Iris provides the highest type and screen throughput on the market—up to 60 types and screens per hour.
NEO Iris performs ABO/Rh D typing, weak D testing, donor confirmation, cytomegalovirus screening, immunoglobulin G direct antiglobulin testing and crossmatching, and antibody identification and screening.
The workflow management tool on Neo Iris has STAT priority and allows operators to run tests in any order at any time, according to Immucor.
The company says NEO Iris can hold up to 224 samples, and “modules can pipette, incubate, centrifuge, and read simultaneously.”
NEO Iris integrates with Immucor’s data management software, ImmuLINK®, to aggregate test results and produce reports with complete testing history.
Another feature of NEO Iris is blud_directSM, which provides technical support from an Immucor representative that can be accessed on the NEO Iris monitor.
For more information on the instrument, see the NEO Iris page on the Immucor website.
The U.S. Food and Drug Administration has granted marketing clearance for the immunohematology instrument NEO Iris™.
NEO Iris is a fully automated blood bank instrument designed for the mid- to high-volume laboratory, according to Immucor, Inc., the company marketing the device.
Immucor says NEO Iris provides the highest type and screen throughput on the market—up to 60 types and screens per hour.
NEO Iris performs ABO/Rh D typing, weak D testing, donor confirmation, cytomegalovirus screening, immunoglobulin G direct antiglobulin testing and crossmatching, and antibody identification and screening.
The workflow management tool on Neo Iris has STAT priority and allows operators to run tests in any order at any time, according to Immucor.
The company says NEO Iris can hold up to 224 samples, and “modules can pipette, incubate, centrifuge, and read simultaneously.”
NEO Iris integrates with Immucor’s data management software, ImmuLINK®, to aggregate test results and produce reports with complete testing history.
Another feature of NEO Iris is blud_directSM, which provides technical support from an Immucor representative that can be accessed on the NEO Iris monitor.
For more information on the instrument, see the NEO Iris page on the Immucor website.
The U.S. Food and Drug Administration has granted marketing clearance for the immunohematology instrument NEO Iris™.
NEO Iris is a fully automated blood bank instrument designed for the mid- to high-volume laboratory, according to Immucor, Inc., the company marketing the device.
Immucor says NEO Iris provides the highest type and screen throughput on the market—up to 60 types and screens per hour.
NEO Iris performs ABO/Rh D typing, weak D testing, donor confirmation, cytomegalovirus screening, immunoglobulin G direct antiglobulin testing and crossmatching, and antibody identification and screening.
The workflow management tool on Neo Iris has STAT priority and allows operators to run tests in any order at any time, according to Immucor.
The company says NEO Iris can hold up to 224 samples, and “modules can pipette, incubate, centrifuge, and read simultaneously.”
NEO Iris integrates with Immucor’s data management software, ImmuLINK®, to aggregate test results and produce reports with complete testing history.
Another feature of NEO Iris is blud_directSM, which provides technical support from an Immucor representative that can be accessed on the NEO Iris monitor.
For more information on the instrument, see the NEO Iris page on the Immucor website.
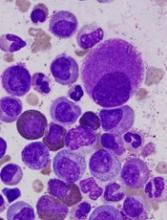
‘Compelling’ new target found for monocytic AML
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
Triplet improves PFS in relapsed/refractory MM
Adding elotuzumab to pomalidomide and low-dose dexamethasone can improve progression-free survival (PFS) in patients with relapsed/refractory multiple myeloma (MM), according to the ELOQUENT-3 trial.
These results support the recent U.S. approval of elotuzumab, pomalidomide, and dexamethasone in adults with MM who have received at least two prior therapies, including lenalidomide and a proteasome inhibitor.
Results from ELOQUENT-3 were recently published in The New England Journal of Medicine and previously presented at the 23rd Congress of the European Hematology Association in June.
The phase 2 trial included patients with refractory or relapsed and refractory MM who had received lenalidomide and a proteasome inhibitor.
The patients were randomized to receive elotuzumab plus pomalidomide and low-dose dexamethasone (EPd, n=60) or pomalidomide and low-dose dexamethasone (Pd, n=57) in 28-day cycles until disease progression or unacceptable toxicity.
The overall response rate was 53% in the EPd arm and 26% in the Pd arm (odds ratio=3.25; P=0.0029).
There were two stringent complete responses (CRs) and three CRs in the EPd arm, and there was one CR in the Pd arm.
The median duration of response was 8.3 months in the Pd arm and was not reached in the EPd arm.
The median PFS was 10.3 months with EPd and 4.7 months with Pd (hazard ratio=0.54, P=0.008).
Overall survival data were not mature at the time of analysis, but there was a trend favoring EPd over Pd (hazard ratio=0.62). There were 13 deaths in the EPd arm and 18 in the Pd arm.
The most common treatment-related adverse events (in the EPd and Pd arms, respectively) were neutropenia (18% and 20%), hyperglycemia (18% and 11%), and anemia (10% and 15%).
This trial was supported by Bristol-Myers Squibb and AbbVie Biotherapeutics, and researchers reported relationships with several other companies.
Adding elotuzumab to pomalidomide and low-dose dexamethasone can improve progression-free survival (PFS) in patients with relapsed/refractory multiple myeloma (MM), according to the ELOQUENT-3 trial.
These results support the recent U.S. approval of elotuzumab, pomalidomide, and dexamethasone in adults with MM who have received at least two prior therapies, including lenalidomide and a proteasome inhibitor.
Results from ELOQUENT-3 were recently published in The New England Journal of Medicine and previously presented at the 23rd Congress of the European Hematology Association in June.
The phase 2 trial included patients with refractory or relapsed and refractory MM who had received lenalidomide and a proteasome inhibitor.
The patients were randomized to receive elotuzumab plus pomalidomide and low-dose dexamethasone (EPd, n=60) or pomalidomide and low-dose dexamethasone (Pd, n=57) in 28-day cycles until disease progression or unacceptable toxicity.
The overall response rate was 53% in the EPd arm and 26% in the Pd arm (odds ratio=3.25; P=0.0029).
There were two stringent complete responses (CRs) and three CRs in the EPd arm, and there was one CR in the Pd arm.
The median duration of response was 8.3 months in the Pd arm and was not reached in the EPd arm.
The median PFS was 10.3 months with EPd and 4.7 months with Pd (hazard ratio=0.54, P=0.008).
Overall survival data were not mature at the time of analysis, but there was a trend favoring EPd over Pd (hazard ratio=0.62). There were 13 deaths in the EPd arm and 18 in the Pd arm.
The most common treatment-related adverse events (in the EPd and Pd arms, respectively) were neutropenia (18% and 20%), hyperglycemia (18% and 11%), and anemia (10% and 15%).
This trial was supported by Bristol-Myers Squibb and AbbVie Biotherapeutics, and researchers reported relationships with several other companies.
Adding elotuzumab to pomalidomide and low-dose dexamethasone can improve progression-free survival (PFS) in patients with relapsed/refractory multiple myeloma (MM), according to the ELOQUENT-3 trial.
These results support the recent U.S. approval of elotuzumab, pomalidomide, and dexamethasone in adults with MM who have received at least two prior therapies, including lenalidomide and a proteasome inhibitor.
Results from ELOQUENT-3 were recently published in The New England Journal of Medicine and previously presented at the 23rd Congress of the European Hematology Association in June.
The phase 2 trial included patients with refractory or relapsed and refractory MM who had received lenalidomide and a proteasome inhibitor.
The patients were randomized to receive elotuzumab plus pomalidomide and low-dose dexamethasone (EPd, n=60) or pomalidomide and low-dose dexamethasone (Pd, n=57) in 28-day cycles until disease progression or unacceptable toxicity.
The overall response rate was 53% in the EPd arm and 26% in the Pd arm (odds ratio=3.25; P=0.0029).
There were two stringent complete responses (CRs) and three CRs in the EPd arm, and there was one CR in the Pd arm.
The median duration of response was 8.3 months in the Pd arm and was not reached in the EPd arm.
The median PFS was 10.3 months with EPd and 4.7 months with Pd (hazard ratio=0.54, P=0.008).
Overall survival data were not mature at the time of analysis, but there was a trend favoring EPd over Pd (hazard ratio=0.62). There were 13 deaths in the EPd arm and 18 in the Pd arm.
The most common treatment-related adverse events (in the EPd and Pd arms, respectively) were neutropenia (18% and 20%), hyperglycemia (18% and 11%), and anemia (10% and 15%).
This trial was supported by Bristol-Myers Squibb and AbbVie Biotherapeutics, and researchers reported relationships with several other companies.
CAR T therapy for MM receives RMAT designation
P-BCMA-101, an autologous chimeric antigen receptor (CAR) T-cell therapy being developed to treat patients with relapsed/refractory multiple myeloma (MM), has received regenerative medicine advanced therapy (RMAT) designation from the U.S. Food and Drug Administration (FDA).
RMAT designation is intended to expedite the development and review of regenerative medicines that are intended to treat, modify, reverse, or cure a serious or life-threatening disease or condition.
For a therapy to receive RMAT designation, preliminary evidence must indicate the therapy has the potential to address unmet medical needs for the disease or condition.
RMAT designation provides all the benefits of fast track and breakthrough therapy designations, including early interactions with the FDA.
About P-BCMA-101
To create P-BCMA-101, patients’ T cells are modified using a non-viral DNA modification system known as piggyBac™. The modified T cells target cells expressing B-cell maturation antigen (BCMA), which is expressed on essentially all MM cells.
Early results from the phase 1 trial (NCT03288493) of P-BCMA-101 were recently reported at the 2018 CAR-TCR Summit by Eric Ostertag, MD, PhD, chief executive officer of Poseida Therapeutics Inc., the company developing P-BCMA-101.
The presentation included data on 11 patients with heavily pretreated MM. Patients were a median age of 60, and 73% were high risk. They had a median of six prior therapies.
Patients received conditioning with fludarabine and cyclophosphamide for 3 days prior to receiving P-BCMA-101. They then received one of three doses of CAR T cells—51×106 (n=3), 152×106 (n=7), or 430×106 (n=1).
The investigators observed no dose-limiting toxicities. Adverse events included neutropenia in eight patients and thrombocytopenia in five.
One patient may have had cytokine release syndrome, but the condition resolved without drug intervention. And investigators observed no neurotoxicity.
Seven of ten patients evaluable for response by International Myeloma Working Group criteria achieved at least a partial response, including very good partial responses and stringent complete response.
The eleventh patient has oligosecretory disease and was only evaluable by PET, which indicated a near complete response.
Additional results from this trial are scheduled to be presented at the 2018 ASH Annual Meeting (abstract 1012).
The study is funded by the California Institute for Regenerative Medicine and Poseida Therapeutics.
P-BCMA-101, an autologous chimeric antigen receptor (CAR) T-cell therapy being developed to treat patients with relapsed/refractory multiple myeloma (MM), has received regenerative medicine advanced therapy (RMAT) designation from the U.S. Food and Drug Administration (FDA).
RMAT designation is intended to expedite the development and review of regenerative medicines that are intended to treat, modify, reverse, or cure a serious or life-threatening disease or condition.
For a therapy to receive RMAT designation, preliminary evidence must indicate the therapy has the potential to address unmet medical needs for the disease or condition.
RMAT designation provides all the benefits of fast track and breakthrough therapy designations, including early interactions with the FDA.
About P-BCMA-101
To create P-BCMA-101, patients’ T cells are modified using a non-viral DNA modification system known as piggyBac™. The modified T cells target cells expressing B-cell maturation antigen (BCMA), which is expressed on essentially all MM cells.
Early results from the phase 1 trial (NCT03288493) of P-BCMA-101 were recently reported at the 2018 CAR-TCR Summit by Eric Ostertag, MD, PhD, chief executive officer of Poseida Therapeutics Inc., the company developing P-BCMA-101.
The presentation included data on 11 patients with heavily pretreated MM. Patients were a median age of 60, and 73% were high risk. They had a median of six prior therapies.
Patients received conditioning with fludarabine and cyclophosphamide for 3 days prior to receiving P-BCMA-101. They then received one of three doses of CAR T cells—51×106 (n=3), 152×106 (n=7), or 430×106 (n=1).
The investigators observed no dose-limiting toxicities. Adverse events included neutropenia in eight patients and thrombocytopenia in five.
One patient may have had cytokine release syndrome, but the condition resolved without drug intervention. And investigators observed no neurotoxicity.
Seven of ten patients evaluable for response by International Myeloma Working Group criteria achieved at least a partial response, including very good partial responses and stringent complete response.
The eleventh patient has oligosecretory disease and was only evaluable by PET, which indicated a near complete response.
Additional results from this trial are scheduled to be presented at the 2018 ASH Annual Meeting (abstract 1012).
The study is funded by the California Institute for Regenerative Medicine and Poseida Therapeutics.
P-BCMA-101, an autologous chimeric antigen receptor (CAR) T-cell therapy being developed to treat patients with relapsed/refractory multiple myeloma (MM), has received regenerative medicine advanced therapy (RMAT) designation from the U.S. Food and Drug Administration (FDA).
RMAT designation is intended to expedite the development and review of regenerative medicines that are intended to treat, modify, reverse, or cure a serious or life-threatening disease or condition.
For a therapy to receive RMAT designation, preliminary evidence must indicate the therapy has the potential to address unmet medical needs for the disease or condition.
RMAT designation provides all the benefits of fast track and breakthrough therapy designations, including early interactions with the FDA.
About P-BCMA-101
To create P-BCMA-101, patients’ T cells are modified using a non-viral DNA modification system known as piggyBac™. The modified T cells target cells expressing B-cell maturation antigen (BCMA), which is expressed on essentially all MM cells.
Early results from the phase 1 trial (NCT03288493) of P-BCMA-101 were recently reported at the 2018 CAR-TCR Summit by Eric Ostertag, MD, PhD, chief executive officer of Poseida Therapeutics Inc., the company developing P-BCMA-101.
The presentation included data on 11 patients with heavily pretreated MM. Patients were a median age of 60, and 73% were high risk. They had a median of six prior therapies.
Patients received conditioning with fludarabine and cyclophosphamide for 3 days prior to receiving P-BCMA-101. They then received one of three doses of CAR T cells—51×106 (n=3), 152×106 (n=7), or 430×106 (n=1).
The investigators observed no dose-limiting toxicities. Adverse events included neutropenia in eight patients and thrombocytopenia in five.
One patient may have had cytokine release syndrome, but the condition resolved without drug intervention. And investigators observed no neurotoxicity.
Seven of ten patients evaluable for response by International Myeloma Working Group criteria achieved at least a partial response, including very good partial responses and stringent complete response.
The eleventh patient has oligosecretory disease and was only evaluable by PET, which indicated a near complete response.
Additional results from this trial are scheduled to be presented at the 2018 ASH Annual Meeting (abstract 1012).
The study is funded by the California Institute for Regenerative Medicine and Poseida Therapeutics.
Vaccine inhibits thrombus formation in mice
Investigators designed an antiplatelet vaccine they say could be a novel therapy to prevent recurrent ischemic stroke.
The vaccine inhibits S100A9 (S100 calcium-binding protein A9)/CD36 (cluster of differentiation 36) signaling in platelets, which is a key signal in arterial thrombosis but not hemostasis.
Mice immunized with the vaccine experienced an antithrombotic effect that lasted 84 days.
The vaccine inhibited thrombus formation without the risk of bleeding or adverse autoimmune responses.
The researchers say the vaccine worked as well as clopidogrel.
“Many stroke patients don’t take their blood thinning drugs as prescribed, which makes it more likely they will have another stroke,” said Hironori Nakagami, MD, of Osaka University Graduate School of Medicine in Japan.
“This vaccine might one day help solve this issue since it would only need to be injected periodically.”
Dr. Nakagami and his colleagues described the vaccine in Hypertension.
S100A9 had previously been reported to be a key molecule in the regulation of thrombus formation. The investigators therefore hypothesized that neutralizing S100A9 with a vaccine would provide sustained antithrombotic effects without increasing the risk of bleeding.
First, the researchers selected a region in S100A9 as a candidate target for the vaccine and conjugated it to keyhole limpet hemocyanin (KLH) as a carrier protein.
They also designed a S100A8 peptide vaccine. S100A8 forms a heterodimer with S100A9 but was not known to play a key role in thrombus formation.
The investigators then randomly assigned male C57BL/6J mice to receive either vaccine or KLH only as control.
The researchers vaccinated mice with S100A9 on days 0, 14, 28, and 126 with either 20 µg or 50 µg of S100A9 peptide. Other mice were vaccinated with S100A8 at a dose of 20 µg on days 0 and 14.
The vaccinated mice developed antibodies against either S100A8 or S100A9, the latter at the 50 µg dose of vaccine. The antibodies recognized the recombinant S100A8 and S100A9 without cross reactivity, which suggested specific binding of each induced antibody.
Assessing thrombus formation
The researchers examined the antithrombotic effects of S100A8 or S100A9 in mouse models of thrombotic middle cerebral artery (MCA) occlusion.
Twenty-one days after the final vaccination, mice immunized with S100A9 had significantly delayed MCA occlusion, with a greater antithrombotic effect in the 50 µg group.
This antithrombotic effect, the investigators said, was comparable to that observed in mice treated with clopidogrel at a dose of 6 mg/kg for 2 days.
The S100A8 vaccine did not prevent MCA occlusion, so the researchers focused on the S100A9 vaccine in subsequent experiments.
They studied the vaccine’s antithrombotic effects in a model of common carotid artery (CCA) occlusion and observed results similar to those seen in the MCA model.
Specifically, there was no significant difference in the CCA occlusion time between mice vaccinated with S100A9 and mice treated with clopidogrel.
The investigators also found the antithrombotic effect of the S100A9 vaccine lasted more than 2 months.
Post-stroke model
Dr. Nakagami and his colleagues then examined the antithrombotic effect of S100A9 in mice after a transient MCA stroke.
The researchers administered the S100A9 vaccine every 2 weeks, starting 1 week after the stroke, for a total of three times.
The vaccine induced anti-S100A9 antibodies, and the mice had significantly prolonged CCA occlusion times compared to KLH control mice. (The investigators said they assessed CCA occlusion because it was impossible to measure the regional cerebral blood flow after a transient MCA occlusion due to post-surgical cranial skin adhesions.)
This result suggested that S100A9 could prevent thrombus formation in mice that had already had a stroke.
Hemostatic parameters and safety
Additional experiments indicated that the S100A9 vaccine had no effect on hemostatic parameters as indicated by tail bleeding time, platelet count, activated partial thromboplastin time, and prothrombin time.
The researchers also evaluated the vaccine’s safety by examining immunologic responses.
Histological analysis at day 147 after the first immunization revealed no pathological changes, such as CD4+ T-cell infiltration or activation of F4/80+ microglia/macrophages.
The immunized mice had predominantly IgG1 responses. This indicated that the S100A9 vaccine selectively induced primary T helper 2 responses.
“We are continuing our research in hopes of being able to start clinical trials between 5 and 10 years from now, but there are differences between mice and humans in how the vaccine will be recognized by the immune system,” Dr. Nakagami explained.
“We should be able to overcome such problems and believe this vaccine provides a very promising strategy in secondary prevention of stroke.”
This research was funded by the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the Japan Cardiovascular Research Foundation, the SENSHIN Medical Research Foundation, and the Japan Agency for Medical Research and Development.
Dr. Nakagami and five of the eleven authors have applied for a patent for the S100A9 vaccine.
Dr. Nakagami and five other authors are affiliated with departments at Osaka University that receive financial support from AnGes, DAICEL, FunPep, Novartis, Shionogi, Boehringer, and Rohto. One author is a founder, stockholder, and former board member of AnGes.
Investigators designed an antiplatelet vaccine they say could be a novel therapy to prevent recurrent ischemic stroke.
The vaccine inhibits S100A9 (S100 calcium-binding protein A9)/CD36 (cluster of differentiation 36) signaling in platelets, which is a key signal in arterial thrombosis but not hemostasis.
Mice immunized with the vaccine experienced an antithrombotic effect that lasted 84 days.
The vaccine inhibited thrombus formation without the risk of bleeding or adverse autoimmune responses.
The researchers say the vaccine worked as well as clopidogrel.
“Many stroke patients don’t take their blood thinning drugs as prescribed, which makes it more likely they will have another stroke,” said Hironori Nakagami, MD, of Osaka University Graduate School of Medicine in Japan.
“This vaccine might one day help solve this issue since it would only need to be injected periodically.”
Dr. Nakagami and his colleagues described the vaccine in Hypertension.
S100A9 had previously been reported to be a key molecule in the regulation of thrombus formation. The investigators therefore hypothesized that neutralizing S100A9 with a vaccine would provide sustained antithrombotic effects without increasing the risk of bleeding.
First, the researchers selected a region in S100A9 as a candidate target for the vaccine and conjugated it to keyhole limpet hemocyanin (KLH) as a carrier protein.
They also designed a S100A8 peptide vaccine. S100A8 forms a heterodimer with S100A9 but was not known to play a key role in thrombus formation.
The investigators then randomly assigned male C57BL/6J mice to receive either vaccine or KLH only as control.
The researchers vaccinated mice with S100A9 on days 0, 14, 28, and 126 with either 20 µg or 50 µg of S100A9 peptide. Other mice were vaccinated with S100A8 at a dose of 20 µg on days 0 and 14.
The vaccinated mice developed antibodies against either S100A8 or S100A9, the latter at the 50 µg dose of vaccine. The antibodies recognized the recombinant S100A8 and S100A9 without cross reactivity, which suggested specific binding of each induced antibody.
Assessing thrombus formation
The researchers examined the antithrombotic effects of S100A8 or S100A9 in mouse models of thrombotic middle cerebral artery (MCA) occlusion.
Twenty-one days after the final vaccination, mice immunized with S100A9 had significantly delayed MCA occlusion, with a greater antithrombotic effect in the 50 µg group.
This antithrombotic effect, the investigators said, was comparable to that observed in mice treated with clopidogrel at a dose of 6 mg/kg for 2 days.
The S100A8 vaccine did not prevent MCA occlusion, so the researchers focused on the S100A9 vaccine in subsequent experiments.
They studied the vaccine’s antithrombotic effects in a model of common carotid artery (CCA) occlusion and observed results similar to those seen in the MCA model.
Specifically, there was no significant difference in the CCA occlusion time between mice vaccinated with S100A9 and mice treated with clopidogrel.
The investigators also found the antithrombotic effect of the S100A9 vaccine lasted more than 2 months.
Post-stroke model
Dr. Nakagami and his colleagues then examined the antithrombotic effect of S100A9 in mice after a transient MCA stroke.
The researchers administered the S100A9 vaccine every 2 weeks, starting 1 week after the stroke, for a total of three times.
The vaccine induced anti-S100A9 antibodies, and the mice had significantly prolonged CCA occlusion times compared to KLH control mice. (The investigators said they assessed CCA occlusion because it was impossible to measure the regional cerebral blood flow after a transient MCA occlusion due to post-surgical cranial skin adhesions.)
This result suggested that S100A9 could prevent thrombus formation in mice that had already had a stroke.
Hemostatic parameters and safety
Additional experiments indicated that the S100A9 vaccine had no effect on hemostatic parameters as indicated by tail bleeding time, platelet count, activated partial thromboplastin time, and prothrombin time.
The researchers also evaluated the vaccine’s safety by examining immunologic responses.
Histological analysis at day 147 after the first immunization revealed no pathological changes, such as CD4+ T-cell infiltration or activation of F4/80+ microglia/macrophages.
The immunized mice had predominantly IgG1 responses. This indicated that the S100A9 vaccine selectively induced primary T helper 2 responses.
“We are continuing our research in hopes of being able to start clinical trials between 5 and 10 years from now, but there are differences between mice and humans in how the vaccine will be recognized by the immune system,” Dr. Nakagami explained.
“We should be able to overcome such problems and believe this vaccine provides a very promising strategy in secondary prevention of stroke.”
This research was funded by the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the Japan Cardiovascular Research Foundation, the SENSHIN Medical Research Foundation, and the Japan Agency for Medical Research and Development.
Dr. Nakagami and five of the eleven authors have applied for a patent for the S100A9 vaccine.
Dr. Nakagami and five other authors are affiliated with departments at Osaka University that receive financial support from AnGes, DAICEL, FunPep, Novartis, Shionogi, Boehringer, and Rohto. One author is a founder, stockholder, and former board member of AnGes.
Investigators designed an antiplatelet vaccine they say could be a novel therapy to prevent recurrent ischemic stroke.
The vaccine inhibits S100A9 (S100 calcium-binding protein A9)/CD36 (cluster of differentiation 36) signaling in platelets, which is a key signal in arterial thrombosis but not hemostasis.
Mice immunized with the vaccine experienced an antithrombotic effect that lasted 84 days.
The vaccine inhibited thrombus formation without the risk of bleeding or adverse autoimmune responses.
The researchers say the vaccine worked as well as clopidogrel.
“Many stroke patients don’t take their blood thinning drugs as prescribed, which makes it more likely they will have another stroke,” said Hironori Nakagami, MD, of Osaka University Graduate School of Medicine in Japan.
“This vaccine might one day help solve this issue since it would only need to be injected periodically.”
Dr. Nakagami and his colleagues described the vaccine in Hypertension.
S100A9 had previously been reported to be a key molecule in the regulation of thrombus formation. The investigators therefore hypothesized that neutralizing S100A9 with a vaccine would provide sustained antithrombotic effects without increasing the risk of bleeding.
First, the researchers selected a region in S100A9 as a candidate target for the vaccine and conjugated it to keyhole limpet hemocyanin (KLH) as a carrier protein.
They also designed a S100A8 peptide vaccine. S100A8 forms a heterodimer with S100A9 but was not known to play a key role in thrombus formation.
The investigators then randomly assigned male C57BL/6J mice to receive either vaccine or KLH only as control.
The researchers vaccinated mice with S100A9 on days 0, 14, 28, and 126 with either 20 µg or 50 µg of S100A9 peptide. Other mice were vaccinated with S100A8 at a dose of 20 µg on days 0 and 14.
The vaccinated mice developed antibodies against either S100A8 or S100A9, the latter at the 50 µg dose of vaccine. The antibodies recognized the recombinant S100A8 and S100A9 without cross reactivity, which suggested specific binding of each induced antibody.
Assessing thrombus formation
The researchers examined the antithrombotic effects of S100A8 or S100A9 in mouse models of thrombotic middle cerebral artery (MCA) occlusion.
Twenty-one days after the final vaccination, mice immunized with S100A9 had significantly delayed MCA occlusion, with a greater antithrombotic effect in the 50 µg group.
This antithrombotic effect, the investigators said, was comparable to that observed in mice treated with clopidogrel at a dose of 6 mg/kg for 2 days.
The S100A8 vaccine did not prevent MCA occlusion, so the researchers focused on the S100A9 vaccine in subsequent experiments.
They studied the vaccine’s antithrombotic effects in a model of common carotid artery (CCA) occlusion and observed results similar to those seen in the MCA model.
Specifically, there was no significant difference in the CCA occlusion time between mice vaccinated with S100A9 and mice treated with clopidogrel.
The investigators also found the antithrombotic effect of the S100A9 vaccine lasted more than 2 months.
Post-stroke model
Dr. Nakagami and his colleagues then examined the antithrombotic effect of S100A9 in mice after a transient MCA stroke.
The researchers administered the S100A9 vaccine every 2 weeks, starting 1 week after the stroke, for a total of three times.
The vaccine induced anti-S100A9 antibodies, and the mice had significantly prolonged CCA occlusion times compared to KLH control mice. (The investigators said they assessed CCA occlusion because it was impossible to measure the regional cerebral blood flow after a transient MCA occlusion due to post-surgical cranial skin adhesions.)
This result suggested that S100A9 could prevent thrombus formation in mice that had already had a stroke.
Hemostatic parameters and safety
Additional experiments indicated that the S100A9 vaccine had no effect on hemostatic parameters as indicated by tail bleeding time, platelet count, activated partial thromboplastin time, and prothrombin time.
The researchers also evaluated the vaccine’s safety by examining immunologic responses.
Histological analysis at day 147 after the first immunization revealed no pathological changes, such as CD4+ T-cell infiltration or activation of F4/80+ microglia/macrophages.
The immunized mice had predominantly IgG1 responses. This indicated that the S100A9 vaccine selectively induced primary T helper 2 responses.
“We are continuing our research in hopes of being able to start clinical trials between 5 and 10 years from now, but there are differences between mice and humans in how the vaccine will be recognized by the immune system,” Dr. Nakagami explained.
“We should be able to overcome such problems and believe this vaccine provides a very promising strategy in secondary prevention of stroke.”
This research was funded by the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the Japan Cardiovascular Research Foundation, the SENSHIN Medical Research Foundation, and the Japan Agency for Medical Research and Development.
Dr. Nakagami and five of the eleven authors have applied for a patent for the S100A9 vaccine.
Dr. Nakagami and five other authors are affiliated with departments at Osaka University that receive financial support from AnGes, DAICEL, FunPep, Novartis, Shionogi, Boehringer, and Rohto. One author is a founder, stockholder, and former board member of AnGes.
Combo worsens platelet recovery in MDS
Results from the phase 3 SUPPORT trial suggest there is no role for the combination of eltrombopag and azacitidine in patients with intermediate- or high-risk myelodysplastic syndromes (MDS), according to investigators.
Adding eltrombopag to azacitidine worsened platelet recovery, reduced the overall response rate, and did not improve overall or progression-free survival when compared to azacitidine plus placebo.
Investigators had hypothesized that eltrombopag would reduce the thrombocytopenia exacerbated by azacitidine treatment.
Not only was this not the case, but the investigators observed a trend toward increased progression to acute myeloid leukemia (AML) in eltrombopag-treated patients.
An independent monitoring committee recommended the trial be terminated early.
Michael Dickinson, MBBS, of Peter MacCallum Cancer Centre and Royal Melbourne Hospital in Melbourne, Australia, and his colleagues reported results from the trial in Blood.
The investigators conducted the SUPPORT study (NCT02158936) to investigate the efficacy and safety of eltrombopag as platelet supportive care in patients with intermediate- to high-risk MDS and thrombocytopenia who were receiving azacitidine.
Study design
Investigators randomized patients 1:1 to eltrombopag or placebo in combination with azacitidine. Patients had to have at least one platelet count less than 75 x 109/L within 28 days prior to the first azacitidine dose.
Patients received azacitidine subcutaneously at a dose of 75 mg/m2 once daily on 7 consecutive days for 28 days.
Patients received eltrombopag or placebo at a starting dose of 200 mg/day (100 mg/day for East Asians), adjusted by 100 mg increments (50 mg increments for East Asians) to a maximum of 300 mg/day (150 mg/day for East Asians).
Patient disposition
Investigators enrolled 356 patients from 30 countries between June 2014 and December 2015, randomizing 179 patients to eltrombopag and 177 to placebo.
Patients were a median age of 70, 66% were male, 83% were white, and 47% had platelet counts between 20 and 50 x 109/L.
At the start of the trial, 19% of patients (n=66) were platelet transfusion-dependent—16% (29/179) in the eltrombopag group and 21% (37/177) in the placebo group.
Treatment exposure
Patients received eltrombopag for a median of 83 days (range, 60 to 148) and placebo for a median of 149 days (range, 8 to 503).
For non-East Asian patients, the mean dose of eltrombopag was 205 mg/day (range, 65 to 293), and the mean dose of placebo was 245 mg/day (range, 107 to 316).
Sixty-eight patients (38%) in the eltrombopag arm received the recommended number of six or more azacitidine cycles, compared to 91 (51%) in the placebo arm.
Two patients in the eltrombopag arm did not receive treatment. Therefore, the safety analyses were conducted on the 177 treated patients in each arm.
Safety
The most common reason for treatment discontinuation was termination of the study. Thirty-two percent of patients in the eltrombopag cohort and 44% in the placebo cohort discontinued for this reason.
Patients had to discontinue eltrombopag or placebo if azacitidine was discontinued, and this occurred in 30% of patients in the eltrombopag group and 26% in the placebo group.
Twenty-two percent of eltrombopag-treated patients discontinued due to adverse events (AEs), compared with 14% in the placebo group.
The most common reasons for azacitidine discontinuation in the eltrombopag and placebo arms, respectively, were study termination (35% and 46%), AEs (25% and 19%), disease progression (15% and 14%), and patient decision (11% and 9%).
AEs with the greatest difference between the eltrombopag and placebo arms, respectively, were febrile neutropenia (31% and 21%), neutropenia (31% and 26%), nausea (31% and 26%), and diarrhea (25% and 14%).
Sixty-three of the 177 AEs in the eltrombopag group were suspected to be treatment-related, compared to 42 of 173 AEs in the placebo group.
One hundred twenty-eight (72%) serious AEs occurred in the eltrombopag arm, compared to 100 (56%) in the placebo arm.
Fifteen percent of the serious AEs were suspected to be related to eltrombopag treatment, compared to 4% of the serious AEs in the placebo group.
At the final analysis, 108 patients had died—57 (32%) in the eltrombopag group and 51 (29%) in the placebo group (hazard ratio [HR] 1.42; 95% CI 0.97, 2.08; nominal P=0.164).
The main causes of death were disease progression (28 in the eltrombopag group and 21 in the placebo group) and sepsis (18 in the eltrombopag group and 13 in the placebo group).
Efficacy
The study’s primary endpoint was the proportion of patients free of platelet transfusions during cycles one to four of azacitidine therapy.
At the final analysis, there were fewer patients in the eltrombopag arm than in the placebo arm who had achieved platelet transfusion independence—16% (28/179) and 31% (55/177), respectively. The odds ratio (OR) was 0.37 (95% CI 0.21, 0.65; two-sided P value=0.001).
Secondary efficacy endpoints included overall survival, disease response, duration of response, progression to AML, progression-free survival, and hematologic improvement.
The investigators pointed out that no formal statistical tests were performed for the secondary endpoints. Therefore, “statistical interpretation should be made with caution,” they wrote.
Overall response by IWG criteria occurred in 20% of patients in the eltrombopag group and 35% of those in the placebo group, according to investigator assessment (OR=0.51; 95% CI 0.30, 0.86; nominal P=0.005).
There was no significant difference in hematologic improvement, overall survival, or progression-free survival between the treatment arms.
The investigators found a higher rate of progression to AML in the eltrombopag arm than in the placebo arm—15% and 9%, respectively (OR=1.59; 95% CI 0.81, 3.14; nominal P=0.079).
They pointed out that these results contrasted with those of recent clinical studies of eltrombopag monotherapy1,2,3,4 in patients with MDS.
“[T]he findings of this trial were unexpected,” the investigators wrote.
They hypothesized that eltrombopag inhibits the effects of azacitidine when the drugs are given concomitantly. The issue is being studied further with ongoing research, they said.
The authors acknowledged financial support for medical editorial assistance provided by Novartis Pharmaceuticals Corporation.
Dr. Dickinson disclosed relationships with Celgene, Novartis, Janssen, Pfizer, and Roche. Senior study author Uwe Platzbecker, MD, disclosed relationships with Amgen, Celgene, GlaxoSmithKline, and Novartis.
1. Platzbecker U, et al Lancet Haematol. 2015;2(10):e417-e426. DOI: https://doi.org/10.1016/S2352-3026(15)00149-0
2. Oliva EN, et al. Lancet Haematol. 2017;4(3):e127-e136. DOI: https://doi.org/10.1016/S2352-3026(17)30012-1
3. Mittelman M, et al. Lancet Haematol. 2017;5(1):e34-e43. DOI: https://doi.org/10.1016/S2352-3026(17)30228-4
4. Buckstein R. Lancet Haematol. 2015;2(10):e396-e397. DOI: https://doi.org/10.1016/S2352-3026(15)00200-8
Results from the phase 3 SUPPORT trial suggest there is no role for the combination of eltrombopag and azacitidine in patients with intermediate- or high-risk myelodysplastic syndromes (MDS), according to investigators.
Adding eltrombopag to azacitidine worsened platelet recovery, reduced the overall response rate, and did not improve overall or progression-free survival when compared to azacitidine plus placebo.
Investigators had hypothesized that eltrombopag would reduce the thrombocytopenia exacerbated by azacitidine treatment.
Not only was this not the case, but the investigators observed a trend toward increased progression to acute myeloid leukemia (AML) in eltrombopag-treated patients.
An independent monitoring committee recommended the trial be terminated early.
Michael Dickinson, MBBS, of Peter MacCallum Cancer Centre and Royal Melbourne Hospital in Melbourne, Australia, and his colleagues reported results from the trial in Blood.
The investigators conducted the SUPPORT study (NCT02158936) to investigate the efficacy and safety of eltrombopag as platelet supportive care in patients with intermediate- to high-risk MDS and thrombocytopenia who were receiving azacitidine.
Study design
Investigators randomized patients 1:1 to eltrombopag or placebo in combination with azacitidine. Patients had to have at least one platelet count less than 75 x 109/L within 28 days prior to the first azacitidine dose.
Patients received azacitidine subcutaneously at a dose of 75 mg/m2 once daily on 7 consecutive days for 28 days.
Patients received eltrombopag or placebo at a starting dose of 200 mg/day (100 mg/day for East Asians), adjusted by 100 mg increments (50 mg increments for East Asians) to a maximum of 300 mg/day (150 mg/day for East Asians).
Patient disposition
Investigators enrolled 356 patients from 30 countries between June 2014 and December 2015, randomizing 179 patients to eltrombopag and 177 to placebo.
Patients were a median age of 70, 66% were male, 83% were white, and 47% had platelet counts between 20 and 50 x 109/L.
At the start of the trial, 19% of patients (n=66) were platelet transfusion-dependent—16% (29/179) in the eltrombopag group and 21% (37/177) in the placebo group.
Treatment exposure
Patients received eltrombopag for a median of 83 days (range, 60 to 148) and placebo for a median of 149 days (range, 8 to 503).
For non-East Asian patients, the mean dose of eltrombopag was 205 mg/day (range, 65 to 293), and the mean dose of placebo was 245 mg/day (range, 107 to 316).
Sixty-eight patients (38%) in the eltrombopag arm received the recommended number of six or more azacitidine cycles, compared to 91 (51%) in the placebo arm.
Two patients in the eltrombopag arm did not receive treatment. Therefore, the safety analyses were conducted on the 177 treated patients in each arm.
Safety
The most common reason for treatment discontinuation was termination of the study. Thirty-two percent of patients in the eltrombopag cohort and 44% in the placebo cohort discontinued for this reason.
Patients had to discontinue eltrombopag or placebo if azacitidine was discontinued, and this occurred in 30% of patients in the eltrombopag group and 26% in the placebo group.
Twenty-two percent of eltrombopag-treated patients discontinued due to adverse events (AEs), compared with 14% in the placebo group.
The most common reasons for azacitidine discontinuation in the eltrombopag and placebo arms, respectively, were study termination (35% and 46%), AEs (25% and 19%), disease progression (15% and 14%), and patient decision (11% and 9%).
AEs with the greatest difference between the eltrombopag and placebo arms, respectively, were febrile neutropenia (31% and 21%), neutropenia (31% and 26%), nausea (31% and 26%), and diarrhea (25% and 14%).
Sixty-three of the 177 AEs in the eltrombopag group were suspected to be treatment-related, compared to 42 of 173 AEs in the placebo group.
One hundred twenty-eight (72%) serious AEs occurred in the eltrombopag arm, compared to 100 (56%) in the placebo arm.
Fifteen percent of the serious AEs were suspected to be related to eltrombopag treatment, compared to 4% of the serious AEs in the placebo group.
At the final analysis, 108 patients had died—57 (32%) in the eltrombopag group and 51 (29%) in the placebo group (hazard ratio [HR] 1.42; 95% CI 0.97, 2.08; nominal P=0.164).
The main causes of death were disease progression (28 in the eltrombopag group and 21 in the placebo group) and sepsis (18 in the eltrombopag group and 13 in the placebo group).
Efficacy
The study’s primary endpoint was the proportion of patients free of platelet transfusions during cycles one to four of azacitidine therapy.
At the final analysis, there were fewer patients in the eltrombopag arm than in the placebo arm who had achieved platelet transfusion independence—16% (28/179) and 31% (55/177), respectively. The odds ratio (OR) was 0.37 (95% CI 0.21, 0.65; two-sided P value=0.001).
Secondary efficacy endpoints included overall survival, disease response, duration of response, progression to AML, progression-free survival, and hematologic improvement.
The investigators pointed out that no formal statistical tests were performed for the secondary endpoints. Therefore, “statistical interpretation should be made with caution,” they wrote.
Overall response by IWG criteria occurred in 20% of patients in the eltrombopag group and 35% of those in the placebo group, according to investigator assessment (OR=0.51; 95% CI 0.30, 0.86; nominal P=0.005).
There was no significant difference in hematologic improvement, overall survival, or progression-free survival between the treatment arms.
The investigators found a higher rate of progression to AML in the eltrombopag arm than in the placebo arm—15% and 9%, respectively (OR=1.59; 95% CI 0.81, 3.14; nominal P=0.079).
They pointed out that these results contrasted with those of recent clinical studies of eltrombopag monotherapy1,2,3,4 in patients with MDS.
“[T]he findings of this trial were unexpected,” the investigators wrote.
They hypothesized that eltrombopag inhibits the effects of azacitidine when the drugs are given concomitantly. The issue is being studied further with ongoing research, they said.
The authors acknowledged financial support for medical editorial assistance provided by Novartis Pharmaceuticals Corporation.
Dr. Dickinson disclosed relationships with Celgene, Novartis, Janssen, Pfizer, and Roche. Senior study author Uwe Platzbecker, MD, disclosed relationships with Amgen, Celgene, GlaxoSmithKline, and Novartis.
1. Platzbecker U, et al Lancet Haematol. 2015;2(10):e417-e426. DOI: https://doi.org/10.1016/S2352-3026(15)00149-0
2. Oliva EN, et al. Lancet Haematol. 2017;4(3):e127-e136. DOI: https://doi.org/10.1016/S2352-3026(17)30012-1
3. Mittelman M, et al. Lancet Haematol. 2017;5(1):e34-e43. DOI: https://doi.org/10.1016/S2352-3026(17)30228-4
4. Buckstein R. Lancet Haematol. 2015;2(10):e396-e397. DOI: https://doi.org/10.1016/S2352-3026(15)00200-8
Results from the phase 3 SUPPORT trial suggest there is no role for the combination of eltrombopag and azacitidine in patients with intermediate- or high-risk myelodysplastic syndromes (MDS), according to investigators.
Adding eltrombopag to azacitidine worsened platelet recovery, reduced the overall response rate, and did not improve overall or progression-free survival when compared to azacitidine plus placebo.
Investigators had hypothesized that eltrombopag would reduce the thrombocytopenia exacerbated by azacitidine treatment.
Not only was this not the case, but the investigators observed a trend toward increased progression to acute myeloid leukemia (AML) in eltrombopag-treated patients.
An independent monitoring committee recommended the trial be terminated early.
Michael Dickinson, MBBS, of Peter MacCallum Cancer Centre and Royal Melbourne Hospital in Melbourne, Australia, and his colleagues reported results from the trial in Blood.
The investigators conducted the SUPPORT study (NCT02158936) to investigate the efficacy and safety of eltrombopag as platelet supportive care in patients with intermediate- to high-risk MDS and thrombocytopenia who were receiving azacitidine.
Study design
Investigators randomized patients 1:1 to eltrombopag or placebo in combination with azacitidine. Patients had to have at least one platelet count less than 75 x 109/L within 28 days prior to the first azacitidine dose.
Patients received azacitidine subcutaneously at a dose of 75 mg/m2 once daily on 7 consecutive days for 28 days.
Patients received eltrombopag or placebo at a starting dose of 200 mg/day (100 mg/day for East Asians), adjusted by 100 mg increments (50 mg increments for East Asians) to a maximum of 300 mg/day (150 mg/day for East Asians).
Patient disposition
Investigators enrolled 356 patients from 30 countries between June 2014 and December 2015, randomizing 179 patients to eltrombopag and 177 to placebo.
Patients were a median age of 70, 66% were male, 83% were white, and 47% had platelet counts between 20 and 50 x 109/L.
At the start of the trial, 19% of patients (n=66) were platelet transfusion-dependent—16% (29/179) in the eltrombopag group and 21% (37/177) in the placebo group.
Treatment exposure
Patients received eltrombopag for a median of 83 days (range, 60 to 148) and placebo for a median of 149 days (range, 8 to 503).
For non-East Asian patients, the mean dose of eltrombopag was 205 mg/day (range, 65 to 293), and the mean dose of placebo was 245 mg/day (range, 107 to 316).
Sixty-eight patients (38%) in the eltrombopag arm received the recommended number of six or more azacitidine cycles, compared to 91 (51%) in the placebo arm.
Two patients in the eltrombopag arm did not receive treatment. Therefore, the safety analyses were conducted on the 177 treated patients in each arm.
Safety
The most common reason for treatment discontinuation was termination of the study. Thirty-two percent of patients in the eltrombopag cohort and 44% in the placebo cohort discontinued for this reason.
Patients had to discontinue eltrombopag or placebo if azacitidine was discontinued, and this occurred in 30% of patients in the eltrombopag group and 26% in the placebo group.
Twenty-two percent of eltrombopag-treated patients discontinued due to adverse events (AEs), compared with 14% in the placebo group.
The most common reasons for azacitidine discontinuation in the eltrombopag and placebo arms, respectively, were study termination (35% and 46%), AEs (25% and 19%), disease progression (15% and 14%), and patient decision (11% and 9%).
AEs with the greatest difference between the eltrombopag and placebo arms, respectively, were febrile neutropenia (31% and 21%), neutropenia (31% and 26%), nausea (31% and 26%), and diarrhea (25% and 14%).
Sixty-three of the 177 AEs in the eltrombopag group were suspected to be treatment-related, compared to 42 of 173 AEs in the placebo group.
One hundred twenty-eight (72%) serious AEs occurred in the eltrombopag arm, compared to 100 (56%) in the placebo arm.
Fifteen percent of the serious AEs were suspected to be related to eltrombopag treatment, compared to 4% of the serious AEs in the placebo group.
At the final analysis, 108 patients had died—57 (32%) in the eltrombopag group and 51 (29%) in the placebo group (hazard ratio [HR] 1.42; 95% CI 0.97, 2.08; nominal P=0.164).
The main causes of death were disease progression (28 in the eltrombopag group and 21 in the placebo group) and sepsis (18 in the eltrombopag group and 13 in the placebo group).
Efficacy
The study’s primary endpoint was the proportion of patients free of platelet transfusions during cycles one to four of azacitidine therapy.
At the final analysis, there were fewer patients in the eltrombopag arm than in the placebo arm who had achieved platelet transfusion independence—16% (28/179) and 31% (55/177), respectively. The odds ratio (OR) was 0.37 (95% CI 0.21, 0.65; two-sided P value=0.001).
Secondary efficacy endpoints included overall survival, disease response, duration of response, progression to AML, progression-free survival, and hematologic improvement.
The investigators pointed out that no formal statistical tests were performed for the secondary endpoints. Therefore, “statistical interpretation should be made with caution,” they wrote.
Overall response by IWG criteria occurred in 20% of patients in the eltrombopag group and 35% of those in the placebo group, according to investigator assessment (OR=0.51; 95% CI 0.30, 0.86; nominal P=0.005).
There was no significant difference in hematologic improvement, overall survival, or progression-free survival between the treatment arms.
The investigators found a higher rate of progression to AML in the eltrombopag arm than in the placebo arm—15% and 9%, respectively (OR=1.59; 95% CI 0.81, 3.14; nominal P=0.079).
They pointed out that these results contrasted with those of recent clinical studies of eltrombopag monotherapy1,2,3,4 in patients with MDS.
“[T]he findings of this trial were unexpected,” the investigators wrote.
They hypothesized that eltrombopag inhibits the effects of azacitidine when the drugs are given concomitantly. The issue is being studied further with ongoing research, they said.
The authors acknowledged financial support for medical editorial assistance provided by Novartis Pharmaceuticals Corporation.
Dr. Dickinson disclosed relationships with Celgene, Novartis, Janssen, Pfizer, and Roche. Senior study author Uwe Platzbecker, MD, disclosed relationships with Amgen, Celgene, GlaxoSmithKline, and Novartis.
1. Platzbecker U, et al Lancet Haematol. 2015;2(10):e417-e426. DOI: https://doi.org/10.1016/S2352-3026(15)00149-0
2. Oliva EN, et al. Lancet Haematol. 2017;4(3):e127-e136. DOI: https://doi.org/10.1016/S2352-3026(17)30012-1
3. Mittelman M, et al. Lancet Haematol. 2017;5(1):e34-e43. DOI: https://doi.org/10.1016/S2352-3026(17)30228-4
4. Buckstein R. Lancet Haematol. 2015;2(10):e396-e397. DOI: https://doi.org/10.1016/S2352-3026(15)00200-8
Molecule enhances PI activity in multiple myeloma
Researchers say they have identified a new class of protein disulfide isomerase (PDI) inhibitors that sensitize multiple myeloma (MM) cells to proteasome inhibitors (PIs).
The investigators screened approximately 20,000 compounds spanning multiple chemical libraries and found the compound E61 to be a “striking hit,” with a six-fold increase in bortezomib cytotoxicity and the ability to re-sensitize PI activity at low micromolar concentrations.
The researchers then synthesized and evaluated 150 E61 derivatives and discovered the lead candidate, E64FC26, which was highly synergistic with PIs at concentrations as low as 200 nM.
They reported that E64FC26 has “several advantages over previously reported PDI inhibitors, including superior potency and a pan-style mode of inhibition.”
“PDI is an attractive target in oncology, but good PDI inhibitors have been hard to find,” said Nathan G. Dolloff, PhD, of the Medical University of South Carolina (MUSC) in Charleston.
“The compounds we discovered have a lot of advantages, including high potency and good drug-like properties. We hope that those strengths translate into an effective new drug that can ultimately help patients.”
Dr. Dolloff and his colleagues reported their discovery in Leukemia.
The investigators detected the synergistic effects of E61 in combination with next-generation PIs, including carfilzomib, ixazomib, and oprozomib in both PI-sensitive and -resistant MM cell lines.
On the other hand, E61 had no effect on dexamethasone activity in dexamethasone-resistant cells. And E61 did not affect lenalidomide or doxorubicin cytotoxicity in PI-resistant cells.
The researchers also determined that E61 was only active in MM cells. E61 did not enhance the cytotoxic effects of PIs in normal cells.
This selective toxicity suggests that E61 may have “a wide therapeutic index in vivo,” the investigators wrote.
In vivo activity
To investigate the anti-MM activity and tolerability of E61 in vivo, the researchers treated a NOD-SCID IL2Rγ−/− mouse model with E61 at a continuous dose of 50 mg/kg/day.
E61 prolonged survival by 11 days in the treated mice (P=0.0007), and four of the 11 treated mice survived to the experiment’s end.
In another experiment, two of eight mice achieved a complete response.
After continuous dosing for 40 or more days, E61 was well tolerated, the investigators reported. The mice showed no overt signs of distress and did not lose weight.
Molecular target of E61
Using click (Cu(I)-catalyzed azide-alkyne cycloaddition) chemistry and a proteomics approach, the researchers then confirmed that PDI family members are the molecular target of E61.
Functional studies indicated that E61 inhibited PDI reductase activity in vitro. E61 also enhanced the accumulation of ubiquitinylated proteins and produced strong endoplasmic reticulum (ER) and oxidative stress responses when combined with PIs.
Anti-MM activity of E64FC26
The investigators used a structure activity relationship program to narrow the candidate molecules down to E64FC26.
E64FC26 demonstrated pan-inhibition in that it inhibited all members of the PDI family tested, including PDIA3, PDIA4, TXNDC5, and PDIA6.
E64FC26 also had greater in vitro potency against PDIA1 and the other PDI isoforms. It was the only compound to sensitize MM cells to PIs, with an average increase in PI sensitivity ranging from six- to seven-fold.
The researchers also noted that E64FC26 was superior to other PDI inhibitors they tested in activating ER stress.
The investigators tested the activity of E64FC26 in vivo using Vk*MYC transgenic mice, a model that closely resembles human MM.
Mice treated with E64FC26 had an immediate anti-MM response. Serum M-protein decreased in all mice by an average of 33 ± 7.9% (P=0.0135).
The investigators observed similar effects in a human xenotransplant MM model.
These mice were randomized to receive treatment with vehicle, E64FC26 (2 mg/kg for 3 days/week), bortezomib (0.25 mg/kg for 2 days/week), or a combination of the two agents.
E64FC26 increased median survival by 2 weeks compared with vehicle-treated mice (P<0.0001). By day 36, no vehicle-treated mouse survived, compared with 100% of the E64FC26-treated mice.
Single-agent bortezomib increased survival by 6 days (P=0.0007).
And the combination produced the greatest improvement in median survival, increasing it by 20 days (P<0.0001).
The investigators reported no overt toxicity or body weight fluctuation for mice treated with E64FC26 or the combination.
“These results provide preclinical proof of concept for the strategy of targeting PDI with this new class of compound for the treatment of MM,” the researchers concluded.
“One of the strengths of this study is that we spanned almost the entire drug discovery process,” Dr. Dolloff said. “We screened thousands of compounds, found an exciting molecule, deconvoluted what its binding target was, synthesized hundreds of derivatives to make it better, and then conducted animal studies.”
“The study has everything from biochemistry and cell biology to medicinal chemistry and animal pharmacology in it. There is still a lot of work to be done before this drug is ready for clinical trials in humans, but it has been a rewarding project, and I’m looking forward to the next steps.”
Dr. Dolloff is founder of Leukogene Therapeutics, Inc., which has licensed patents from MUSC, and a second study author is an inventor on patents. The other authors declared no conflicts of interest.
The research was supported by the National Institutes of Health/National Cancer Institute, the South Carolina Clinical & Translational Research Institute, the MUSC Hollings Cancer Center, and by the Hollings Cancer Center T32 Ruth L. Kirschstein National Research Service Award Training Program.
Researchers say they have identified a new class of protein disulfide isomerase (PDI) inhibitors that sensitize multiple myeloma (MM) cells to proteasome inhibitors (PIs).
The investigators screened approximately 20,000 compounds spanning multiple chemical libraries and found the compound E61 to be a “striking hit,” with a six-fold increase in bortezomib cytotoxicity and the ability to re-sensitize PI activity at low micromolar concentrations.
The researchers then synthesized and evaluated 150 E61 derivatives and discovered the lead candidate, E64FC26, which was highly synergistic with PIs at concentrations as low as 200 nM.
They reported that E64FC26 has “several advantages over previously reported PDI inhibitors, including superior potency and a pan-style mode of inhibition.”
“PDI is an attractive target in oncology, but good PDI inhibitors have been hard to find,” said Nathan G. Dolloff, PhD, of the Medical University of South Carolina (MUSC) in Charleston.
“The compounds we discovered have a lot of advantages, including high potency and good drug-like properties. We hope that those strengths translate into an effective new drug that can ultimately help patients.”
Dr. Dolloff and his colleagues reported their discovery in Leukemia.
The investigators detected the synergistic effects of E61 in combination with next-generation PIs, including carfilzomib, ixazomib, and oprozomib in both PI-sensitive and -resistant MM cell lines.
On the other hand, E61 had no effect on dexamethasone activity in dexamethasone-resistant cells. And E61 did not affect lenalidomide or doxorubicin cytotoxicity in PI-resistant cells.
The researchers also determined that E61 was only active in MM cells. E61 did not enhance the cytotoxic effects of PIs in normal cells.
This selective toxicity suggests that E61 may have “a wide therapeutic index in vivo,” the investigators wrote.
In vivo activity
To investigate the anti-MM activity and tolerability of E61 in vivo, the researchers treated a NOD-SCID IL2Rγ−/− mouse model with E61 at a continuous dose of 50 mg/kg/day.
E61 prolonged survival by 11 days in the treated mice (P=0.0007), and four of the 11 treated mice survived to the experiment’s end.
In another experiment, two of eight mice achieved a complete response.
After continuous dosing for 40 or more days, E61 was well tolerated, the investigators reported. The mice showed no overt signs of distress and did not lose weight.
Molecular target of E61
Using click (Cu(I)-catalyzed azide-alkyne cycloaddition) chemistry and a proteomics approach, the researchers then confirmed that PDI family members are the molecular target of E61.
Functional studies indicated that E61 inhibited PDI reductase activity in vitro. E61 also enhanced the accumulation of ubiquitinylated proteins and produced strong endoplasmic reticulum (ER) and oxidative stress responses when combined with PIs.
Anti-MM activity of E64FC26
The investigators used a structure activity relationship program to narrow the candidate molecules down to E64FC26.
E64FC26 demonstrated pan-inhibition in that it inhibited all members of the PDI family tested, including PDIA3, PDIA4, TXNDC5, and PDIA6.
E64FC26 also had greater in vitro potency against PDIA1 and the other PDI isoforms. It was the only compound to sensitize MM cells to PIs, with an average increase in PI sensitivity ranging from six- to seven-fold.
The researchers also noted that E64FC26 was superior to other PDI inhibitors they tested in activating ER stress.
The investigators tested the activity of E64FC26 in vivo using Vk*MYC transgenic mice, a model that closely resembles human MM.
Mice treated with E64FC26 had an immediate anti-MM response. Serum M-protein decreased in all mice by an average of 33 ± 7.9% (P=0.0135).
The investigators observed similar effects in a human xenotransplant MM model.
These mice were randomized to receive treatment with vehicle, E64FC26 (2 mg/kg for 3 days/week), bortezomib (0.25 mg/kg for 2 days/week), or a combination of the two agents.
E64FC26 increased median survival by 2 weeks compared with vehicle-treated mice (P<0.0001). By day 36, no vehicle-treated mouse survived, compared with 100% of the E64FC26-treated mice.
Single-agent bortezomib increased survival by 6 days (P=0.0007).
And the combination produced the greatest improvement in median survival, increasing it by 20 days (P<0.0001).
The investigators reported no overt toxicity or body weight fluctuation for mice treated with E64FC26 or the combination.
“These results provide preclinical proof of concept for the strategy of targeting PDI with this new class of compound for the treatment of MM,” the researchers concluded.
“One of the strengths of this study is that we spanned almost the entire drug discovery process,” Dr. Dolloff said. “We screened thousands of compounds, found an exciting molecule, deconvoluted what its binding target was, synthesized hundreds of derivatives to make it better, and then conducted animal studies.”
“The study has everything from biochemistry and cell biology to medicinal chemistry and animal pharmacology in it. There is still a lot of work to be done before this drug is ready for clinical trials in humans, but it has been a rewarding project, and I’m looking forward to the next steps.”
Dr. Dolloff is founder of Leukogene Therapeutics, Inc., which has licensed patents from MUSC, and a second study author is an inventor on patents. The other authors declared no conflicts of interest.
The research was supported by the National Institutes of Health/National Cancer Institute, the South Carolina Clinical & Translational Research Institute, the MUSC Hollings Cancer Center, and by the Hollings Cancer Center T32 Ruth L. Kirschstein National Research Service Award Training Program.
Researchers say they have identified a new class of protein disulfide isomerase (PDI) inhibitors that sensitize multiple myeloma (MM) cells to proteasome inhibitors (PIs).
The investigators screened approximately 20,000 compounds spanning multiple chemical libraries and found the compound E61 to be a “striking hit,” with a six-fold increase in bortezomib cytotoxicity and the ability to re-sensitize PI activity at low micromolar concentrations.
The researchers then synthesized and evaluated 150 E61 derivatives and discovered the lead candidate, E64FC26, which was highly synergistic with PIs at concentrations as low as 200 nM.
They reported that E64FC26 has “several advantages over previously reported PDI inhibitors, including superior potency and a pan-style mode of inhibition.”
“PDI is an attractive target in oncology, but good PDI inhibitors have been hard to find,” said Nathan G. Dolloff, PhD, of the Medical University of South Carolina (MUSC) in Charleston.
“The compounds we discovered have a lot of advantages, including high potency and good drug-like properties. We hope that those strengths translate into an effective new drug that can ultimately help patients.”
Dr. Dolloff and his colleagues reported their discovery in Leukemia.
The investigators detected the synergistic effects of E61 in combination with next-generation PIs, including carfilzomib, ixazomib, and oprozomib in both PI-sensitive and -resistant MM cell lines.
On the other hand, E61 had no effect on dexamethasone activity in dexamethasone-resistant cells. And E61 did not affect lenalidomide or doxorubicin cytotoxicity in PI-resistant cells.
The researchers also determined that E61 was only active in MM cells. E61 did not enhance the cytotoxic effects of PIs in normal cells.
This selective toxicity suggests that E61 may have “a wide therapeutic index in vivo,” the investigators wrote.
In vivo activity
To investigate the anti-MM activity and tolerability of E61 in vivo, the researchers treated a NOD-SCID IL2Rγ−/− mouse model with E61 at a continuous dose of 50 mg/kg/day.
E61 prolonged survival by 11 days in the treated mice (P=0.0007), and four of the 11 treated mice survived to the experiment’s end.
In another experiment, two of eight mice achieved a complete response.
After continuous dosing for 40 or more days, E61 was well tolerated, the investigators reported. The mice showed no overt signs of distress and did not lose weight.
Molecular target of E61
Using click (Cu(I)-catalyzed azide-alkyne cycloaddition) chemistry and a proteomics approach, the researchers then confirmed that PDI family members are the molecular target of E61.
Functional studies indicated that E61 inhibited PDI reductase activity in vitro. E61 also enhanced the accumulation of ubiquitinylated proteins and produced strong endoplasmic reticulum (ER) and oxidative stress responses when combined with PIs.
Anti-MM activity of E64FC26
The investigators used a structure activity relationship program to narrow the candidate molecules down to E64FC26.
E64FC26 demonstrated pan-inhibition in that it inhibited all members of the PDI family tested, including PDIA3, PDIA4, TXNDC5, and PDIA6.
E64FC26 also had greater in vitro potency against PDIA1 and the other PDI isoforms. It was the only compound to sensitize MM cells to PIs, with an average increase in PI sensitivity ranging from six- to seven-fold.
The researchers also noted that E64FC26 was superior to other PDI inhibitors they tested in activating ER stress.
The investigators tested the activity of E64FC26 in vivo using Vk*MYC transgenic mice, a model that closely resembles human MM.
Mice treated with E64FC26 had an immediate anti-MM response. Serum M-protein decreased in all mice by an average of 33 ± 7.9% (P=0.0135).
The investigators observed similar effects in a human xenotransplant MM model.
These mice were randomized to receive treatment with vehicle, E64FC26 (2 mg/kg for 3 days/week), bortezomib (0.25 mg/kg for 2 days/week), or a combination of the two agents.
E64FC26 increased median survival by 2 weeks compared with vehicle-treated mice (P<0.0001). By day 36, no vehicle-treated mouse survived, compared with 100% of the E64FC26-treated mice.
Single-agent bortezomib increased survival by 6 days (P=0.0007).
And the combination produced the greatest improvement in median survival, increasing it by 20 days (P<0.0001).
The investigators reported no overt toxicity or body weight fluctuation for mice treated with E64FC26 or the combination.
“These results provide preclinical proof of concept for the strategy of targeting PDI with this new class of compound for the treatment of MM,” the researchers concluded.
“One of the strengths of this study is that we spanned almost the entire drug discovery process,” Dr. Dolloff said. “We screened thousands of compounds, found an exciting molecule, deconvoluted what its binding target was, synthesized hundreds of derivatives to make it better, and then conducted animal studies.”
“The study has everything from biochemistry and cell biology to medicinal chemistry and animal pharmacology in it. There is still a lot of work to be done before this drug is ready for clinical trials in humans, but it has been a rewarding project, and I’m looking forward to the next steps.”
Dr. Dolloff is founder of Leukogene Therapeutics, Inc., which has licensed patents from MUSC, and a second study author is an inventor on patents. The other authors declared no conflicts of interest.
The research was supported by the National Institutes of Health/National Cancer Institute, the South Carolina Clinical & Translational Research Institute, the MUSC Hollings Cancer Center, and by the Hollings Cancer Center T32 Ruth L. Kirschstein National Research Service Award Training Program.