User login
PCI and CABG for left main disease have equal outcomes at 5 years
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: While PCI with drug-eluting stents has become more accepted as treatment for some patients with left main disease, long-term outcomes from randomized control trials comparing PCI with CABG have yet to be clearly established.
Study design: International, open-label, multicenter, randomized trial.
Setting: A total of 126 sites in 17 countries.
Synopsis: Patients with low or intermediate anatomical complexity with 70% visual stenosis of the left main coronary artery or 50%-70% stenosis by noninvasive testing were randomized to either PCI (948) or CABG (957). Dual-antiplatelet therapy was given to PCI patients and aspirin to CABG patients. At 5 years there was no significant difference in the rate of the composite of death, stroke, or myocardial infarction (22.0% with PCI vs. 19.2% with CABG; difference, 2.8 percentage points; 95% CI, –0.9 to 6.5; P = .13). This was consistent across subgroups.
There were numerical differences in nonpowered secondary outcomes that may represent effects but should be interpreted cautiously: ischemia-driven revascularization (16.9% with PCI vs. 10% with CABG), transient ischemic attack plus stroke (3.3% with PCI vs. 5.2% with CABG), and death from any cause (3% with PCI vs. 9.9% with CABG). There was no significant difference in cardiovascular events, MI, or stroke.
One interesting limitation was that patients who had PCI were more commonly on dual-antiplatelet therapy and angiotensin converting–enzyme inhibitors, whereas CABG patients were more often on beta-blockers, diuretics, anticoagulants, and antiarrhythmics.
Bottom line: PCI and CABG treatments for left main disease have no significant difference in the composite outcome of death, stroke, or MI at 5 years.
Citation: Stone GW et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
New guidelines on the diagnosis and treatment of adults with CAP
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: More than a decade has passed since the last CAP guidelines. Since then there have been new trials and epidemiological studies. There have also been changes to the process for guideline development. This guideline has moved away from the narrative style of guidelines to the GRADE format and PICO framework with hopes of answering specific questions by looking at the quality of evidence.
Study design: Multidisciplinary panel conducted pragmatic systemic reviews of high-quality studies.
Setting: The panel revised and built upon the 2007 guidelines, addressing 16 clinical questions to be used in immunocompetent patients with radiographic evidence of CAP in the United States with no recent foreign travel.
Synopsis: Changes from the 2007 guidelines are as follows: Sputum and blood cultures, previously recommended only in patients with severe CAP, are now also recommended for inpatients being empirically treated for Pseudomonas or methicillin-resistant Staphylococcus aureus (MRSA) and for those who have received IV antibiotics in the previous 90 days; use of procalcitonin is not recommended to decide whether to withhold antibiotics; steroids are not recommended unless being used for shock; HCAP categorization should be abandoned and need for empiric coverage of MRSA and Pseudomonas should be based on local epidemiology and local validated risk factors; B-lactam/macrolide is favored over fluoroquinolone for severe CAP therapy; and routine follow-up chest x-ray is not recommended.
Other recommendations include not routinely testing for urine pneumococcal or legionella antigens in nonsevere CAP; using PSI over CURB-65, in addition to clinical judgment, to determine need for inpatient care; using severe CAP criteria and clinical judgment for determining ICU need; not adding anaerobic coverage for aspiration pneumonia; and treating most cases of CAP that are clinically stable and uncomplicated for 5-7 days.
Bottom line: Given new data, updated recommendations have been made to help optimize CAP therapy.
Citation: Metlay JP et al. Diagnosis and treatment of adults with community-acquired pneumonia: An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-67.
Dr. Horton is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Multifaceted Intervention Reduces Cost
Healthcare costs continue to increase and are estimated to be approximately $3.1 trillion per year in the United States.[1] Waste is a major contributor to this cost, accounting for an estimated $910 billion/year.[2] Laboratory tests are well documented to contribute to healthcare waste, with an estimated 30% to 50% of tests for hospitalized patients being unnecessary.[3, 4, 5] This issue has been highlighted by the American Board of Internal Medicine Foundation's Choosing Wisely campaign as an area to reduce waste.[6] Evaluating this concern locally, a University Health Systems Consortium 2011 analysis indicated that the University of Utah general internal medicine hospitalist service had a higher average direct lab cost per discharge compared to top performers, indicating an opportunity for improvement.
Multiple interventions have been described in the literature to address excessive laboratory utilization, including physician education, audit and feedback, cost information display, and administrative rules restricting certain types of ordering.[7, 8, 9, 10, 11] Despite these interventions, barriers remain common and not all interventions are sustained. For example, interventions focused mainly on education see a small improvement initially that is not sustained.[4, 12, 13] Additionally, although most studies focus on individual interventions, those that target multiple factors have been found to be more successful at producing and sustaining change.[14] Therefore, the opportunity existed to incorporate multiple etiologies into a single intervention and apply a checklist to laboratory ordering to see if combined modalities could be effective at reducing laboratory costs in a sustainable manner.
In addition to cost, there is potential patient harm resulting from unnecessary laboratory testing. For prolonged hospitalizations, anemia is a well‐recognized side effect of phlebotomy,[15, 16] and a recent evaluation of cardiac surgery patients found an average cumulative blood loss due to phlebotomy of 454 mL/hospital stay.[17] The sheer number of tests ordered can lead to false positive tests that result in additional testing and monitoring. Furthermore, patients subjected to laboratory blood draws are often awakened early in the morning, which is unpleasant and could adversely affect the patient experience.
Recognizing laboratory cost as a problem, the University of Utah general internal medicine hospitalist service implemented a multifaceted quality‐improvement initiative with a goal to reduce laboratory testing. At the time of this project, University of Utah Health Care (UUHC) developed a Value Driven Outcomes (VDO) tool to give direct data related to costs of care, including the actual cost paid by the hospital to the university‐owned laboratory vendor (ARUP Laboratories, Salt Lake City, UT) for testing.[18] The hospitalist group incorporated VDO into the initiative for routine cost feedback. This study evaluates the impact of this intervention on laboratory costs.
METHODS
Design
A retrospective, controlled, interrupted time series (ITS) study was performed to compare changes in lab costs between hospitalists (intervention study group) and other providers (control study group). The intervention initiation date was February 1, 2013. The baseline period was July 1, 2012 to January 31, 2013, as that was the period in which the VDO tool became available for cost analysis prior to intervention. The intervention period was February 1, 2013 to April 30, 2014, as there was a change in the electronic health record (EHR) in May 2014 that affected data flow and could act as a major confounder. The institutional review board classified this project as quality improvement and did not require review and oversight.
Setting
UUHC is a 500‐bed academic medical center in Salt Lake City, Utah. The hospitalist service is a teaching service composed of 4 teams with internal medicine residents and medical students. The nonhospitalist services include all surgical services, as well as pulmonary, cardiology, hematology, and oncology services on which internal medicine residents rotate. All services at UUHC are staffed by academic physicians affiliated with the University of Utah School of Medicine.
Population
All patients 18 years and older admitted to the hospital to a service other than obstetrics, rehabilitation, or psychiatry between July 1, 2012 and April 30, 2014 were evaluated. Patients with missing data for outcomes or covariates were excluded.
Intervention
Initial evaluation included an informal review of patient charts and discussion with hospitalist group members, both indicating laboratory overuse. A working group was then established including hospitalists and process engineers to evaluate the workflow by which laboratory tests were ordered. Concurrently, a literature review was performed to help identify the scope of the problem and evaluate methods that had been successful at other institutions. Through this review, it was noted that interns were the most frequent orderers of tests and the largest contributors to variation of testing for inpatients.[19] Two specific studies with direct applicability to this project demonstrated that discussion of costs with attendings in a trauma intensive care unit resulted in a 30% reduction of tests ordered,[20] and discussion of testing with a senior resident in an internal medicine inpatient setting demonstrated a 20% reduction in laboratory testing.[21]
Our laboratory reduction intervention expanded on the current literature to incorporate education, process change, cost feedback, and financial incentives. Specifically, starting February 1, 2013, the following interventions were performed:
- Education of all providers involved, including the hospitalist group and all internal medicine residents at the start of their rotation with the hospitalist service. Education included a 30‐minute discussion of laboratory overuse, costs associated with laboratory overuse, previous interventions and their success, and current intervention with goals. Each resident was provided a pocket card with the most common lab tests and associated charges. Charges were used instead of costs due to concerns regarding the possible public dissemination of institutional costs.
- Standardization of the rounding process including a checklist review (see Supporting Information, Appendix, in the online version of this article) for all patients that ensured discussion of labs, telemetry, pain, lines/tubes, nursing presence, and follow‐up needed. The expectation was that all plans for lab testing would be discussed during rounds. The third‐year medical student was responsible to ensure that all items were covered daily on each patient.
- Monthly feedback at the hospitalist group meeting regarding laboratory costs using the VDO tool. Data were presented as a monthly group average and compared to preintervention baseline costs. Individual performance could be viewed and compared to other providers within the group.
- Financial incentive through a program that shares 50% of cost savings realized by the hospital with the Division of General Internal Medicine. The incentive could be used to support future quality‐improvement projects, but there was no individual physician incentive.
Data Collection and Preparation
Clinical data were collected in the inpatient EHR (Cerner Corp., Kansas City, MO) and later imported into the enterprise data warehouse (EDW) as part of the normal data flow. Billing data were imported into the EDW from the billing system. Cost data were estimated using the VDO tool developed by the University of Utah to identify clinical costs to the UUHC system.[18]
Clinical and Cost Outcomes
We hypothesized that following the intervention, the number of tests and lab costs would decrease greater for patients in the intervention group than in the control group, with no adverse effect on length of stay (LOS) or 30‐day readmissions.
Lab cost per day was calculated as the total lab cost per visit divided by the LOS. We adjusted all lab costs to 2013 US dollars using Consumer Price Index inflation data.[22] To account for different LOS, we used LOS as a weight variable when estimating descriptive characteristics and P values for lab cost per day and the number of tests. Thirty‐day readmissions included inpatient encounters followed by another inpatient encounter within 30 days excluding obstetrics, rehabilitation, and psychiatry visits.
Descriptive Variables
We included information on age at admission in years and Charlson Comorbidity Index (CCI) to evaluate differences in control and intervention groups.[23]
Statistical Analysis
First, unadjusted descriptive statistics were calculated for study outcomes and visit characteristics. Descriptive statistics were expressed as n (%) and mean standard deviation. Simple comparisons were performed based on 2 tests of homogeneity for categorical variables and on t tests for continuous variables.
Second, an ITS analysis was conducted to evaluate the impact of the intervention while accounting for baseline trends.[24] In this analysis, the dependent variable (yt) was the difference in aggregated outcome measures between the intervention and control groups every 2 weeks (eg, difference in average lab costs in a given 2‐week period between the 2 groups). Intervention impact was then evaluated in terms of changes in the level of the outcome (b2) as well as in the trend over time (b3) compared to the initial difference in means (b0) and baseline trend (b1). The following difference‐in‐differences segmented regression model was fitted using the autoreg procedure in SAS: yt = b0 + b1*timet + b2*study periodt + b3*time after the interventiont + errort, where timet is biweekly intervals after the beginning of the study, time after the interventiont is biweekly intervals after the intervention date, and study periodt is 1 postintervention and 0 preintervention. The models were fitted using maximum likelihood and stepwise autoregression to test 24 lags.
P values <0.05 were considered significant. SAS (version 9.3; SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
We analyzed 48,327 inpatient visits that met inclusion criteria. We excluded 15,659 obstetrics, rehabilitation, and psychiatry visits. Seven hundred seventy‐two (2.4%) of the remaining visits were excluded due to missing data. A total of 31,896 inpatient visits by 22,545 patients were included in the analysis. There were 10,136 visits before the intervention and 21,760 visits after. Characteristics of the study groups for the full study timeframe (July 1, 2012April 30, 2014) are summarized in Table 1.
| Characteristic | Study Group* | |||
|---|---|---|---|---|
| Overall, N = 31,896 | Control, N = 25,586 | Intervention, N = 6,310 | P Value | |
| ||||
| Patient characteristics | ||||
| Age, y | 55.47 17.61 | 55.27 17.13 | 56.30 19.39 | <0.001 |
| Female gender | 14,995 (47%) | 11,753 (46%) | 3,242 (51%) | <0.001 |
| CCI | 3.73 3.25 | 3.61 3.17 | 4.20 3.54 | <0.001 |
| Outcomes | ||||
| Cost per day, $ | 130.95 392.16 | 131.57 423.94 | 127.68 220.40 | 0.022 |
| Cost per visit, $ | 733.75 1,693.98 | 772.30 1,847.65 | 577.40 795.29 | <0.001 |
| BMP tests per day | 0.73 1.17 | 0.74 1.19 | 0.67 1.05 | <0.001 |
| CMP tests per day | 0.20 0.67 | 0.19 0.68 | 0.26 0.62 | <0.001 |
| CBC tests per day | 0.83 1.10 | 0.84 1.15 | 0.73 0.82 | <0.001 |
| PT/INR tests per day | 0.36 1.03 | 0.36 1.07 | 0.34 0.83 | <.001 |
| LOS, d | 5.60 7.12 | 5.87 7.55 | 4.52 4.82 | <0.001 |
| 30‐day readmissions | 4,374 (14%) | 3,603 (14%) | 771 (12%) | <0.001 |
During the study period, there were 25,586 visits in the control group and 6310 visits in the intervention group. Patients in the intervention group were on average older than patients in the control group. There were more female patients in the intervention group. Mean CCI was 4.2 in the intervention group and 3.6 in the control group. The intervention group had lower LOS and 30‐day readmissions than the control group.
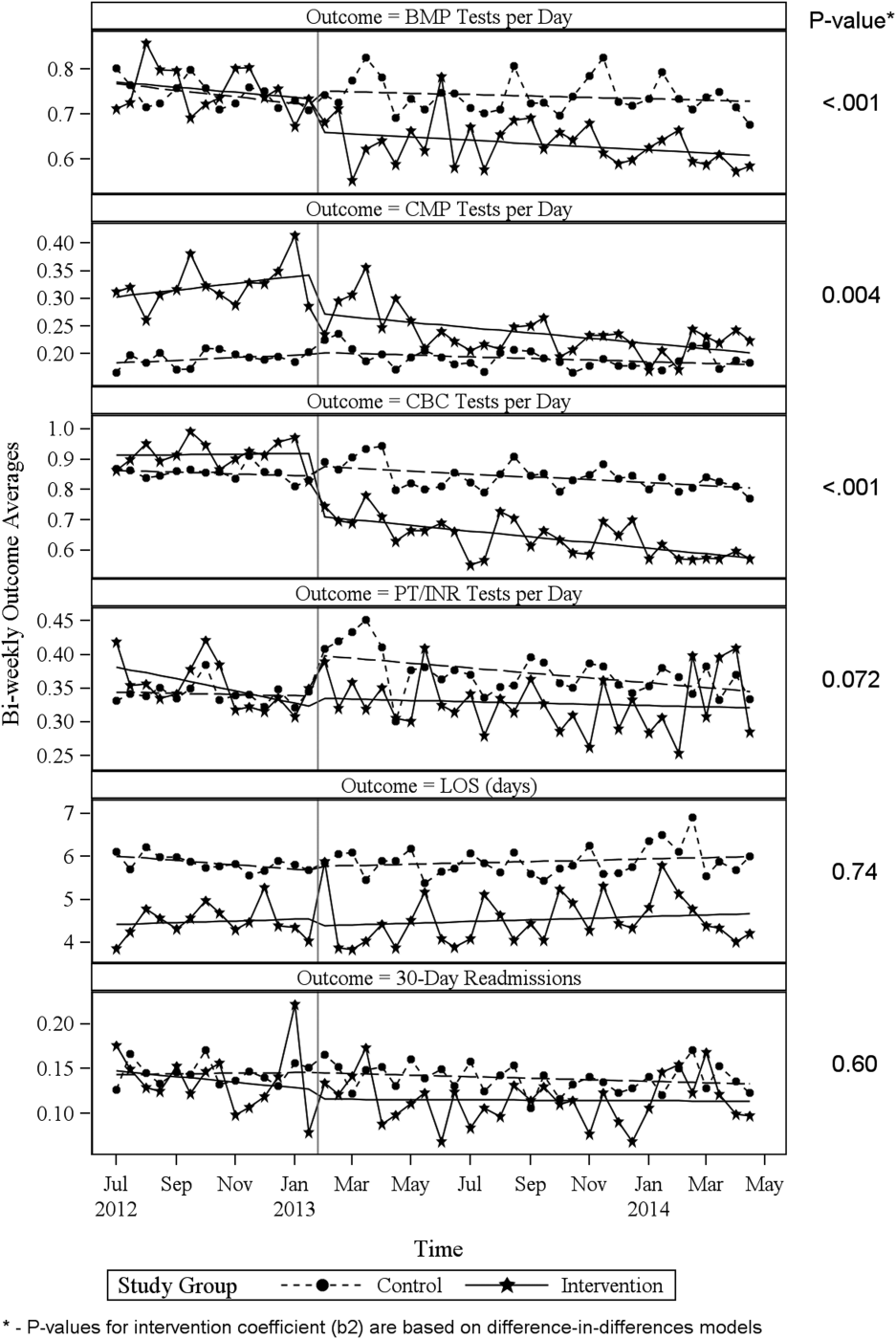
Descriptive statistics and simple comparisons of covariates and outcomes before and after the intervention are shown in Table 2. Age and gender distributions remained unchanged in both groups. CCI increased in the control group by 0.24 (P < 0.001) and remained unchanged in the intervention group. In the intervention group, lab cost per day was reduced from $138 before the intervention to $123 after the intervention (P < 0.001). In contrast, among control patients, cost per day increased nonsignificantly from $130 preintervention to $132 postintervention (P = 0.37). Number of tests per day significantly decreased for all specific tests in the intervention group. Readmission rates decreased significantly from 14% to 11% in the intervention group (P = 0.01). LOS remained constant in both groups.
| Characteristic* | Control | Intervention | ||||
|---|---|---|---|---|---|---|
| Preintervention, N = 8,102 | Postintervention, N = 17,484 | P Value | Preintervention, N = 2,034 | Postintervention, N = 4,276 | P Value | |
| ||||||
| Patient characteristics | ||||||
| Age, yr | 55.17 17.46 | 55.31 16.98 | 0.55 | 55.90 19.47 | 56.50 19.35 | 0.25 |
| Female gender | 3,707 (46%) | 8,046 (46%) | 0.69 | 1,039 (51%) | 2,203 (52%) | 0.74 |
| CCI | 3.45 3.06 | 3.69 3.21 | <0.001 | 4.19 3.51 | 4.20 3.56 | 0.89 |
| Outcomes | ||||||
| Cost per day, $ | 130.1 431.8 | 132.2 420.3 | 0.37 | 137.9 232.9 | 122.9 213.5 | <0.001 |
| Cost per visit, $ | 760.4 1,813.6 | 777.8 1,863.3 | 0.48 | 617.8 844.1 | 558.2 770.3 | 0.005 |
| BMP tests per day | 0.74 1.21 | 0.74 1.18 | 0.67 | 0.75 1.03 | 0.63 1.05 | <0.001 |
| CMP tests per day | 0.19 0.68 | 0.19 0.68 | 0.85 | 0.32 0.68 | 0.23 0.58 | <0.001 |
| CBC tests per day | 0.85 1.14 | 0.84 1.15 | 0.045 | 0.92 0.79 | 0.64 0.76 | <0.001 |
| PT/INR tests per day | 0.34 1.04 | 0.37 1.08 | <0.001 | 0.35 0.82 | 0.33 0.84 | 0.020 |
| LOS, d | 5.84 7.66 | 5.88 7.50 | 0.71 | 4.48 5.12 | 4.54 4.67 | 0.63 |
| 30‐day readmissions | 1,173 (14%) | 2,430 (14%) | 0.22 | 280 (14%) | 491 (11%) | 0.010 |
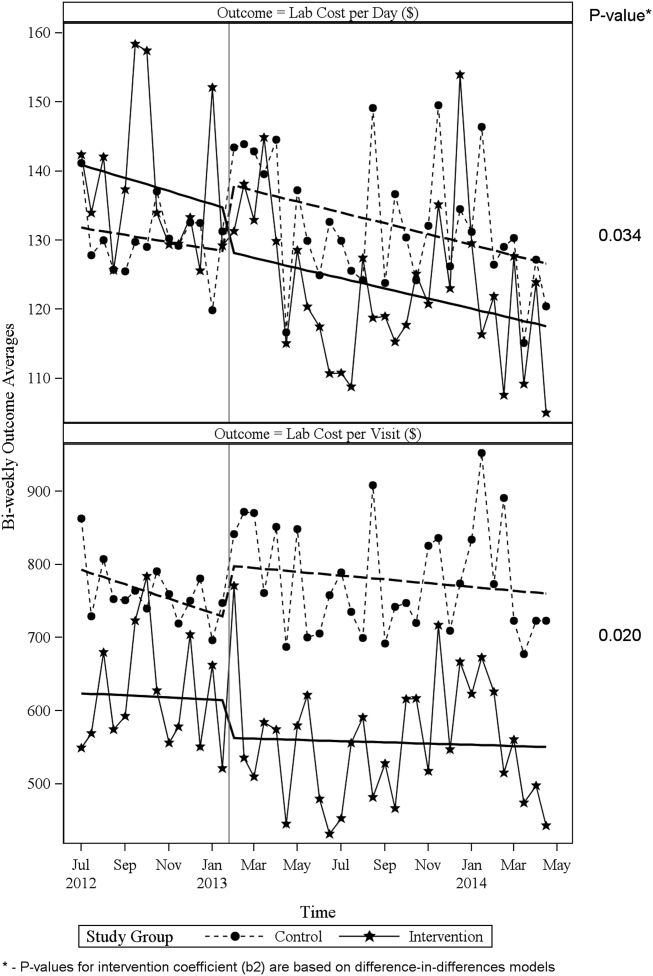
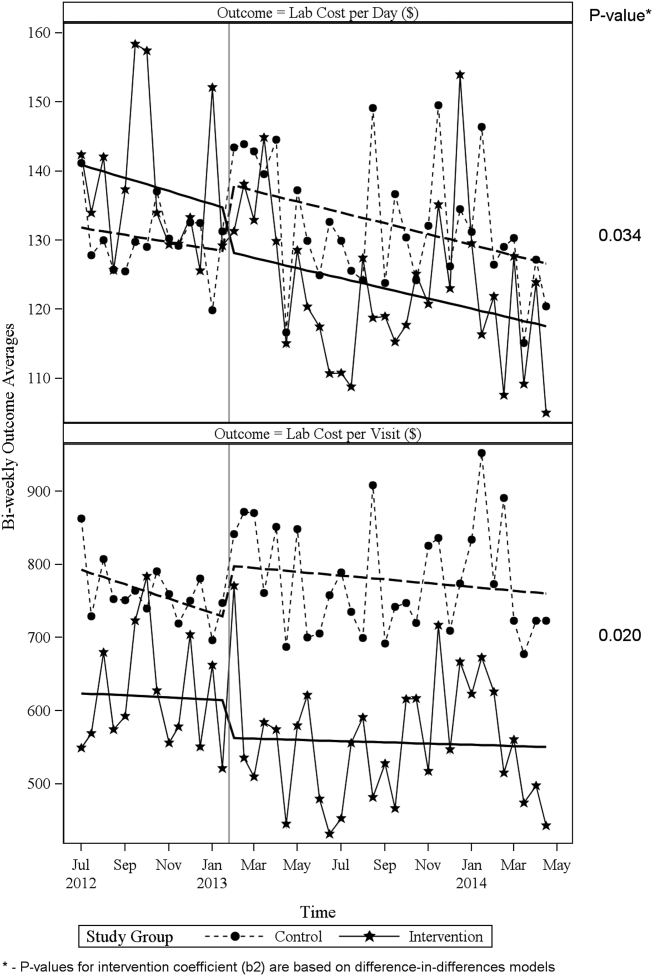
ITS analysis results are shown in Table 3. After the intervention, the difference in monthly means between the 2 groups dropped by $16 for cost per day (P = 0.034) and by $128 for cost per visit (P = 0.02). The decreased cost in the intervention group amounts to approximately $251,427 (95% confidence interval [CI]: $20,370‐$482,484) savings over the first year. If the intervention was rolled out for the control group and had a similar impact, it could have led to an additional cost savings of $1,321,669 (95% CI: 107,081‐2,536,256). Moreover, the number of basic metabolic panel, comprehensive metabolic panel, and complete blood count test per day were reduced significantly more in the intervention group compared to the control group (<0.001, 0.004, and <0.001).
| Outcome | Parameter* | Parameter Estimate | Standard Error | t Value | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| Lab cost per day ($) | Baseline difference level (b0) | 9.3450 | 6.5640 | 1.4237 | 0.16 |
| Baseline difference trend (b1) | 0.2150 | 0.7709 | 0.2789 | 0.78 | |
| Change in difference level after intervention(b2) | 16.1200 | 7.3297 | 2.1993 | 0.034 | |
| Change in difference trend after intervention (b3) | 0.2388 | 0.8090 | 0.2952 | 0.77 | |
| Lab cost per visit ($) | Baseline difference level (b0) | 166.081 | 48.3425 | 3.4355 | 0.001 |
| Baseline difference trend (b1) | 3.6663 | 5.8571 | 0.6260 | 0.53 | |
| Change in difference level after intervention(b2) | 128.527 | 53.0278 | 2.4238 | 0.020 | |
| Change in difference trend after intervention (b3) | 2.2586 | 5.8463 | 0.3863 | 0.70 | |
| BMP tests per day | Baseline difference level (b0) | 0.0061 | 0.0250 | 0.2439 | 0.81 |
| Baseline difference trend (b1) | 0.0004 | 0.0030 | 0.1449 | 0.89 | |
| Change in difference level after intervention(b2) | 0.1034 | 0.0276 | 3.7426 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0014 | 0.0030 | 0.4588 | 0.65 | |
| CMP tests per day | Baseline difference level (b0) | 0.1226 | 0.0226 | 5.4302 | <0.001 |
| Baseline difference trend (b1) | 0.0015 | 0.0028 | 0.5539 | 0.58 | |
| Change in difference level after intervention(b2) | 0.0754 | 0.0248 | 3.0397 | 0.004 | |
| Change in difference trend after intervention (b3) | 0.0030 | 0.0028 | 1.0937 | 0.28 | |
| CBC tests per day | Baseline difference level (b0) | 0.0539 | 0.0190 | 2.8338 | 0.007 |
| Baseline difference trend (b1) | 0.0013 | 0.0023 | 0.5594 | 0.58 | |
| Change in difference level after intervention(b2) | 0.2343 | 0.0213 | 10.997 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0036 | 0.0023 | 1.5539 | 0.13 | |
| PT/INR tests per day | Baseline difference level (b0) | 0.0413 | 0.0242 | 1.7063 | 0.096 |
| Baseline difference trend (b1) | 0.0040 | 0.0028 | 1.4095 | 0.17 | |
| Change in difference level after intervention(b2) | 0.0500 | 0.0270 | 1.8507 | 0.072 | |
| Change in difference trend after intervention (b3) | 0.0054 | 0.0030 | 1.7940 | 0.080 | |
| LOS, d | Baseline difference level (b0) | 1.4211 | 0.2746 | 5.1743 | <0.001 |
| Baseline difference trend (b1) | 0.0093 | 0.0333 | 0.2807 | 0.78 | |
| Change in difference level after intervention(b2) | 0.1007 | 0.2988 | 0.3368 | 0.74 | |
| Change in difference trend after intervention (b3) | 0.0053 | 0.0331 | 0.1588 | 0.87 | |
| 30‐day readmissions | Baseline difference level (b0) | 0.0057 | 0.0185 | 0.3084 | 0.76 |
| Baseline difference trend (b1) | 0.0017 | 0.0022 | 0.8016 | 0.43 | |
| Change in difference level after intervention(b2) | 0.0110 | 0.0206 | 0.5315 | 0.60 | |
| Change in difference trend after intervention (b3) | 0.0021 | 0.0023 | 0.9111 | 0.37 | |
Figure 1 shows a graphical representation of the biweekly means for the 2 primary outcomeslab cost per day and lab cost per visit. Figure 2 shows all other outcomes. To the right of each figure, P values are provided for the b2 coefficients from Table 3.
DISCUSSION
Through a multifaceted quality‐improvement initiative, the UUHC hospitalist group was able to reduce lab cost per day and per visit as well as commonly ordered routine labs as compared to an institutional control group. A multifaceted approach was selected given the literature supporting this approach as the most likely method to sustain improvement.[14] At the same time, the use of a multifaceted intervention makes it difficult to rigorously determine the relative impact of different components of the intervention. In discussing this issue, however, the hospitalist group felt that the driving factors for change were those related to process change, specifically, the use of a standardized rounding checklist to discuss lab testing and the routine review of lab costs at group meetings. The ultimate goal was to change the culture of routine test ordering into a thoughtful process of needed tests and thereby reduce costs. Prior to this intervention, the least experienced person on this team (the intern) ordered any test he or she wanted, usually without discussion. The intervention focused on this issue through standardized supervision and explicit discussion of laboratory tests. Importantly, although improvements from education initiatives typically decrease over time, the incorporation of process change in this intervention was felt to likely contribute to the sustained reduction seen at 15 months. Although use of the rounding checklist added another step to daily rounds, the routine cost feedback, including comparisons to peers, helped encourage use of the checklist. Thus, we feel that routine feedback was essential to sustaining the intervention and its impact.
Inappropriate and unnecessary testing has been recognized for decades, and multiple interventions have been attempted, including a recent article that demonstrated a 10% reduction in common laboratory ordering through an initiative mainly focused on education and ordering feedback.[25] Despite reported success of several interventions, none have combined multiple interventions and explicitly required discussion of laboratory tests on rounds. For example, although the UUHC intervention used Attali et al.[21] and Barie and Hydo's[20] work to develop the intervention, neither of these studies described how laboratory testing was discussed with the attending or supervising resident. The UUHC intervention thus builds on the current literature by combining other successful modalities with explicit discussion of laboratory testing via a rounding checklist and feedback with the novel VDO tool to reduce laboratory costs. A major strength of this intervention is the relatively low cost and the generalizability of implementing rounding checklists. Initial support from the hospital was needed to provide accurate VDO information to the hospitalist group. However, ongoing costs were minimal and related to any additional time spent during rounds to discuss laboratory tests. Thus, we feel that this intervention is feasible for wide replication.
Another strength of the study is the use of the VDO tool to measure actual costs. Whereas previous studies have relied on estimated costs with extrapolation to potential cost savings, this study used direct costs to the institution as a more accurate marker of cost savings. Additionally, most studies on lab utilization have used a before/after analysis without a control group. The presence of a control group for this analysis is important to help assess for institutional trends that may not be reflected in a before/after intervention. The reduction in cost in the intervention group despite a trend toward increased cost in the institutional control group supports the impact of this intervention.
Limitations of this study include that it was a single‐center, controlled ITS study and not a randomized controlled trial. Related to this limitation, the control group reflected a different patient population compared to the intervention group, with a longer LOS, lower CCI, and inclusion of nonmedical patients. However, these differences were relatively stable before and after the intervention. Also, ITS is considered one of the most robust research designs outside of randomized controlled trials, and it accounts for baseline differences in both levels and trends.[24] Nevertheless, it remains possible that secular trends existed that we did not capture and that affected the 2 populations differently.
A further limitation is that the baseline period was only 7 months and the intervention was 15 months. As the 7 months started in July, this could have reflected the time when interns were least experienced with ordering. Unfortunately, we did not have VDO availability for a full year prior to the intervention. We believe that any major effect due to this shortened baseline period should have been seen in the control group as well, and therefore accounted for in the analysis. Additionally, it is possible that there was spillover of the intervention to the control group, as internal medicine residents rotated throughout the hospital to other medical services (pulmonary, cardiology, hematology, and oncology). However, any effect of their rotation should have been to lower the control lab cost, thus making differences less profound.
CONCLUSIONS
A multifaceted approach to laboratory reduction through education, process change, cost feedback, and financial incentive resulted in a significant reduction in laboratory cost per day, laboratory cost per visit, and the ordering of common laboratory tests at a major academic medical center.
Acknowledgements
The authors thank Mr. Michael Swanicke for his assistance in process engineering, Mr. Tony Clawson for his routine provision of VDO data, and Ms. Selma Lopez for her editorial support.
Disclosures: K.K. is or has been a consultant on clinical decision support (CDS) or electronic clinical quality measurement to the US Office of the National Coordinator for Health IT, ARUP Laboratories, McKesson InterQual, ESAC, Inc., JBS International, Inc., Inflexxion, Inc., Intelligent Automation, Inc., Partners HealthCare, Mayo Clinic, and the RAND Corporation. K.K. receives royalties for a Duke University‐owned CDS technology for infectious disease management known as CustomID that he helped develop. K.K. was formerly a consultant for Religent, Inc. and a co‐owner and consultant for Clinica Software, Inc., both of which provide commercial CDS services, including through use of a CDS technology known as SEBASTIAN that K.K. developed. K.K. no longer has a financial relationship with either Religent or Clinica Software. K.K. has no competing interest with any specific product or intervention evaluated in this article. All other authors declare no competing interests.
- , , , et al. National health expenditure projections, 2014–24: spending growth faster than recent trends. Health Aff (Millwood). 2015;34(8):1407–1417.
- , . Eliminating waste in US health care. JAMA. 2012;307(14):1513–1516.
- , , , et al. Utilization of arterial blood gas measurements in a large tertiary care hospital. Am J Clin Pathol. 2007;127:604–609.
- , . Strategies to promote rational clinical chemistry test utilization. Clin Biochem. 1996;29:291–299.
- , , , , . The landscape of inappropriate laboratory testing: a 15‐year meta‐analysis. PLoS One. 2013;8:e78962.
- ABIM Choosing Wisely Society of Hospital Medicine–Adult Hospital Medicine. Five things physicians and patients should question. Available at: http://www.choosingwisely.org/societies/society‐of‐hospital‐medicine‐adult. Published February 21, 2013. Accessed September 2, 2015.
- , , , , , . Effect of daily charge feedback on inpatient charges and physician knowledge and behavior. Arch Intern Med. 1989;149:426–429.
- , , , et al. A utilization management intervention to reduce unnecessary testing in the coronary care unit. Arch Intern Med. 2002;162:1885–1890.
- , , , et al. The impact of peer management on test‐ordering behavior. Ann Intern Med. 2004;141:196–204.
- , , , , . An administrative intervention to improve the utilization of laboratory tests within a university hospital. Int J Qual Health Care. 2005;17:243–248.
- , , , et al. Impact of providing fee data on laboratory test ordering. JAMA Intern Med. 2013;173(10):903–908.
- , , , et al. The failure of physician education as a cost containment strategy. JAMA. 1984;252:225–230.
- . Is that lab test necessary? Am J Clin Pathol. 2006;126:335–336.
- , , , . Techniques to improve physicians' use of diagnostic tests. JAMA. 1998;280:2020–2027.
- , , . Laboratory testing in the intensive care unit. Crit Care Clin. 2007;23:435–465.
- . Complications of critical care: lab testing and iatrogenic anemia. MLO Med Lab Obs. 200;33(10):28–31.
- , , , et al. Contemporary bloodletting in cardiac surgical care. Ann Thorac Surg. 2015;99:779–785.
- , , , et al. Value Driven Outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015:22:223–235.
- , , . The impact of residents, interns, and attendings on inpatient laboratory ordering patterns: a report from one university's hospitalist service. Acad Med. 2011;86:139–145.
- , . Learning to not know: results of a program for ancillary cost reduction in surgical care. J Trauma. 1996;41:714–720.
- , , , et al. A cost‐effective method for reducing the volume of laboratory tests in a university‐associated teaching hospital. Mt Sinai J Med. 2006;73:787–794.
- US Bureau of Labor Statistics. CPI inflation calculator. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed May 22, 2015.
- , , , et al. Coding algorithms for defining comorbidities in ICD‐9‐CM and ICD‐10 administrative data. Med Care. 2005;43:113–1139.
- , , , . Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther. 2002;27(4):299–309.
- , , , et al. A multifaceted hospitalist quality improvement intervention: decreased frequency of common labs. J Hosp Med. 2015;10:390–395.
Healthcare costs continue to increase and are estimated to be approximately $3.1 trillion per year in the United States.[1] Waste is a major contributor to this cost, accounting for an estimated $910 billion/year.[2] Laboratory tests are well documented to contribute to healthcare waste, with an estimated 30% to 50% of tests for hospitalized patients being unnecessary.[3, 4, 5] This issue has been highlighted by the American Board of Internal Medicine Foundation's Choosing Wisely campaign as an area to reduce waste.[6] Evaluating this concern locally, a University Health Systems Consortium 2011 analysis indicated that the University of Utah general internal medicine hospitalist service had a higher average direct lab cost per discharge compared to top performers, indicating an opportunity for improvement.
Multiple interventions have been described in the literature to address excessive laboratory utilization, including physician education, audit and feedback, cost information display, and administrative rules restricting certain types of ordering.[7, 8, 9, 10, 11] Despite these interventions, barriers remain common and not all interventions are sustained. For example, interventions focused mainly on education see a small improvement initially that is not sustained.[4, 12, 13] Additionally, although most studies focus on individual interventions, those that target multiple factors have been found to be more successful at producing and sustaining change.[14] Therefore, the opportunity existed to incorporate multiple etiologies into a single intervention and apply a checklist to laboratory ordering to see if combined modalities could be effective at reducing laboratory costs in a sustainable manner.
In addition to cost, there is potential patient harm resulting from unnecessary laboratory testing. For prolonged hospitalizations, anemia is a well‐recognized side effect of phlebotomy,[15, 16] and a recent evaluation of cardiac surgery patients found an average cumulative blood loss due to phlebotomy of 454 mL/hospital stay.[17] The sheer number of tests ordered can lead to false positive tests that result in additional testing and monitoring. Furthermore, patients subjected to laboratory blood draws are often awakened early in the morning, which is unpleasant and could adversely affect the patient experience.
Recognizing laboratory cost as a problem, the University of Utah general internal medicine hospitalist service implemented a multifaceted quality‐improvement initiative with a goal to reduce laboratory testing. At the time of this project, University of Utah Health Care (UUHC) developed a Value Driven Outcomes (VDO) tool to give direct data related to costs of care, including the actual cost paid by the hospital to the university‐owned laboratory vendor (ARUP Laboratories, Salt Lake City, UT) for testing.[18] The hospitalist group incorporated VDO into the initiative for routine cost feedback. This study evaluates the impact of this intervention on laboratory costs.
METHODS
Design
A retrospective, controlled, interrupted time series (ITS) study was performed to compare changes in lab costs between hospitalists (intervention study group) and other providers (control study group). The intervention initiation date was February 1, 2013. The baseline period was July 1, 2012 to January 31, 2013, as that was the period in which the VDO tool became available for cost analysis prior to intervention. The intervention period was February 1, 2013 to April 30, 2014, as there was a change in the electronic health record (EHR) in May 2014 that affected data flow and could act as a major confounder. The institutional review board classified this project as quality improvement and did not require review and oversight.
Setting
UUHC is a 500‐bed academic medical center in Salt Lake City, Utah. The hospitalist service is a teaching service composed of 4 teams with internal medicine residents and medical students. The nonhospitalist services include all surgical services, as well as pulmonary, cardiology, hematology, and oncology services on which internal medicine residents rotate. All services at UUHC are staffed by academic physicians affiliated with the University of Utah School of Medicine.
Population
All patients 18 years and older admitted to the hospital to a service other than obstetrics, rehabilitation, or psychiatry between July 1, 2012 and April 30, 2014 were evaluated. Patients with missing data for outcomes or covariates were excluded.
Intervention
Initial evaluation included an informal review of patient charts and discussion with hospitalist group members, both indicating laboratory overuse. A working group was then established including hospitalists and process engineers to evaluate the workflow by which laboratory tests were ordered. Concurrently, a literature review was performed to help identify the scope of the problem and evaluate methods that had been successful at other institutions. Through this review, it was noted that interns were the most frequent orderers of tests and the largest contributors to variation of testing for inpatients.[19] Two specific studies with direct applicability to this project demonstrated that discussion of costs with attendings in a trauma intensive care unit resulted in a 30% reduction of tests ordered,[20] and discussion of testing with a senior resident in an internal medicine inpatient setting demonstrated a 20% reduction in laboratory testing.[21]
Our laboratory reduction intervention expanded on the current literature to incorporate education, process change, cost feedback, and financial incentives. Specifically, starting February 1, 2013, the following interventions were performed:
- Education of all providers involved, including the hospitalist group and all internal medicine residents at the start of their rotation with the hospitalist service. Education included a 30‐minute discussion of laboratory overuse, costs associated with laboratory overuse, previous interventions and their success, and current intervention with goals. Each resident was provided a pocket card with the most common lab tests and associated charges. Charges were used instead of costs due to concerns regarding the possible public dissemination of institutional costs.
- Standardization of the rounding process including a checklist review (see Supporting Information, Appendix, in the online version of this article) for all patients that ensured discussion of labs, telemetry, pain, lines/tubes, nursing presence, and follow‐up needed. The expectation was that all plans for lab testing would be discussed during rounds. The third‐year medical student was responsible to ensure that all items were covered daily on each patient.
- Monthly feedback at the hospitalist group meeting regarding laboratory costs using the VDO tool. Data were presented as a monthly group average and compared to preintervention baseline costs. Individual performance could be viewed and compared to other providers within the group.
- Financial incentive through a program that shares 50% of cost savings realized by the hospital with the Division of General Internal Medicine. The incentive could be used to support future quality‐improvement projects, but there was no individual physician incentive.
Data Collection and Preparation
Clinical data were collected in the inpatient EHR (Cerner Corp., Kansas City, MO) and later imported into the enterprise data warehouse (EDW) as part of the normal data flow. Billing data were imported into the EDW from the billing system. Cost data were estimated using the VDO tool developed by the University of Utah to identify clinical costs to the UUHC system.[18]
Clinical and Cost Outcomes
We hypothesized that following the intervention, the number of tests and lab costs would decrease greater for patients in the intervention group than in the control group, with no adverse effect on length of stay (LOS) or 30‐day readmissions.
Lab cost per day was calculated as the total lab cost per visit divided by the LOS. We adjusted all lab costs to 2013 US dollars using Consumer Price Index inflation data.[22] To account for different LOS, we used LOS as a weight variable when estimating descriptive characteristics and P values for lab cost per day and the number of tests. Thirty‐day readmissions included inpatient encounters followed by another inpatient encounter within 30 days excluding obstetrics, rehabilitation, and psychiatry visits.
Descriptive Variables
We included information on age at admission in years and Charlson Comorbidity Index (CCI) to evaluate differences in control and intervention groups.[23]
Statistical Analysis
First, unadjusted descriptive statistics were calculated for study outcomes and visit characteristics. Descriptive statistics were expressed as n (%) and mean standard deviation. Simple comparisons were performed based on 2 tests of homogeneity for categorical variables and on t tests for continuous variables.
Second, an ITS analysis was conducted to evaluate the impact of the intervention while accounting for baseline trends.[24] In this analysis, the dependent variable (yt) was the difference in aggregated outcome measures between the intervention and control groups every 2 weeks (eg, difference in average lab costs in a given 2‐week period between the 2 groups). Intervention impact was then evaluated in terms of changes in the level of the outcome (b2) as well as in the trend over time (b3) compared to the initial difference in means (b0) and baseline trend (b1). The following difference‐in‐differences segmented regression model was fitted using the autoreg procedure in SAS: yt = b0 + b1*timet + b2*study periodt + b3*time after the interventiont + errort, where timet is biweekly intervals after the beginning of the study, time after the interventiont is biweekly intervals after the intervention date, and study periodt is 1 postintervention and 0 preintervention. The models were fitted using maximum likelihood and stepwise autoregression to test 24 lags.
P values <0.05 were considered significant. SAS (version 9.3; SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
We analyzed 48,327 inpatient visits that met inclusion criteria. We excluded 15,659 obstetrics, rehabilitation, and psychiatry visits. Seven hundred seventy‐two (2.4%) of the remaining visits were excluded due to missing data. A total of 31,896 inpatient visits by 22,545 patients were included in the analysis. There were 10,136 visits before the intervention and 21,760 visits after. Characteristics of the study groups for the full study timeframe (July 1, 2012April 30, 2014) are summarized in Table 1.
| Characteristic | Study Group* | |||
|---|---|---|---|---|
| Overall, N = 31,896 | Control, N = 25,586 | Intervention, N = 6,310 | P Value | |
| ||||
| Patient characteristics | ||||
| Age, y | 55.47 17.61 | 55.27 17.13 | 56.30 19.39 | <0.001 |
| Female gender | 14,995 (47%) | 11,753 (46%) | 3,242 (51%) | <0.001 |
| CCI | 3.73 3.25 | 3.61 3.17 | 4.20 3.54 | <0.001 |
| Outcomes | ||||
| Cost per day, $ | 130.95 392.16 | 131.57 423.94 | 127.68 220.40 | 0.022 |
| Cost per visit, $ | 733.75 1,693.98 | 772.30 1,847.65 | 577.40 795.29 | <0.001 |
| BMP tests per day | 0.73 1.17 | 0.74 1.19 | 0.67 1.05 | <0.001 |
| CMP tests per day | 0.20 0.67 | 0.19 0.68 | 0.26 0.62 | <0.001 |
| CBC tests per day | 0.83 1.10 | 0.84 1.15 | 0.73 0.82 | <0.001 |
| PT/INR tests per day | 0.36 1.03 | 0.36 1.07 | 0.34 0.83 | <.001 |
| LOS, d | 5.60 7.12 | 5.87 7.55 | 4.52 4.82 | <0.001 |
| 30‐day readmissions | 4,374 (14%) | 3,603 (14%) | 771 (12%) | <0.001 |
During the study period, there were 25,586 visits in the control group and 6310 visits in the intervention group. Patients in the intervention group were on average older than patients in the control group. There were more female patients in the intervention group. Mean CCI was 4.2 in the intervention group and 3.6 in the control group. The intervention group had lower LOS and 30‐day readmissions than the control group.
Descriptive statistics and simple comparisons of covariates and outcomes before and after the intervention are shown in Table 2. Age and gender distributions remained unchanged in both groups. CCI increased in the control group by 0.24 (P < 0.001) and remained unchanged in the intervention group. In the intervention group, lab cost per day was reduced from $138 before the intervention to $123 after the intervention (P < 0.001). In contrast, among control patients, cost per day increased nonsignificantly from $130 preintervention to $132 postintervention (P = 0.37). Number of tests per day significantly decreased for all specific tests in the intervention group. Readmission rates decreased significantly from 14% to 11% in the intervention group (P = 0.01). LOS remained constant in both groups.
| Characteristic* | Control | Intervention | ||||
|---|---|---|---|---|---|---|
| Preintervention, N = 8,102 | Postintervention, N = 17,484 | P Value | Preintervention, N = 2,034 | Postintervention, N = 4,276 | P Value | |
| ||||||
| Patient characteristics | ||||||
| Age, yr | 55.17 17.46 | 55.31 16.98 | 0.55 | 55.90 19.47 | 56.50 19.35 | 0.25 |
| Female gender | 3,707 (46%) | 8,046 (46%) | 0.69 | 1,039 (51%) | 2,203 (52%) | 0.74 |
| CCI | 3.45 3.06 | 3.69 3.21 | <0.001 | 4.19 3.51 | 4.20 3.56 | 0.89 |
| Outcomes | ||||||
| Cost per day, $ | 130.1 431.8 | 132.2 420.3 | 0.37 | 137.9 232.9 | 122.9 213.5 | <0.001 |
| Cost per visit, $ | 760.4 1,813.6 | 777.8 1,863.3 | 0.48 | 617.8 844.1 | 558.2 770.3 | 0.005 |
| BMP tests per day | 0.74 1.21 | 0.74 1.18 | 0.67 | 0.75 1.03 | 0.63 1.05 | <0.001 |
| CMP tests per day | 0.19 0.68 | 0.19 0.68 | 0.85 | 0.32 0.68 | 0.23 0.58 | <0.001 |
| CBC tests per day | 0.85 1.14 | 0.84 1.15 | 0.045 | 0.92 0.79 | 0.64 0.76 | <0.001 |
| PT/INR tests per day | 0.34 1.04 | 0.37 1.08 | <0.001 | 0.35 0.82 | 0.33 0.84 | 0.020 |
| LOS, d | 5.84 7.66 | 5.88 7.50 | 0.71 | 4.48 5.12 | 4.54 4.67 | 0.63 |
| 30‐day readmissions | 1,173 (14%) | 2,430 (14%) | 0.22 | 280 (14%) | 491 (11%) | 0.010 |
ITS analysis results are shown in Table 3. After the intervention, the difference in monthly means between the 2 groups dropped by $16 for cost per day (P = 0.034) and by $128 for cost per visit (P = 0.02). The decreased cost in the intervention group amounts to approximately $251,427 (95% confidence interval [CI]: $20,370‐$482,484) savings over the first year. If the intervention was rolled out for the control group and had a similar impact, it could have led to an additional cost savings of $1,321,669 (95% CI: 107,081‐2,536,256). Moreover, the number of basic metabolic panel, comprehensive metabolic panel, and complete blood count test per day were reduced significantly more in the intervention group compared to the control group (<0.001, 0.004, and <0.001).
| Outcome | Parameter* | Parameter Estimate | Standard Error | t Value | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| Lab cost per day ($) | Baseline difference level (b0) | 9.3450 | 6.5640 | 1.4237 | 0.16 |
| Baseline difference trend (b1) | 0.2150 | 0.7709 | 0.2789 | 0.78 | |
| Change in difference level after intervention(b2) | 16.1200 | 7.3297 | 2.1993 | 0.034 | |
| Change in difference trend after intervention (b3) | 0.2388 | 0.8090 | 0.2952 | 0.77 | |
| Lab cost per visit ($) | Baseline difference level (b0) | 166.081 | 48.3425 | 3.4355 | 0.001 |
| Baseline difference trend (b1) | 3.6663 | 5.8571 | 0.6260 | 0.53 | |
| Change in difference level after intervention(b2) | 128.527 | 53.0278 | 2.4238 | 0.020 | |
| Change in difference trend after intervention (b3) | 2.2586 | 5.8463 | 0.3863 | 0.70 | |
| BMP tests per day | Baseline difference level (b0) | 0.0061 | 0.0250 | 0.2439 | 0.81 |
| Baseline difference trend (b1) | 0.0004 | 0.0030 | 0.1449 | 0.89 | |
| Change in difference level after intervention(b2) | 0.1034 | 0.0276 | 3.7426 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0014 | 0.0030 | 0.4588 | 0.65 | |
| CMP tests per day | Baseline difference level (b0) | 0.1226 | 0.0226 | 5.4302 | <0.001 |
| Baseline difference trend (b1) | 0.0015 | 0.0028 | 0.5539 | 0.58 | |
| Change in difference level after intervention(b2) | 0.0754 | 0.0248 | 3.0397 | 0.004 | |
| Change in difference trend after intervention (b3) | 0.0030 | 0.0028 | 1.0937 | 0.28 | |
| CBC tests per day | Baseline difference level (b0) | 0.0539 | 0.0190 | 2.8338 | 0.007 |
| Baseline difference trend (b1) | 0.0013 | 0.0023 | 0.5594 | 0.58 | |
| Change in difference level after intervention(b2) | 0.2343 | 0.0213 | 10.997 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0036 | 0.0023 | 1.5539 | 0.13 | |
| PT/INR tests per day | Baseline difference level (b0) | 0.0413 | 0.0242 | 1.7063 | 0.096 |
| Baseline difference trend (b1) | 0.0040 | 0.0028 | 1.4095 | 0.17 | |
| Change in difference level after intervention(b2) | 0.0500 | 0.0270 | 1.8507 | 0.072 | |
| Change in difference trend after intervention (b3) | 0.0054 | 0.0030 | 1.7940 | 0.080 | |
| LOS, d | Baseline difference level (b0) | 1.4211 | 0.2746 | 5.1743 | <0.001 |
| Baseline difference trend (b1) | 0.0093 | 0.0333 | 0.2807 | 0.78 | |
| Change in difference level after intervention(b2) | 0.1007 | 0.2988 | 0.3368 | 0.74 | |
| Change in difference trend after intervention (b3) | 0.0053 | 0.0331 | 0.1588 | 0.87 | |
| 30‐day readmissions | Baseline difference level (b0) | 0.0057 | 0.0185 | 0.3084 | 0.76 |
| Baseline difference trend (b1) | 0.0017 | 0.0022 | 0.8016 | 0.43 | |
| Change in difference level after intervention(b2) | 0.0110 | 0.0206 | 0.5315 | 0.60 | |
| Change in difference trend after intervention (b3) | 0.0021 | 0.0023 | 0.9111 | 0.37 | |
Figure 1 shows a graphical representation of the biweekly means for the 2 primary outcomeslab cost per day and lab cost per visit. Figure 2 shows all other outcomes. To the right of each figure, P values are provided for the b2 coefficients from Table 3.


DISCUSSION
Through a multifaceted quality‐improvement initiative, the UUHC hospitalist group was able to reduce lab cost per day and per visit as well as commonly ordered routine labs as compared to an institutional control group. A multifaceted approach was selected given the literature supporting this approach as the most likely method to sustain improvement.[14] At the same time, the use of a multifaceted intervention makes it difficult to rigorously determine the relative impact of different components of the intervention. In discussing this issue, however, the hospitalist group felt that the driving factors for change were those related to process change, specifically, the use of a standardized rounding checklist to discuss lab testing and the routine review of lab costs at group meetings. The ultimate goal was to change the culture of routine test ordering into a thoughtful process of needed tests and thereby reduce costs. Prior to this intervention, the least experienced person on this team (the intern) ordered any test he or she wanted, usually without discussion. The intervention focused on this issue through standardized supervision and explicit discussion of laboratory tests. Importantly, although improvements from education initiatives typically decrease over time, the incorporation of process change in this intervention was felt to likely contribute to the sustained reduction seen at 15 months. Although use of the rounding checklist added another step to daily rounds, the routine cost feedback, including comparisons to peers, helped encourage use of the checklist. Thus, we feel that routine feedback was essential to sustaining the intervention and its impact.
Inappropriate and unnecessary testing has been recognized for decades, and multiple interventions have been attempted, including a recent article that demonstrated a 10% reduction in common laboratory ordering through an initiative mainly focused on education and ordering feedback.[25] Despite reported success of several interventions, none have combined multiple interventions and explicitly required discussion of laboratory tests on rounds. For example, although the UUHC intervention used Attali et al.[21] and Barie and Hydo's[20] work to develop the intervention, neither of these studies described how laboratory testing was discussed with the attending or supervising resident. The UUHC intervention thus builds on the current literature by combining other successful modalities with explicit discussion of laboratory testing via a rounding checklist and feedback with the novel VDO tool to reduce laboratory costs. A major strength of this intervention is the relatively low cost and the generalizability of implementing rounding checklists. Initial support from the hospital was needed to provide accurate VDO information to the hospitalist group. However, ongoing costs were minimal and related to any additional time spent during rounds to discuss laboratory tests. Thus, we feel that this intervention is feasible for wide replication.
Another strength of the study is the use of the VDO tool to measure actual costs. Whereas previous studies have relied on estimated costs with extrapolation to potential cost savings, this study used direct costs to the institution as a more accurate marker of cost savings. Additionally, most studies on lab utilization have used a before/after analysis without a control group. The presence of a control group for this analysis is important to help assess for institutional trends that may not be reflected in a before/after intervention. The reduction in cost in the intervention group despite a trend toward increased cost in the institutional control group supports the impact of this intervention.
Limitations of this study include that it was a single‐center, controlled ITS study and not a randomized controlled trial. Related to this limitation, the control group reflected a different patient population compared to the intervention group, with a longer LOS, lower CCI, and inclusion of nonmedical patients. However, these differences were relatively stable before and after the intervention. Also, ITS is considered one of the most robust research designs outside of randomized controlled trials, and it accounts for baseline differences in both levels and trends.[24] Nevertheless, it remains possible that secular trends existed that we did not capture and that affected the 2 populations differently.
A further limitation is that the baseline period was only 7 months and the intervention was 15 months. As the 7 months started in July, this could have reflected the time when interns were least experienced with ordering. Unfortunately, we did not have VDO availability for a full year prior to the intervention. We believe that any major effect due to this shortened baseline period should have been seen in the control group as well, and therefore accounted for in the analysis. Additionally, it is possible that there was spillover of the intervention to the control group, as internal medicine residents rotated throughout the hospital to other medical services (pulmonary, cardiology, hematology, and oncology). However, any effect of their rotation should have been to lower the control lab cost, thus making differences less profound.
CONCLUSIONS
A multifaceted approach to laboratory reduction through education, process change, cost feedback, and financial incentive resulted in a significant reduction in laboratory cost per day, laboratory cost per visit, and the ordering of common laboratory tests at a major academic medical center.
Acknowledgements
The authors thank Mr. Michael Swanicke for his assistance in process engineering, Mr. Tony Clawson for his routine provision of VDO data, and Ms. Selma Lopez for her editorial support.
Disclosures: K.K. is or has been a consultant on clinical decision support (CDS) or electronic clinical quality measurement to the US Office of the National Coordinator for Health IT, ARUP Laboratories, McKesson InterQual, ESAC, Inc., JBS International, Inc., Inflexxion, Inc., Intelligent Automation, Inc., Partners HealthCare, Mayo Clinic, and the RAND Corporation. K.K. receives royalties for a Duke University‐owned CDS technology for infectious disease management known as CustomID that he helped develop. K.K. was formerly a consultant for Religent, Inc. and a co‐owner and consultant for Clinica Software, Inc., both of which provide commercial CDS services, including through use of a CDS technology known as SEBASTIAN that K.K. developed. K.K. no longer has a financial relationship with either Religent or Clinica Software. K.K. has no competing interest with any specific product or intervention evaluated in this article. All other authors declare no competing interests.
Healthcare costs continue to increase and are estimated to be approximately $3.1 trillion per year in the United States.[1] Waste is a major contributor to this cost, accounting for an estimated $910 billion/year.[2] Laboratory tests are well documented to contribute to healthcare waste, with an estimated 30% to 50% of tests for hospitalized patients being unnecessary.[3, 4, 5] This issue has been highlighted by the American Board of Internal Medicine Foundation's Choosing Wisely campaign as an area to reduce waste.[6] Evaluating this concern locally, a University Health Systems Consortium 2011 analysis indicated that the University of Utah general internal medicine hospitalist service had a higher average direct lab cost per discharge compared to top performers, indicating an opportunity for improvement.
Multiple interventions have been described in the literature to address excessive laboratory utilization, including physician education, audit and feedback, cost information display, and administrative rules restricting certain types of ordering.[7, 8, 9, 10, 11] Despite these interventions, barriers remain common and not all interventions are sustained. For example, interventions focused mainly on education see a small improvement initially that is not sustained.[4, 12, 13] Additionally, although most studies focus on individual interventions, those that target multiple factors have been found to be more successful at producing and sustaining change.[14] Therefore, the opportunity existed to incorporate multiple etiologies into a single intervention and apply a checklist to laboratory ordering to see if combined modalities could be effective at reducing laboratory costs in a sustainable manner.
In addition to cost, there is potential patient harm resulting from unnecessary laboratory testing. For prolonged hospitalizations, anemia is a well‐recognized side effect of phlebotomy,[15, 16] and a recent evaluation of cardiac surgery patients found an average cumulative blood loss due to phlebotomy of 454 mL/hospital stay.[17] The sheer number of tests ordered can lead to false positive tests that result in additional testing and monitoring. Furthermore, patients subjected to laboratory blood draws are often awakened early in the morning, which is unpleasant and could adversely affect the patient experience.
Recognizing laboratory cost as a problem, the University of Utah general internal medicine hospitalist service implemented a multifaceted quality‐improvement initiative with a goal to reduce laboratory testing. At the time of this project, University of Utah Health Care (UUHC) developed a Value Driven Outcomes (VDO) tool to give direct data related to costs of care, including the actual cost paid by the hospital to the university‐owned laboratory vendor (ARUP Laboratories, Salt Lake City, UT) for testing.[18] The hospitalist group incorporated VDO into the initiative for routine cost feedback. This study evaluates the impact of this intervention on laboratory costs.
METHODS
Design
A retrospective, controlled, interrupted time series (ITS) study was performed to compare changes in lab costs between hospitalists (intervention study group) and other providers (control study group). The intervention initiation date was February 1, 2013. The baseline period was July 1, 2012 to January 31, 2013, as that was the period in which the VDO tool became available for cost analysis prior to intervention. The intervention period was February 1, 2013 to April 30, 2014, as there was a change in the electronic health record (EHR) in May 2014 that affected data flow and could act as a major confounder. The institutional review board classified this project as quality improvement and did not require review and oversight.
Setting
UUHC is a 500‐bed academic medical center in Salt Lake City, Utah. The hospitalist service is a teaching service composed of 4 teams with internal medicine residents and medical students. The nonhospitalist services include all surgical services, as well as pulmonary, cardiology, hematology, and oncology services on which internal medicine residents rotate. All services at UUHC are staffed by academic physicians affiliated with the University of Utah School of Medicine.
Population
All patients 18 years and older admitted to the hospital to a service other than obstetrics, rehabilitation, or psychiatry between July 1, 2012 and April 30, 2014 were evaluated. Patients with missing data for outcomes or covariates were excluded.
Intervention
Initial evaluation included an informal review of patient charts and discussion with hospitalist group members, both indicating laboratory overuse. A working group was then established including hospitalists and process engineers to evaluate the workflow by which laboratory tests were ordered. Concurrently, a literature review was performed to help identify the scope of the problem and evaluate methods that had been successful at other institutions. Through this review, it was noted that interns were the most frequent orderers of tests and the largest contributors to variation of testing for inpatients.[19] Two specific studies with direct applicability to this project demonstrated that discussion of costs with attendings in a trauma intensive care unit resulted in a 30% reduction of tests ordered,[20] and discussion of testing with a senior resident in an internal medicine inpatient setting demonstrated a 20% reduction in laboratory testing.[21]
Our laboratory reduction intervention expanded on the current literature to incorporate education, process change, cost feedback, and financial incentives. Specifically, starting February 1, 2013, the following interventions were performed:
- Education of all providers involved, including the hospitalist group and all internal medicine residents at the start of their rotation with the hospitalist service. Education included a 30‐minute discussion of laboratory overuse, costs associated with laboratory overuse, previous interventions and their success, and current intervention with goals. Each resident was provided a pocket card with the most common lab tests and associated charges. Charges were used instead of costs due to concerns regarding the possible public dissemination of institutional costs.
- Standardization of the rounding process including a checklist review (see Supporting Information, Appendix, in the online version of this article) for all patients that ensured discussion of labs, telemetry, pain, lines/tubes, nursing presence, and follow‐up needed. The expectation was that all plans for lab testing would be discussed during rounds. The third‐year medical student was responsible to ensure that all items were covered daily on each patient.
- Monthly feedback at the hospitalist group meeting regarding laboratory costs using the VDO tool. Data were presented as a monthly group average and compared to preintervention baseline costs. Individual performance could be viewed and compared to other providers within the group.
- Financial incentive through a program that shares 50% of cost savings realized by the hospital with the Division of General Internal Medicine. The incentive could be used to support future quality‐improvement projects, but there was no individual physician incentive.
Data Collection and Preparation
Clinical data were collected in the inpatient EHR (Cerner Corp., Kansas City, MO) and later imported into the enterprise data warehouse (EDW) as part of the normal data flow. Billing data were imported into the EDW from the billing system. Cost data were estimated using the VDO tool developed by the University of Utah to identify clinical costs to the UUHC system.[18]
Clinical and Cost Outcomes
We hypothesized that following the intervention, the number of tests and lab costs would decrease greater for patients in the intervention group than in the control group, with no adverse effect on length of stay (LOS) or 30‐day readmissions.
Lab cost per day was calculated as the total lab cost per visit divided by the LOS. We adjusted all lab costs to 2013 US dollars using Consumer Price Index inflation data.[22] To account for different LOS, we used LOS as a weight variable when estimating descriptive characteristics and P values for lab cost per day and the number of tests. Thirty‐day readmissions included inpatient encounters followed by another inpatient encounter within 30 days excluding obstetrics, rehabilitation, and psychiatry visits.
Descriptive Variables
We included information on age at admission in years and Charlson Comorbidity Index (CCI) to evaluate differences in control and intervention groups.[23]
Statistical Analysis
First, unadjusted descriptive statistics were calculated for study outcomes and visit characteristics. Descriptive statistics were expressed as n (%) and mean standard deviation. Simple comparisons were performed based on 2 tests of homogeneity for categorical variables and on t tests for continuous variables.
Second, an ITS analysis was conducted to evaluate the impact of the intervention while accounting for baseline trends.[24] In this analysis, the dependent variable (yt) was the difference in aggregated outcome measures between the intervention and control groups every 2 weeks (eg, difference in average lab costs in a given 2‐week period between the 2 groups). Intervention impact was then evaluated in terms of changes in the level of the outcome (b2) as well as in the trend over time (b3) compared to the initial difference in means (b0) and baseline trend (b1). The following difference‐in‐differences segmented regression model was fitted using the autoreg procedure in SAS: yt = b0 + b1*timet + b2*study periodt + b3*time after the interventiont + errort, where timet is biweekly intervals after the beginning of the study, time after the interventiont is biweekly intervals after the intervention date, and study periodt is 1 postintervention and 0 preintervention. The models were fitted using maximum likelihood and stepwise autoregression to test 24 lags.
P values <0.05 were considered significant. SAS (version 9.3; SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
We analyzed 48,327 inpatient visits that met inclusion criteria. We excluded 15,659 obstetrics, rehabilitation, and psychiatry visits. Seven hundred seventy‐two (2.4%) of the remaining visits were excluded due to missing data. A total of 31,896 inpatient visits by 22,545 patients were included in the analysis. There were 10,136 visits before the intervention and 21,760 visits after. Characteristics of the study groups for the full study timeframe (July 1, 2012April 30, 2014) are summarized in Table 1.
| Characteristic | Study Group* | |||
|---|---|---|---|---|
| Overall, N = 31,896 | Control, N = 25,586 | Intervention, N = 6,310 | P Value | |
| ||||
| Patient characteristics | ||||
| Age, y | 55.47 17.61 | 55.27 17.13 | 56.30 19.39 | <0.001 |
| Female gender | 14,995 (47%) | 11,753 (46%) | 3,242 (51%) | <0.001 |
| CCI | 3.73 3.25 | 3.61 3.17 | 4.20 3.54 | <0.001 |
| Outcomes | ||||
| Cost per day, $ | 130.95 392.16 | 131.57 423.94 | 127.68 220.40 | 0.022 |
| Cost per visit, $ | 733.75 1,693.98 | 772.30 1,847.65 | 577.40 795.29 | <0.001 |
| BMP tests per day | 0.73 1.17 | 0.74 1.19 | 0.67 1.05 | <0.001 |
| CMP tests per day | 0.20 0.67 | 0.19 0.68 | 0.26 0.62 | <0.001 |
| CBC tests per day | 0.83 1.10 | 0.84 1.15 | 0.73 0.82 | <0.001 |
| PT/INR tests per day | 0.36 1.03 | 0.36 1.07 | 0.34 0.83 | <.001 |
| LOS, d | 5.60 7.12 | 5.87 7.55 | 4.52 4.82 | <0.001 |
| 30‐day readmissions | 4,374 (14%) | 3,603 (14%) | 771 (12%) | <0.001 |
During the study period, there were 25,586 visits in the control group and 6310 visits in the intervention group. Patients in the intervention group were on average older than patients in the control group. There were more female patients in the intervention group. Mean CCI was 4.2 in the intervention group and 3.6 in the control group. The intervention group had lower LOS and 30‐day readmissions than the control group.
Descriptive statistics and simple comparisons of covariates and outcomes before and after the intervention are shown in Table 2. Age and gender distributions remained unchanged in both groups. CCI increased in the control group by 0.24 (P < 0.001) and remained unchanged in the intervention group. In the intervention group, lab cost per day was reduced from $138 before the intervention to $123 after the intervention (P < 0.001). In contrast, among control patients, cost per day increased nonsignificantly from $130 preintervention to $132 postintervention (P = 0.37). Number of tests per day significantly decreased for all specific tests in the intervention group. Readmission rates decreased significantly from 14% to 11% in the intervention group (P = 0.01). LOS remained constant in both groups.
| Characteristic* | Control | Intervention | ||||
|---|---|---|---|---|---|---|
| Preintervention, N = 8,102 | Postintervention, N = 17,484 | P Value | Preintervention, N = 2,034 | Postintervention, N = 4,276 | P Value | |
| ||||||
| Patient characteristics | ||||||
| Age, yr | 55.17 17.46 | 55.31 16.98 | 0.55 | 55.90 19.47 | 56.50 19.35 | 0.25 |
| Female gender | 3,707 (46%) | 8,046 (46%) | 0.69 | 1,039 (51%) | 2,203 (52%) | 0.74 |
| CCI | 3.45 3.06 | 3.69 3.21 | <0.001 | 4.19 3.51 | 4.20 3.56 | 0.89 |
| Outcomes | ||||||
| Cost per day, $ | 130.1 431.8 | 132.2 420.3 | 0.37 | 137.9 232.9 | 122.9 213.5 | <0.001 |
| Cost per visit, $ | 760.4 1,813.6 | 777.8 1,863.3 | 0.48 | 617.8 844.1 | 558.2 770.3 | 0.005 |
| BMP tests per day | 0.74 1.21 | 0.74 1.18 | 0.67 | 0.75 1.03 | 0.63 1.05 | <0.001 |
| CMP tests per day | 0.19 0.68 | 0.19 0.68 | 0.85 | 0.32 0.68 | 0.23 0.58 | <0.001 |
| CBC tests per day | 0.85 1.14 | 0.84 1.15 | 0.045 | 0.92 0.79 | 0.64 0.76 | <0.001 |
| PT/INR tests per day | 0.34 1.04 | 0.37 1.08 | <0.001 | 0.35 0.82 | 0.33 0.84 | 0.020 |
| LOS, d | 5.84 7.66 | 5.88 7.50 | 0.71 | 4.48 5.12 | 4.54 4.67 | 0.63 |
| 30‐day readmissions | 1,173 (14%) | 2,430 (14%) | 0.22 | 280 (14%) | 491 (11%) | 0.010 |
ITS analysis results are shown in Table 3. After the intervention, the difference in monthly means between the 2 groups dropped by $16 for cost per day (P = 0.034) and by $128 for cost per visit (P = 0.02). The decreased cost in the intervention group amounts to approximately $251,427 (95% confidence interval [CI]: $20,370‐$482,484) savings over the first year. If the intervention was rolled out for the control group and had a similar impact, it could have led to an additional cost savings of $1,321,669 (95% CI: 107,081‐2,536,256). Moreover, the number of basic metabolic panel, comprehensive metabolic panel, and complete blood count test per day were reduced significantly more in the intervention group compared to the control group (<0.001, 0.004, and <0.001).
| Outcome | Parameter* | Parameter Estimate | Standard Error | t Value | Pr > |t| |
|---|---|---|---|---|---|
| |||||
| Lab cost per day ($) | Baseline difference level (b0) | 9.3450 | 6.5640 | 1.4237 | 0.16 |
| Baseline difference trend (b1) | 0.2150 | 0.7709 | 0.2789 | 0.78 | |
| Change in difference level after intervention(b2) | 16.1200 | 7.3297 | 2.1993 | 0.034 | |
| Change in difference trend after intervention (b3) | 0.2388 | 0.8090 | 0.2952 | 0.77 | |
| Lab cost per visit ($) | Baseline difference level (b0) | 166.081 | 48.3425 | 3.4355 | 0.001 |
| Baseline difference trend (b1) | 3.6663 | 5.8571 | 0.6260 | 0.53 | |
| Change in difference level after intervention(b2) | 128.527 | 53.0278 | 2.4238 | 0.020 | |
| Change in difference trend after intervention (b3) | 2.2586 | 5.8463 | 0.3863 | 0.70 | |
| BMP tests per day | Baseline difference level (b0) | 0.0061 | 0.0250 | 0.2439 | 0.81 |
| Baseline difference trend (b1) | 0.0004 | 0.0030 | 0.1449 | 0.89 | |
| Change in difference level after intervention(b2) | 0.1034 | 0.0276 | 3.7426 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0014 | 0.0030 | 0.4588 | 0.65 | |
| CMP tests per day | Baseline difference level (b0) | 0.1226 | 0.0226 | 5.4302 | <0.001 |
| Baseline difference trend (b1) | 0.0015 | 0.0028 | 0.5539 | 0.58 | |
| Change in difference level after intervention(b2) | 0.0754 | 0.0248 | 3.0397 | 0.004 | |
| Change in difference trend after intervention (b3) | 0.0030 | 0.0028 | 1.0937 | 0.28 | |
| CBC tests per day | Baseline difference level (b0) | 0.0539 | 0.0190 | 2.8338 | 0.007 |
| Baseline difference trend (b1) | 0.0013 | 0.0023 | 0.5594 | 0.58 | |
| Change in difference level after intervention(b2) | 0.2343 | 0.0213 | 10.997 | <0.001 | |
| Change in difference trend after intervention (b3) | 0.0036 | 0.0023 | 1.5539 | 0.13 | |
| PT/INR tests per day | Baseline difference level (b0) | 0.0413 | 0.0242 | 1.7063 | 0.096 |
| Baseline difference trend (b1) | 0.0040 | 0.0028 | 1.4095 | 0.17 | |
| Change in difference level after intervention(b2) | 0.0500 | 0.0270 | 1.8507 | 0.072 | |
| Change in difference trend after intervention (b3) | 0.0054 | 0.0030 | 1.7940 | 0.080 | |
| LOS, d | Baseline difference level (b0) | 1.4211 | 0.2746 | 5.1743 | <0.001 |
| Baseline difference trend (b1) | 0.0093 | 0.0333 | 0.2807 | 0.78 | |
| Change in difference level after intervention(b2) | 0.1007 | 0.2988 | 0.3368 | 0.74 | |
| Change in difference trend after intervention (b3) | 0.0053 | 0.0331 | 0.1588 | 0.87 | |
| 30‐day readmissions | Baseline difference level (b0) | 0.0057 | 0.0185 | 0.3084 | 0.76 |
| Baseline difference trend (b1) | 0.0017 | 0.0022 | 0.8016 | 0.43 | |
| Change in difference level after intervention(b2) | 0.0110 | 0.0206 | 0.5315 | 0.60 | |
| Change in difference trend after intervention (b3) | 0.0021 | 0.0023 | 0.9111 | 0.37 | |
Figure 1 shows a graphical representation of the biweekly means for the 2 primary outcomeslab cost per day and lab cost per visit. Figure 2 shows all other outcomes. To the right of each figure, P values are provided for the b2 coefficients from Table 3.


DISCUSSION
Through a multifaceted quality‐improvement initiative, the UUHC hospitalist group was able to reduce lab cost per day and per visit as well as commonly ordered routine labs as compared to an institutional control group. A multifaceted approach was selected given the literature supporting this approach as the most likely method to sustain improvement.[14] At the same time, the use of a multifaceted intervention makes it difficult to rigorously determine the relative impact of different components of the intervention. In discussing this issue, however, the hospitalist group felt that the driving factors for change were those related to process change, specifically, the use of a standardized rounding checklist to discuss lab testing and the routine review of lab costs at group meetings. The ultimate goal was to change the culture of routine test ordering into a thoughtful process of needed tests and thereby reduce costs. Prior to this intervention, the least experienced person on this team (the intern) ordered any test he or she wanted, usually without discussion. The intervention focused on this issue through standardized supervision and explicit discussion of laboratory tests. Importantly, although improvements from education initiatives typically decrease over time, the incorporation of process change in this intervention was felt to likely contribute to the sustained reduction seen at 15 months. Although use of the rounding checklist added another step to daily rounds, the routine cost feedback, including comparisons to peers, helped encourage use of the checklist. Thus, we feel that routine feedback was essential to sustaining the intervention and its impact.
Inappropriate and unnecessary testing has been recognized for decades, and multiple interventions have been attempted, including a recent article that demonstrated a 10% reduction in common laboratory ordering through an initiative mainly focused on education and ordering feedback.[25] Despite reported success of several interventions, none have combined multiple interventions and explicitly required discussion of laboratory tests on rounds. For example, although the UUHC intervention used Attali et al.[21] and Barie and Hydo's[20] work to develop the intervention, neither of these studies described how laboratory testing was discussed with the attending or supervising resident. The UUHC intervention thus builds on the current literature by combining other successful modalities with explicit discussion of laboratory testing via a rounding checklist and feedback with the novel VDO tool to reduce laboratory costs. A major strength of this intervention is the relatively low cost and the generalizability of implementing rounding checklists. Initial support from the hospital was needed to provide accurate VDO information to the hospitalist group. However, ongoing costs were minimal and related to any additional time spent during rounds to discuss laboratory tests. Thus, we feel that this intervention is feasible for wide replication.
Another strength of the study is the use of the VDO tool to measure actual costs. Whereas previous studies have relied on estimated costs with extrapolation to potential cost savings, this study used direct costs to the institution as a more accurate marker of cost savings. Additionally, most studies on lab utilization have used a before/after analysis without a control group. The presence of a control group for this analysis is important to help assess for institutional trends that may not be reflected in a before/after intervention. The reduction in cost in the intervention group despite a trend toward increased cost in the institutional control group supports the impact of this intervention.
Limitations of this study include that it was a single‐center, controlled ITS study and not a randomized controlled trial. Related to this limitation, the control group reflected a different patient population compared to the intervention group, with a longer LOS, lower CCI, and inclusion of nonmedical patients. However, these differences were relatively stable before and after the intervention. Also, ITS is considered one of the most robust research designs outside of randomized controlled trials, and it accounts for baseline differences in both levels and trends.[24] Nevertheless, it remains possible that secular trends existed that we did not capture and that affected the 2 populations differently.
A further limitation is that the baseline period was only 7 months and the intervention was 15 months. As the 7 months started in July, this could have reflected the time when interns were least experienced with ordering. Unfortunately, we did not have VDO availability for a full year prior to the intervention. We believe that any major effect due to this shortened baseline period should have been seen in the control group as well, and therefore accounted for in the analysis. Additionally, it is possible that there was spillover of the intervention to the control group, as internal medicine residents rotated throughout the hospital to other medical services (pulmonary, cardiology, hematology, and oncology). However, any effect of their rotation should have been to lower the control lab cost, thus making differences less profound.
CONCLUSIONS
A multifaceted approach to laboratory reduction through education, process change, cost feedback, and financial incentive resulted in a significant reduction in laboratory cost per day, laboratory cost per visit, and the ordering of common laboratory tests at a major academic medical center.
Acknowledgements
The authors thank Mr. Michael Swanicke for his assistance in process engineering, Mr. Tony Clawson for his routine provision of VDO data, and Ms. Selma Lopez for her editorial support.
Disclosures: K.K. is or has been a consultant on clinical decision support (CDS) or electronic clinical quality measurement to the US Office of the National Coordinator for Health IT, ARUP Laboratories, McKesson InterQual, ESAC, Inc., JBS International, Inc., Inflexxion, Inc., Intelligent Automation, Inc., Partners HealthCare, Mayo Clinic, and the RAND Corporation. K.K. receives royalties for a Duke University‐owned CDS technology for infectious disease management known as CustomID that he helped develop. K.K. was formerly a consultant for Religent, Inc. and a co‐owner and consultant for Clinica Software, Inc., both of which provide commercial CDS services, including through use of a CDS technology known as SEBASTIAN that K.K. developed. K.K. no longer has a financial relationship with either Religent or Clinica Software. K.K. has no competing interest with any specific product or intervention evaluated in this article. All other authors declare no competing interests.
- , , , et al. National health expenditure projections, 2014–24: spending growth faster than recent trends. Health Aff (Millwood). 2015;34(8):1407–1417.
- , . Eliminating waste in US health care. JAMA. 2012;307(14):1513–1516.
- , , , et al. Utilization of arterial blood gas measurements in a large tertiary care hospital. Am J Clin Pathol. 2007;127:604–609.
- , . Strategies to promote rational clinical chemistry test utilization. Clin Biochem. 1996;29:291–299.
- , , , , . The landscape of inappropriate laboratory testing: a 15‐year meta‐analysis. PLoS One. 2013;8:e78962.
- ABIM Choosing Wisely Society of Hospital Medicine–Adult Hospital Medicine. Five things physicians and patients should question. Available at: http://www.choosingwisely.org/societies/society‐of‐hospital‐medicine‐adult. Published February 21, 2013. Accessed September 2, 2015.
- , , , , , . Effect of daily charge feedback on inpatient charges and physician knowledge and behavior. Arch Intern Med. 1989;149:426–429.
- , , , et al. A utilization management intervention to reduce unnecessary testing in the coronary care unit. Arch Intern Med. 2002;162:1885–1890.
- , , , et al. The impact of peer management on test‐ordering behavior. Ann Intern Med. 2004;141:196–204.
- , , , , . An administrative intervention to improve the utilization of laboratory tests within a university hospital. Int J Qual Health Care. 2005;17:243–248.
- , , , et al. Impact of providing fee data on laboratory test ordering. JAMA Intern Med. 2013;173(10):903–908.
- , , , et al. The failure of physician education as a cost containment strategy. JAMA. 1984;252:225–230.
- . Is that lab test necessary? Am J Clin Pathol. 2006;126:335–336.
- , , , . Techniques to improve physicians' use of diagnostic tests. JAMA. 1998;280:2020–2027.
- , , . Laboratory testing in the intensive care unit. Crit Care Clin. 2007;23:435–465.
- . Complications of critical care: lab testing and iatrogenic anemia. MLO Med Lab Obs. 200;33(10):28–31.
- , , , et al. Contemporary bloodletting in cardiac surgical care. Ann Thorac Surg. 2015;99:779–785.
- , , , et al. Value Driven Outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015:22:223–235.
- , , . The impact of residents, interns, and attendings on inpatient laboratory ordering patterns: a report from one university's hospitalist service. Acad Med. 2011;86:139–145.
- , . Learning to not know: results of a program for ancillary cost reduction in surgical care. J Trauma. 1996;41:714–720.
- , , , et al. A cost‐effective method for reducing the volume of laboratory tests in a university‐associated teaching hospital. Mt Sinai J Med. 2006;73:787–794.
- US Bureau of Labor Statistics. CPI inflation calculator. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed May 22, 2015.
- , , , et al. Coding algorithms for defining comorbidities in ICD‐9‐CM and ICD‐10 administrative data. Med Care. 2005;43:113–1139.
- , , , . Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther. 2002;27(4):299–309.
- , , , et al. A multifaceted hospitalist quality improvement intervention: decreased frequency of common labs. J Hosp Med. 2015;10:390–395.
- , , , et al. National health expenditure projections, 2014–24: spending growth faster than recent trends. Health Aff (Millwood). 2015;34(8):1407–1417.
- , . Eliminating waste in US health care. JAMA. 2012;307(14):1513–1516.
- , , , et al. Utilization of arterial blood gas measurements in a large tertiary care hospital. Am J Clin Pathol. 2007;127:604–609.
- , . Strategies to promote rational clinical chemistry test utilization. Clin Biochem. 1996;29:291–299.
- , , , , . The landscape of inappropriate laboratory testing: a 15‐year meta‐analysis. PLoS One. 2013;8:e78962.
- ABIM Choosing Wisely Society of Hospital Medicine–Adult Hospital Medicine. Five things physicians and patients should question. Available at: http://www.choosingwisely.org/societies/society‐of‐hospital‐medicine‐adult. Published February 21, 2013. Accessed September 2, 2015.
- , , , , , . Effect of daily charge feedback on inpatient charges and physician knowledge and behavior. Arch Intern Med. 1989;149:426–429.
- , , , et al. A utilization management intervention to reduce unnecessary testing in the coronary care unit. Arch Intern Med. 2002;162:1885–1890.
- , , , et al. The impact of peer management on test‐ordering behavior. Ann Intern Med. 2004;141:196–204.
- , , , , . An administrative intervention to improve the utilization of laboratory tests within a university hospital. Int J Qual Health Care. 2005;17:243–248.
- , , , et al. Impact of providing fee data on laboratory test ordering. JAMA Intern Med. 2013;173(10):903–908.
- , , , et al. The failure of physician education as a cost containment strategy. JAMA. 1984;252:225–230.
- . Is that lab test necessary? Am J Clin Pathol. 2006;126:335–336.
- , , , . Techniques to improve physicians' use of diagnostic tests. JAMA. 1998;280:2020–2027.
- , , . Laboratory testing in the intensive care unit. Crit Care Clin. 2007;23:435–465.
- . Complications of critical care: lab testing and iatrogenic anemia. MLO Med Lab Obs. 200;33(10):28–31.
- , , , et al. Contemporary bloodletting in cardiac surgical care. Ann Thorac Surg. 2015;99:779–785.
- , , , et al. Value Driven Outcomes (VDO): a pragmatic, modular, and extensible software framework for understanding and improving health care costs and outcomes. J Am Med Inform Assoc. 2015:22:223–235.
- , , . The impact of residents, interns, and attendings on inpatient laboratory ordering patterns: a report from one university's hospitalist service. Acad Med. 2011;86:139–145.
- , . Learning to not know: results of a program for ancillary cost reduction in surgical care. J Trauma. 1996;41:714–720.
- , , , et al. A cost‐effective method for reducing the volume of laboratory tests in a university‐associated teaching hospital. Mt Sinai J Med. 2006;73:787–794.
- US Bureau of Labor Statistics. CPI inflation calculator. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed May 22, 2015.
- , , , et al. Coding algorithms for defining comorbidities in ICD‐9‐CM and ICD‐10 administrative data. Med Care. 2005;43:113–1139.
- , , , . Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther. 2002;27(4):299–309.
- , , , et al. A multifaceted hospitalist quality improvement intervention: decreased frequency of common labs. J Hosp Med. 2015;10:390–395.
© 2016 Society of Hospital Medicine
Thrombectomy within Eight Hours of Stroke Onset Reduces Poststroke Disability
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin scale.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups. The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. New Engl J Med. 2015;372(24):2296–2306.
Visit our website for more hospitalist reviews of HM-focused research.
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin scale.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups. The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. New Engl J Med. 2015;372(24):2296–2306.
Visit our website for more hospitalist reviews of HM-focused research.
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin scale.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups. The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. New Engl J Med. 2015;372(24):2296–2306.
Visit our website for more hospitalist reviews of HM-focused research.
High-Flow Nasal Oxygen Therapy Noninferior to BiPAP in Post-Operative Respiratory Failure
Clinical question: In post-operative cardiothoracic surgery patients, is high-flow nasal oxygen therapy inferior to BiPAP for resolution of acute respiratory failure?
Background: Acute respiratory failure is common following cardiothoracic surgery, and noninvasive ventilation often is used to avoid intubation. Noninvasive ventilation is resource-intensive and might be uncomfortable to patients. High-flow nasal oxygen therapy is an alternative modality, which provides large amounts of oxygen with more ease and patient comfort.
Study design: Multi-center, randomized, noninferiority trial.
Setting: Six ICUs in France.
Synopsis: Investigators randomized 830 patients who met criteria (obesity, heart failure, or failure of spontaneous breathing trial) after cardiothoracic surgery. These patients were prophylactically treated with high-flow nasal oxygen or BiPAP. Patients with sleep apnea, nausea/vomiting, agitation/confusion, or hemodynamic instability were excluded. Data collected included arterial blood gas, respiratory rate, and patient-rated dyspnea. The primary outcome was treatment failure as defined by reintubation and mechanical ventilation, a switch to the other study treatment, or study treatment discontinuation.
Complications included pneumothorax, colonic pseudoobstruction, and nosocomial pneumonia. The expected rate of failure for BiPAP was 20%. High-flow nasal oxygen therapy was not inferior to BiPAP, with similar treatment failure rates occurring in both groups (21.9% in BiPAP patients vs. 21% of high-flow nasal oxygen patients); 20% of patients experienced persistent discomfort with either treatment method.
There were no significant differences in complications between the two study groups. Limitations included lack of blinding and potential for bias, leading to treatment failure and crossover.
Bottom line: High-flow nasal oxygen was noninferior to BiPAP in patients with respiratory failure after cardiothoracic surgery.
Citation: Stéphan F, Barrucand B, Petit P, et al. High-flow nasal oxygen vs noninvasive positive airway pressure in hypoxemic patients after cardiothoracic surgery: a randomized clinical trial. JAMA. 2015;313(23):2331-2339.
Clinical question: In post-operative cardiothoracic surgery patients, is high-flow nasal oxygen therapy inferior to BiPAP for resolution of acute respiratory failure?
Background: Acute respiratory failure is common following cardiothoracic surgery, and noninvasive ventilation often is used to avoid intubation. Noninvasive ventilation is resource-intensive and might be uncomfortable to patients. High-flow nasal oxygen therapy is an alternative modality, which provides large amounts of oxygen with more ease and patient comfort.
Study design: Multi-center, randomized, noninferiority trial.
Setting: Six ICUs in France.
Synopsis: Investigators randomized 830 patients who met criteria (obesity, heart failure, or failure of spontaneous breathing trial) after cardiothoracic surgery. These patients were prophylactically treated with high-flow nasal oxygen or BiPAP. Patients with sleep apnea, nausea/vomiting, agitation/confusion, or hemodynamic instability were excluded. Data collected included arterial blood gas, respiratory rate, and patient-rated dyspnea. The primary outcome was treatment failure as defined by reintubation and mechanical ventilation, a switch to the other study treatment, or study treatment discontinuation.
Complications included pneumothorax, colonic pseudoobstruction, and nosocomial pneumonia. The expected rate of failure for BiPAP was 20%. High-flow nasal oxygen therapy was not inferior to BiPAP, with similar treatment failure rates occurring in both groups (21.9% in BiPAP patients vs. 21% of high-flow nasal oxygen patients); 20% of patients experienced persistent discomfort with either treatment method.
There were no significant differences in complications between the two study groups. Limitations included lack of blinding and potential for bias, leading to treatment failure and crossover.
Bottom line: High-flow nasal oxygen was noninferior to BiPAP in patients with respiratory failure after cardiothoracic surgery.
Citation: Stéphan F, Barrucand B, Petit P, et al. High-flow nasal oxygen vs noninvasive positive airway pressure in hypoxemic patients after cardiothoracic surgery: a randomized clinical trial. JAMA. 2015;313(23):2331-2339.
Clinical question: In post-operative cardiothoracic surgery patients, is high-flow nasal oxygen therapy inferior to BiPAP for resolution of acute respiratory failure?
Background: Acute respiratory failure is common following cardiothoracic surgery, and noninvasive ventilation often is used to avoid intubation. Noninvasive ventilation is resource-intensive and might be uncomfortable to patients. High-flow nasal oxygen therapy is an alternative modality, which provides large amounts of oxygen with more ease and patient comfort.
Study design: Multi-center, randomized, noninferiority trial.
Setting: Six ICUs in France.
Synopsis: Investigators randomized 830 patients who met criteria (obesity, heart failure, or failure of spontaneous breathing trial) after cardiothoracic surgery. These patients were prophylactically treated with high-flow nasal oxygen or BiPAP. Patients with sleep apnea, nausea/vomiting, agitation/confusion, or hemodynamic instability were excluded. Data collected included arterial blood gas, respiratory rate, and patient-rated dyspnea. The primary outcome was treatment failure as defined by reintubation and mechanical ventilation, a switch to the other study treatment, or study treatment discontinuation.
Complications included pneumothorax, colonic pseudoobstruction, and nosocomial pneumonia. The expected rate of failure for BiPAP was 20%. High-flow nasal oxygen therapy was not inferior to BiPAP, with similar treatment failure rates occurring in both groups (21.9% in BiPAP patients vs. 21% of high-flow nasal oxygen patients); 20% of patients experienced persistent discomfort with either treatment method.
There were no significant differences in complications between the two study groups. Limitations included lack of blinding and potential for bias, leading to treatment failure and crossover.
Bottom line: High-flow nasal oxygen was noninferior to BiPAP in patients with respiratory failure after cardiothoracic surgery.
Citation: Stéphan F, Barrucand B, Petit P, et al. High-flow nasal oxygen vs noninvasive positive airway pressure in hypoxemic patients after cardiothoracic surgery: a randomized clinical trial. JAMA. 2015;313(23):2331-2339.
Supplemental Oxygen During STEMI Might Increase Myocardial Injury
Clinical question: Does routine oxygen supplementation in patients with STEMI increase myocardial injury?
Background: Because of physiologic and clinical studies conducted before the era of acute coronary intervention, supplemental oxygen routinely is administered to patients with STEMI, regardless of oxygen saturation; however, recent studies have shown possible adverse effects of oxygen, including increased reperfusion injury and increased adverse outcomes in small clinical trials.
Study design: Multicenter, prospective, randomized, controlled trial (RCT).
Setting: Nine metropolitan hospitals.
Synopsis: This multicenter study included 441 patients with STEMI who were 18 years or older and were randomized by paramedics to receive either 8 L/min of oxygen or no supplemental oxygen. All patients then received protocolized care. The primary endpoint of myocardial infarct size, determined by mean peak of creatine kinase, was significantly increased in the oxygen group compared to the no oxygen group (1948 vs. 1543 U/L; means ratio, 1.27; 95% confidence interval, 1.04-1.52; P=0.01). There were nonsignificant increases of secondary endpoints in the oxygen group, including rate of recurrent myocardial infarction (5.5% vs. 0.9%; P=0.006), frequency of arrhythmia (40.4% vs. 31.4%; P=0.05), and size of infarct on six-month cardiac MRI (n=139; 20.3 vs. 13.1 g; P=0.04).
This study has several limitations: It was powered to detect differences in biomarkers (not clinical endpoints) and the treatment was not blinded to paramedics, patients, or cardiology teams.
Bottom line: Supplemental oxygen administration in patients with STEMI might increase infarct size and lead to poorer clinical outcomes; however, larger clinical trials are warranted.
Citation: Stub D, Smith K, Bernard S, et al. Air versus oxygen in ST-segment-elevation myocardial infarction. Circulation. 2015;131(24):2143-2150.
Clinical question: Does routine oxygen supplementation in patients with STEMI increase myocardial injury?
Background: Because of physiologic and clinical studies conducted before the era of acute coronary intervention, supplemental oxygen routinely is administered to patients with STEMI, regardless of oxygen saturation; however, recent studies have shown possible adverse effects of oxygen, including increased reperfusion injury and increased adverse outcomes in small clinical trials.
Study design: Multicenter, prospective, randomized, controlled trial (RCT).
Setting: Nine metropolitan hospitals.
Synopsis: This multicenter study included 441 patients with STEMI who were 18 years or older and were randomized by paramedics to receive either 8 L/min of oxygen or no supplemental oxygen. All patients then received protocolized care. The primary endpoint of myocardial infarct size, determined by mean peak of creatine kinase, was significantly increased in the oxygen group compared to the no oxygen group (1948 vs. 1543 U/L; means ratio, 1.27; 95% confidence interval, 1.04-1.52; P=0.01). There were nonsignificant increases of secondary endpoints in the oxygen group, including rate of recurrent myocardial infarction (5.5% vs. 0.9%; P=0.006), frequency of arrhythmia (40.4% vs. 31.4%; P=0.05), and size of infarct on six-month cardiac MRI (n=139; 20.3 vs. 13.1 g; P=0.04).
This study has several limitations: It was powered to detect differences in biomarkers (not clinical endpoints) and the treatment was not blinded to paramedics, patients, or cardiology teams.
Bottom line: Supplemental oxygen administration in patients with STEMI might increase infarct size and lead to poorer clinical outcomes; however, larger clinical trials are warranted.
Citation: Stub D, Smith K, Bernard S, et al. Air versus oxygen in ST-segment-elevation myocardial infarction. Circulation. 2015;131(24):2143-2150.
Clinical question: Does routine oxygen supplementation in patients with STEMI increase myocardial injury?
Background: Because of physiologic and clinical studies conducted before the era of acute coronary intervention, supplemental oxygen routinely is administered to patients with STEMI, regardless of oxygen saturation; however, recent studies have shown possible adverse effects of oxygen, including increased reperfusion injury and increased adverse outcomes in small clinical trials.
Study design: Multicenter, prospective, randomized, controlled trial (RCT).
Setting: Nine metropolitan hospitals.
Synopsis: This multicenter study included 441 patients with STEMI who were 18 years or older and were randomized by paramedics to receive either 8 L/min of oxygen or no supplemental oxygen. All patients then received protocolized care. The primary endpoint of myocardial infarct size, determined by mean peak of creatine kinase, was significantly increased in the oxygen group compared to the no oxygen group (1948 vs. 1543 U/L; means ratio, 1.27; 95% confidence interval, 1.04-1.52; P=0.01). There were nonsignificant increases of secondary endpoints in the oxygen group, including rate of recurrent myocardial infarction (5.5% vs. 0.9%; P=0.006), frequency of arrhythmia (40.4% vs. 31.4%; P=0.05), and size of infarct on six-month cardiac MRI (n=139; 20.3 vs. 13.1 g; P=0.04).
This study has several limitations: It was powered to detect differences in biomarkers (not clinical endpoints) and the treatment was not blinded to paramedics, patients, or cardiology teams.
Bottom line: Supplemental oxygen administration in patients with STEMI might increase infarct size and lead to poorer clinical outcomes; however, larger clinical trials are warranted.
Citation: Stub D, Smith K, Bernard S, et al. Air versus oxygen in ST-segment-elevation myocardial infarction. Circulation. 2015;131(24):2143-2150.
Thrombectomy Within Eight Hours of Stroke Onset Reduces Post-Stroke Disability
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin score.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups.
The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptoms onset in ischemic stroke. New Engl J Med. 2015;372(24):2296-2306.
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin score.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups.
The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptoms onset in ischemic stroke. New Engl J Med. 2015;372(24):2296-2306.
Clinical question: Does thrombectomy, in conjunction with medical therapy, improve functional independence in patients with an acute proximal anterior stroke?
Background: Revascularization of proximal anterior strokes with alteplase alone occurs less than 50% of the time. First-generation thrombectomy devices (i.e., Merci and Penumbra) have not shown improvement in revascularization or functional outcomes; however, the development of thrombectomy stent retriever devices has led to more promising results, with several recent studies demonstrating functional improvement using endovascular retrieval in addition to medical therapy in proximal anterior circulation strokes.
Study design: Prospective, multicenter, randomized, sequential, open-label, phase 3 study with blinded evaluation.
Setting: Four hospitals in Spain.
Synopsis: Approximately 200 patients who were diagnosed within eight hours of onset of a large vessel anterior stroke were randomly assigned to medical therapy (alteplase) plus endovascular treatment versus medical therapy alone. In order to reduce selection bias, the study was conducted within a population-based registry of acute stroke patients from the same area. The major exclusion criterion was evidence of a large infarct on imaging. The primary outcome was severity of disability at 90 days based on the modified Rankin score.
Study results showed a significant improvement in functional status in the thrombectomy group, with 66% of patients demonstrating revascularization. The rate of death and intracranial hemorrhage was similar between both groups.
The trial stopped recruitment after the first interim analysis given lack of equipoise, with emerging literature supporting endovascular therapy.
Bottom line: Thrombectomy performed in proximal, large vessel anterior circulation strokes within eight hours of onset of symptoms improves functional status at 90 days.
Citation: Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptoms onset in ischemic stroke. New Engl J Med. 2015;372(24):2296-2306.
Interdisciplinary Team Care on General Medical Units Does Not Improve Patient Outcomes
Clinical question: Does interdisciplinary care on general medical wards improve patient outcomes?
Background: Patients on general medical wards might experience errors and/or preventable deaths. One mechanism that could reduce the risk of errors or preventable death is the utilization of interdisciplinary team care.
Study design: Systematic review.
Setting: Studies published in English from 1998 through 2013 in Embase, MEDLINE, and PsycINFO.
Synopsis: Reports of interdisciplinary team care interventions on general medical wards in which the care was evaluated against an objective patient outcome were reviewed. Outcomes were grouped into early (less than 30 days) or late (30 days to 12 months). Thirty studies of more than 66,000 total patients were included; these were composed of RCTs, nonrandomized cluster trials, and controlled before-after studies.
Interventions either altered the composition of the care team or addressed the logistics of team practice. Studies evaluated complications of care, length of stay, readmissions, and mortality. Although some evidence showed interdisciplinary care reduces complications, the majority of studies did not show significant improvements in any other outcomes.
Bottom line: In a systematic review, interdisciplinary team care in general medical wards is not associated with reduced complications, length of stay, readmissions, or mortality.
Citation: Pannick S, Davis R, Ashrafian H, et al. Effects of interdisciplinary team care interventions on general medical wards: a systematic review. JAMA Intern Med. 2015;175(8):1288-1298.
Clinical question: Does interdisciplinary care on general medical wards improve patient outcomes?
Background: Patients on general medical wards might experience errors and/or preventable deaths. One mechanism that could reduce the risk of errors or preventable death is the utilization of interdisciplinary team care.
Study design: Systematic review.
Setting: Studies published in English from 1998 through 2013 in Embase, MEDLINE, and PsycINFO.
Synopsis: Reports of interdisciplinary team care interventions on general medical wards in which the care was evaluated against an objective patient outcome were reviewed. Outcomes were grouped into early (less than 30 days) or late (30 days to 12 months). Thirty studies of more than 66,000 total patients were included; these were composed of RCTs, nonrandomized cluster trials, and controlled before-after studies.
Interventions either altered the composition of the care team or addressed the logistics of team practice. Studies evaluated complications of care, length of stay, readmissions, and mortality. Although some evidence showed interdisciplinary care reduces complications, the majority of studies did not show significant improvements in any other outcomes.
Bottom line: In a systematic review, interdisciplinary team care in general medical wards is not associated with reduced complications, length of stay, readmissions, or mortality.
Citation: Pannick S, Davis R, Ashrafian H, et al. Effects of interdisciplinary team care interventions on general medical wards: a systematic review. JAMA Intern Med. 2015;175(8):1288-1298.
Clinical question: Does interdisciplinary care on general medical wards improve patient outcomes?
Background: Patients on general medical wards might experience errors and/or preventable deaths. One mechanism that could reduce the risk of errors or preventable death is the utilization of interdisciplinary team care.
Study design: Systematic review.
Setting: Studies published in English from 1998 through 2013 in Embase, MEDLINE, and PsycINFO.
Synopsis: Reports of interdisciplinary team care interventions on general medical wards in which the care was evaluated against an objective patient outcome were reviewed. Outcomes were grouped into early (less than 30 days) or late (30 days to 12 months). Thirty studies of more than 66,000 total patients were included; these were composed of RCTs, nonrandomized cluster trials, and controlled before-after studies.
Interventions either altered the composition of the care team or addressed the logistics of team practice. Studies evaluated complications of care, length of stay, readmissions, and mortality. Although some evidence showed interdisciplinary care reduces complications, the majority of studies did not show significant improvements in any other outcomes.
Bottom line: In a systematic review, interdisciplinary team care in general medical wards is not associated with reduced complications, length of stay, readmissions, or mortality.
Citation: Pannick S, Davis R, Ashrafian H, et al. Effects of interdisciplinary team care interventions on general medical wards: a systematic review. JAMA Intern Med. 2015;175(8):1288-1298.
Patient Satisfaction Scores, Objective Measures of Surgical Quality Have Positive Association
Clinical question: Is there an association between objective measures of surgical quality and patient satisfaction as measured by the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey?
Background: The Centers for Medicare and Medicaid Services (CMS) has tied financial reimbursement to patient satisfaction scores. It is unknown whether high-quality surgery correlates with higher patient satisfaction scores.
Study design: Retrospective, observational study.
Setting: One hundred eighty hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP).
Synopsis: The study included 103,866 Medicare patients 65 and older who had surgery at a participating ACS NSQIP hospital between 2004-2008. Data regarding these patients were collected from a linked database (including Medicare inpatient claims, ACS NSQIP, the American Hospital Association annual survey, and Hospital Compare). Analysis of five 30-day outcomes was performed in order to assess surgical quality:
- Post-operative mortality;
- Major complication;
- Minor complication;
- Death following a complication; and
- Readmission.
Participating hospitals were grouped into quartiles based upon their performance on the HCAHPS survey.
Hospitals that performed in the highest quartile on the HCAHPS survey had significantly lower 30-day mortality, death following complications, and minor complications. No differences were detected in hospital readmissions or major complications based on patient satisfaction.
The results of this study may not be generalizable to all hospitals given the fact that the dataset is from Medicare patients only and participation in the ACS NSQIP is voluntary.
Bottom line: Using data from a national database, researchers found a positive association between patient satisfaction scores and objective measures of surgical quality.
Citation: Sacks GD, Lawson EH, Dawes AJ, et al. Relationship between hospital performance on a patient satisfaction survey and surgical quality [published online ahead of print June 24, 2015]. JAMA Surg. doi:10.1001/jamasurg.2015.1108.
Clinical question: Is there an association between objective measures of surgical quality and patient satisfaction as measured by the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey?
Background: The Centers for Medicare and Medicaid Services (CMS) has tied financial reimbursement to patient satisfaction scores. It is unknown whether high-quality surgery correlates with higher patient satisfaction scores.
Study design: Retrospective, observational study.
Setting: One hundred eighty hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP).
Synopsis: The study included 103,866 Medicare patients 65 and older who had surgery at a participating ACS NSQIP hospital between 2004-2008. Data regarding these patients were collected from a linked database (including Medicare inpatient claims, ACS NSQIP, the American Hospital Association annual survey, and Hospital Compare). Analysis of five 30-day outcomes was performed in order to assess surgical quality:
- Post-operative mortality;
- Major complication;
- Minor complication;
- Death following a complication; and
- Readmission.
Participating hospitals were grouped into quartiles based upon their performance on the HCAHPS survey.
Hospitals that performed in the highest quartile on the HCAHPS survey had significantly lower 30-day mortality, death following complications, and minor complications. No differences were detected in hospital readmissions or major complications based on patient satisfaction.
The results of this study may not be generalizable to all hospitals given the fact that the dataset is from Medicare patients only and participation in the ACS NSQIP is voluntary.
Bottom line: Using data from a national database, researchers found a positive association between patient satisfaction scores and objective measures of surgical quality.
Citation: Sacks GD, Lawson EH, Dawes AJ, et al. Relationship between hospital performance on a patient satisfaction survey and surgical quality [published online ahead of print June 24, 2015]. JAMA Surg. doi:10.1001/jamasurg.2015.1108.
Clinical question: Is there an association between objective measures of surgical quality and patient satisfaction as measured by the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey?
Background: The Centers for Medicare and Medicaid Services (CMS) has tied financial reimbursement to patient satisfaction scores. It is unknown whether high-quality surgery correlates with higher patient satisfaction scores.
Study design: Retrospective, observational study.
Setting: One hundred eighty hospitals participating in the American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP).
Synopsis: The study included 103,866 Medicare patients 65 and older who had surgery at a participating ACS NSQIP hospital between 2004-2008. Data regarding these patients were collected from a linked database (including Medicare inpatient claims, ACS NSQIP, the American Hospital Association annual survey, and Hospital Compare). Analysis of five 30-day outcomes was performed in order to assess surgical quality:
- Post-operative mortality;
- Major complication;
- Minor complication;
- Death following a complication; and
- Readmission.
Participating hospitals were grouped into quartiles based upon their performance on the HCAHPS survey.
Hospitals that performed in the highest quartile on the HCAHPS survey had significantly lower 30-day mortality, death following complications, and minor complications. No differences were detected in hospital readmissions or major complications based on patient satisfaction.
The results of this study may not be generalizable to all hospitals given the fact that the dataset is from Medicare patients only and participation in the ACS NSQIP is voluntary.
Bottom line: Using data from a national database, researchers found a positive association between patient satisfaction scores and objective measures of surgical quality.
Citation: Sacks GD, Lawson EH, Dawes AJ, et al. Relationship between hospital performance on a patient satisfaction survey and surgical quality [published online ahead of print June 24, 2015]. JAMA Surg. doi:10.1001/jamasurg.2015.1108.
Extended Anticoagulation Therapy and Recurrent Rates of Venous Thromboembolism
Clinical question: Among patients with a first episode of unprovoked pulmonary embolism (PE), what are the benefits and harms of extending the duration of anticoagulation for secondary prophylaxis against recurrent VTE?
Background: Optimal duration of anticoagulation after initial unprovoked PE is not known. Prior studies demonstrated risk reduction of recurrent VTE while on therapy but have had inadequate long-term monitoring of patients or enrollment of patients with PE, who are known to have a higher case-fatality rate of recurrent VTE than patients with DVT.
Study design: Multicenter, randomized, double-blinded, parallel-grouped, placebo-controlled trial.
Setting: Fourteen French hospitals from 2007 to 2012.
Synopsis: Investigators randomized 371 patients with a first episode of symptomatic unprovoked PE who had completed six months of warfarin to 18 additional months of warfarin or placebo. Patients were followed for 24 months after discontinuation of therapy.
During the treatment period, the primary outcome (composite of recurrent VTE and major bleeding) occurred in 3.3% of the warfarin group vs. 13.5% of the placebo group (HR 0.22). This difference was primarily due to reduction in risk of recurrent VTE (1.7% vs. 13.5%, HR 0.15), with only minimal increased bleeding risk (2.2% vs. 0.5%, NS).
There was no significant difference in the composite outcome (20.8% in warfarin group vs. 24% in placebo) on analysis of the entire study period (treatment and follow-up), however, suggesting the benefit of extended warfarin therapy upon recurrent VTE risk diminished upon cessation.
Bottom line: Patients treated with extended-therapy warfarin after a first unprovoked PE have a decreased risk of recurrent VTE compared to standard therapy only while on treatment; the risk of recurrent VTE returns upon cessation of therapy.
Citation: Couturand F, Sanchez O, Pernod G, et al. Six months vs extended oral anticoagulation after a first episode of pulmonary embolism: The PADIS-PE randomized clinical trial. JAMA. 2015;314(1):31-40.
Clinical question: Among patients with a first episode of unprovoked pulmonary embolism (PE), what are the benefits and harms of extending the duration of anticoagulation for secondary prophylaxis against recurrent VTE?
Background: Optimal duration of anticoagulation after initial unprovoked PE is not known. Prior studies demonstrated risk reduction of recurrent VTE while on therapy but have had inadequate long-term monitoring of patients or enrollment of patients with PE, who are known to have a higher case-fatality rate of recurrent VTE than patients with DVT.
Study design: Multicenter, randomized, double-blinded, parallel-grouped, placebo-controlled trial.
Setting: Fourteen French hospitals from 2007 to 2012.
Synopsis: Investigators randomized 371 patients with a first episode of symptomatic unprovoked PE who had completed six months of warfarin to 18 additional months of warfarin or placebo. Patients were followed for 24 months after discontinuation of therapy.
During the treatment period, the primary outcome (composite of recurrent VTE and major bleeding) occurred in 3.3% of the warfarin group vs. 13.5% of the placebo group (HR 0.22). This difference was primarily due to reduction in risk of recurrent VTE (1.7% vs. 13.5%, HR 0.15), with only minimal increased bleeding risk (2.2% vs. 0.5%, NS).
There was no significant difference in the composite outcome (20.8% in warfarin group vs. 24% in placebo) on analysis of the entire study period (treatment and follow-up), however, suggesting the benefit of extended warfarin therapy upon recurrent VTE risk diminished upon cessation.
Bottom line: Patients treated with extended-therapy warfarin after a first unprovoked PE have a decreased risk of recurrent VTE compared to standard therapy only while on treatment; the risk of recurrent VTE returns upon cessation of therapy.
Citation: Couturand F, Sanchez O, Pernod G, et al. Six months vs extended oral anticoagulation after a first episode of pulmonary embolism: The PADIS-PE randomized clinical trial. JAMA. 2015;314(1):31-40.
Clinical question: Among patients with a first episode of unprovoked pulmonary embolism (PE), what are the benefits and harms of extending the duration of anticoagulation for secondary prophylaxis against recurrent VTE?
Background: Optimal duration of anticoagulation after initial unprovoked PE is not known. Prior studies demonstrated risk reduction of recurrent VTE while on therapy but have had inadequate long-term monitoring of patients or enrollment of patients with PE, who are known to have a higher case-fatality rate of recurrent VTE than patients with DVT.
Study design: Multicenter, randomized, double-blinded, parallel-grouped, placebo-controlled trial.
Setting: Fourteen French hospitals from 2007 to 2012.
Synopsis: Investigators randomized 371 patients with a first episode of symptomatic unprovoked PE who had completed six months of warfarin to 18 additional months of warfarin or placebo. Patients were followed for 24 months after discontinuation of therapy.
During the treatment period, the primary outcome (composite of recurrent VTE and major bleeding) occurred in 3.3% of the warfarin group vs. 13.5% of the placebo group (HR 0.22). This difference was primarily due to reduction in risk of recurrent VTE (1.7% vs. 13.5%, HR 0.15), with only minimal increased bleeding risk (2.2% vs. 0.5%, NS).
There was no significant difference in the composite outcome (20.8% in warfarin group vs. 24% in placebo) on analysis of the entire study period (treatment and follow-up), however, suggesting the benefit of extended warfarin therapy upon recurrent VTE risk diminished upon cessation.
Bottom line: Patients treated with extended-therapy warfarin after a first unprovoked PE have a decreased risk of recurrent VTE compared to standard therapy only while on treatment; the risk of recurrent VTE returns upon cessation of therapy.
Citation: Couturand F, Sanchez O, Pernod G, et al. Six months vs extended oral anticoagulation after a first episode of pulmonary embolism: The PADIS-PE randomized clinical trial. JAMA. 2015;314(1):31-40.