User login
At 5% to 6%, mantle cell lymphoma (MCL) is diagnosed in a relatively small proportion of patients with non-Hodgkin lymphoma. However, MCL is important to recognize because of its relatively poorer prognosis and the important role of autologous hematopoietic stem cell transplantation (HCT) as an adjunct to first-line treatment and, to a lesser extent, in later lines of therapy.
Treatment Options
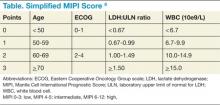
Though pathologic features are beyond the scope of this manuscript, when a definitive diagnosis is made, it is important to differentiate the more aggressive blastoid variant from the more typical pathologic patterns. The indolent form of MCL is diagnosed by clinical presentation as described below. In addition, quantitation of Ki-67 can add prognostic value.1-6 Patients with tumors that express higher levels of Ki-67 have higher relapse rates and shorter overall survivals.2,3,5 The Mantle Cell Lymphoma International Prognostic Index (MIPI) segregates patients into low-, intermediate-, and high-risk groups based on the clinical factors of patient age, performance status, serum lactate dehydrogenase, and total white blood cell count (WBC) (Table). Use of the MIPI at both initial diagnosis and before first-line autologous HCT can also offer significant prognostic value.1-7 Patients with higher MIPI scores have shorter overall survivals.7,8
For patients who present with indolent clinical features such as a stable leukemic phase, splenomegaly without adenopathy, and low tumor burden, watchful waiting can be utilized. However, approximately 80% of patients will require initial treatment with chemotherapy.1-6
For younger patients and those with good performance status and physiologic reserve, randomized trials have not clearly identified a preferred initial regimen, though initial therapy is typically with the hyperCVAD regimen along with the addition of rituximab.5,9 This regimen is fairly aggressive, requires inpatient hospitalization, is associated with cytopenia and risk of infection, and has not been rigorously proven to be superior in prospective randomized studies, but based on select single-arm studies and retrospective controls, this is commonly used as first-line therapy in the U.S.5,9
Of note, the recent SWOG 1106 U.S. Intergroup study comparing initial therapy with R-hyperCVAD to rituximab+bendamustine was closed early due to poor peripheral blood stem cell (PBSC) mobilization in the R-hyperCVAD arm. R-CHOP or R-bendamustine are considered less aggressive alternative regimens for older patients and for those with a poorer overall performance status.
For younger patients, the incorporation of high-dose cytarabine in various combinations during induction has been consistently identified as superior to those regimens without high-dose cytarabine. In general, the comparative studies have rather complex treatment regimens and are not routinely used in the U.S.
Other drugs with proven activity, though currently without a clear therapeutic sequence, include bortezomib, lenalidomide, bendamustine, temsirolimus, and most recently ibrutinib.1-5
Autologous HCT Recommendations
Following initial chemotherapy, autologous HCT is recommended for patients aged < 65 years and with good performance status. Earlier single-arm trials showed that the addition of dose intensification with autologous HCT led to more durable remissions. Both the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) support dose intensification with autologous HCT in first remission.
There are no absolute age restrictions for autologous HCT, though patients must have adequate physiologic reserve and good overall performance status. Prognostic physiologic parameters are not as well characterized for autologous HCT as they are for reduced-intensity allogeneic transplantation and autologous HCT for multiple myeloma. However, risk indices are being developed for patients with non-Hodgkin lymphoma undergoing autologous HCT.
While the addition of rituximab has improved the overall response rate of all chemotherapy regimens in patients with MCL, the most convincing survival plateaus still occur with autologous HCT in first remission. Nonetheless, the best first-line therapy has not been proven in prospective randomized fashion.1-6,9-13
Single-arm studies have shown that the R-HyperCVAD regimen can induce complete responses of 58% to 87% of first-line patients.1-3,5 From a practical perspective, for patients receiving R-hyperCVAD and proceeding with autologous HCT, PBSC are typically harvested after the completion of cycle 1B and patients proceed with autologous HCT after cycle 2B.5,9
The best preparative regimen for autologous HCT has not been clearly identified. Options for dose intensification include the more traditional total body irradiation (TBI)-based regimen as well as chemotherapy only, such as the BEAM (carmustine, etoposide, cytarabine, melphalan) regimen. While there are no comparative studies, a small retrospective analysis suggested benefit for a TBI-based preparative regimen, with a larger and recent European Group for Blood and Marrow Transplantation (EBMT) review suggesting that the benefit of TBI may be limited to those patients who have not achieved complete remission (CR) before autologous HCT.3
Mantle cell lymphoma is known to be a radiosensitive malignancy, and the use of radioimmunotherapy (RIT) along with HCT has shown promising results in single-arm studies when compared with historical control groups. The current unavailability of radioiodine-based RIT (tositumomab) and the unproven benefit of yttrium-based RIT (ibritumomab tiuxetan) makes this approach still of uncertain benefit. Nonetheless, the suggestion of benefit based on retrospective case control studies suggests that the addition of RIT to autologous HCT for MCL is worthy of further investigation.
After remission induction by initial therapy, maintenance rituximab therapy has been evaluated for patients who have received chemotherapy only or those with chemotherapy and autologous HCT.
Currently, the only prospective trial showing overall survival (OS) benefit is in the nontransplant setting following R-CHOP or R-FC (rituximab/fludarabine/cyclophosphamide) chemotherapy performed by the European Mantle Cell Lymphoma Network. This study showed a 4-year OS of 87% for those receiving rituximab maintenance compared with 63% for those receiving interferon alpha maintenance.14
In the autologous HCT setting, support for rituximab maintenance therapy comes from a number of sources. The CALGB 59909 study was a single-arm study showing the efficacy of rituximab along with induction therapy and dose-intensive therapy with autologous HCT followed by a short course of rituximab maintenance. This study showed the feasibility of additional rituximab with 2-year and 5-year PFS of 76% and 56%, respectively.11
Using a preemptive approach, the Nordic MCL-2 study showed both feasibility and a suggestion of delayed time to clinical relapse for intervention with rituximab in those patients who showed molecular relapse. In this study, molecular relapse was defined by increasing PCR-detectable markers following induction and autologous HCT using a BEAM transplant regimen.15 The prospective randomized French GOELAMS LyMa trial compared rituximab maintenance therapy for 3 years compared with no further therapy following first-line autologous HCT. This trial has recently closed and the results have not yet been presented.
While we currently await results of the LyMa trial, it is not possible to uniformly recommend rituximab maintenance to all patients following autologous HCT. Nonetheless, the Nordic MCL-2 study with intervention for molecular relapse and the demonstrated benefit in the nontransplant setting in older patients are compelling, and the generally well-tolerated administration of rituximab, all suggest consideration of rituximab maintenance in select patients until the outcome of the LyMa study is available for review.
Other agents that have demonstrated activity in MCL and have been considered as maintenance following autologous HCT include bortezomib, lenalidomide, and ibrutinib, with lenalidomide being currently studied by the Italian Lymphoma Foundation.
Other Considerations
For those patients who relapse following initial chemotherapy, autologous HCT can be considered following effective debulking chemotherapy. While historically, this group of patients was considered incurable with either chemotherapy or autologous HCT, newer evidence suggests that certain subsets of those patients can be effectively treated with autologous HCT.16 Depending on the number of adverse factors identified, the 5-year progression-free and overall survivals can range from 58% to 15% and from 76% to 32%, respectively.
For those patients who relapse following front-line autologous HCT, select patients with responsive disease, good performance status, and an available donor can be considered for reduced-intensity allogeneic transplantation.17
With the addition of new drugs and potential combinations, it is possible that dose intensification with autologous HCT will come to play a smaller role in the overall therapy of patients with MCL. However, this will require careful assessment in prospective randomized trials, along with better identification of specific patient subsets as well as a more thorough understanding of molecular prognostic and predictive factors.
For patients beyond first remission, autologous HCT can still be of value in those without prior HCT, and in select situations, reduced-intensity allogeneic transplantation also can be considered. Given all these issues, it is strongly encouraged that treating physicians work in concert with HCT programs soon after initial diagnosis so that decisions regarding initial therapy and timing of transplantation can be optimized.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Dreyling M, Ferrero S, Hermine O. How I manage mantle cell lymphoma [published online ahead of print May 23, 2014]. Leukemia.
2. Gordon LI, Bernstein SH, Jares P, Kahl BS, Witzig TE, Dreyling M. Recent advances in mantle cell lymphoma: Report of the 2013 Mantle Cell Lymphoma Consortium Workshop [published online ahead of print April 17, 2014]. Leuk Lymphoma.
3. Dreyling M, Kluin-Nelemans HC, Beà S, et al; European MCL Network. Update on the molecular pathogenesis and clinical treatment of mantle cell lymphoma: Report of the 11th annual conference of the European Mantle Cell Lymphoma Network. Leuk Lymphoma. 2013;54(4):699-707.
4. Dreyling M, Thieblemont C, Gallamini A, et al. ESMO Consensus conferences: Guidelines on malignant lymphoma. Part 2: Marginal zone lymphoma, mantle cell lymphoma, peripheral T-cell lymphoma. Ann Oncol. 2013;24(4):857-877.
5. Williams ME. Transplantation for mantle cell lymphoma: Is it the right thing to do? Hematology Am Soc Hematol Educ Program. 2013;2013(1):568-574.
6. Dreyling M, Hiddemann W; European MCL Network. Current treatment standards and emerging strategies in mantle cell lymphoma. Hematology Am Soc Hematol Educ Program. 2009;2009(1):542-551.
7. Budde LE, Guthrie KA, Till BG, et al. Mantle cell lymphoma international prognostic index but not pretransplantation induction regimen predicts survival for patients with mantle-cell lymphoma receiving high-dose therapy and autologous stem-cell transplantation. J Clin Oncol. 2011;29(22):3023-3029.
8. Hoster E, Dreyling M, Klapper W, et al; German Low Grade Lymphoma Study Group (GLSG), European MCL Network. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood. 2008;111(2):558-565.
9. Geisler CH, Kolstad A, Laurell A, et al; Nordic Lymphoma Group. Long-term progression-free survival of mantle cell lymphoma after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: A nonrandomized phase 2 multicenter study by the Nordic Lymphoma Group. Blood. 2008;112(7):2687-2693.
10. Delarue R, Haioun C, Ribrag V, et al; Groupe d’Etude des Lymphomes de l’Adulte (GELA). CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: A phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood. 2013;121(1):48-53.
11. Damon LE, Johnson JL, Niedzwiecki D, et al. Immunochemotherapy and autologous stem-cell transplantation for untreated patients with mantle-cell lymphoma: CALGB 59909. J Clin Oncol. 2009;27(36):6101-6108.
12. Dreyling M, Lenz G, Hoster E, et al. Early consolidation by myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission significantly prolongs progression-free survival in mantle-cell lymphoma:Results of a prospective randomized trial of the European MCL Network. Blood. 2005;105(7):2677-2684.
13. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: Results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol. 2005;23(9):1984-1992.
14. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med. 2012;367(6):520-531.
15. Andersen NS, Pedersen LB, Laurell A, et al. Pre-emptive treatment with rituximab of molecular relapse after autologous stem cell transplantation in mantle cell lymphoma. J Clin Oncol. 2009;27(26):4365-4370.
16. Cassaday RD, Guthrie KA, Budde EL, et al. Specific features identify patients with relapsed or refractory mantle cell lymphoma benefitting from autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2013;19(9):1403-1406.
17. Maris MB, Sandmaier BM, Storer BE, et al. Allogeneic hematopoietic cell transplantation after fludarabine and 2 Gy total body irradiation for relapsed and refractory mantle cell lymphoma. Blood. 2004;104(12):3535-3542.
At 5% to 6%, mantle cell lymphoma (MCL) is diagnosed in a relatively small proportion of patients with non-Hodgkin lymphoma. However, MCL is important to recognize because of its relatively poorer prognosis and the important role of autologous hematopoietic stem cell transplantation (HCT) as an adjunct to first-line treatment and, to a lesser extent, in later lines of therapy.
Treatment Options
Though pathologic features are beyond the scope of this manuscript, when a definitive diagnosis is made, it is important to differentiate the more aggressive blastoid variant from the more typical pathologic patterns. The indolent form of MCL is diagnosed by clinical presentation as described below. In addition, quantitation of Ki-67 can add prognostic value.1-6 Patients with tumors that express higher levels of Ki-67 have higher relapse rates and shorter overall survivals.2,3,5 The Mantle Cell Lymphoma International Prognostic Index (MIPI) segregates patients into low-, intermediate-, and high-risk groups based on the clinical factors of patient age, performance status, serum lactate dehydrogenase, and total white blood cell count (WBC) (Table). Use of the MIPI at both initial diagnosis and before first-line autologous HCT can also offer significant prognostic value.1-7 Patients with higher MIPI scores have shorter overall survivals.7,8
For patients who present with indolent clinical features such as a stable leukemic phase, splenomegaly without adenopathy, and low tumor burden, watchful waiting can be utilized. However, approximately 80% of patients will require initial treatment with chemotherapy.1-6
For younger patients and those with good performance status and physiologic reserve, randomized trials have not clearly identified a preferred initial regimen, though initial therapy is typically with the hyperCVAD regimen along with the addition of rituximab.5,9 This regimen is fairly aggressive, requires inpatient hospitalization, is associated with cytopenia and risk of infection, and has not been rigorously proven to be superior in prospective randomized studies, but based on select single-arm studies and retrospective controls, this is commonly used as first-line therapy in the U.S.5,9
Of note, the recent SWOG 1106 U.S. Intergroup study comparing initial therapy with R-hyperCVAD to rituximab+bendamustine was closed early due to poor peripheral blood stem cell (PBSC) mobilization in the R-hyperCVAD arm. R-CHOP or R-bendamustine are considered less aggressive alternative regimens for older patients and for those with a poorer overall performance status.
For younger patients, the incorporation of high-dose cytarabine in various combinations during induction has been consistently identified as superior to those regimens without high-dose cytarabine. In general, the comparative studies have rather complex treatment regimens and are not routinely used in the U.S.
Other drugs with proven activity, though currently without a clear therapeutic sequence, include bortezomib, lenalidomide, bendamustine, temsirolimus, and most recently ibrutinib.1-5
Autologous HCT Recommendations
Following initial chemotherapy, autologous HCT is recommended for patients aged < 65 years and with good performance status. Earlier single-arm trials showed that the addition of dose intensification with autologous HCT led to more durable remissions. Both the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) support dose intensification with autologous HCT in first remission.
There are no absolute age restrictions for autologous HCT, though patients must have adequate physiologic reserve and good overall performance status. Prognostic physiologic parameters are not as well characterized for autologous HCT as they are for reduced-intensity allogeneic transplantation and autologous HCT for multiple myeloma. However, risk indices are being developed for patients with non-Hodgkin lymphoma undergoing autologous HCT.
While the addition of rituximab has improved the overall response rate of all chemotherapy regimens in patients with MCL, the most convincing survival plateaus still occur with autologous HCT in first remission. Nonetheless, the best first-line therapy has not been proven in prospective randomized fashion.1-6,9-13
Single-arm studies have shown that the R-HyperCVAD regimen can induce complete responses of 58% to 87% of first-line patients.1-3,5 From a practical perspective, for patients receiving R-hyperCVAD and proceeding with autologous HCT, PBSC are typically harvested after the completion of cycle 1B and patients proceed with autologous HCT after cycle 2B.5,9
The best preparative regimen for autologous HCT has not been clearly identified. Options for dose intensification include the more traditional total body irradiation (TBI)-based regimen as well as chemotherapy only, such as the BEAM (carmustine, etoposide, cytarabine, melphalan) regimen. While there are no comparative studies, a small retrospective analysis suggested benefit for a TBI-based preparative regimen, with a larger and recent European Group for Blood and Marrow Transplantation (EBMT) review suggesting that the benefit of TBI may be limited to those patients who have not achieved complete remission (CR) before autologous HCT.3
Mantle cell lymphoma is known to be a radiosensitive malignancy, and the use of radioimmunotherapy (RIT) along with HCT has shown promising results in single-arm studies when compared with historical control groups. The current unavailability of radioiodine-based RIT (tositumomab) and the unproven benefit of yttrium-based RIT (ibritumomab tiuxetan) makes this approach still of uncertain benefit. Nonetheless, the suggestion of benefit based on retrospective case control studies suggests that the addition of RIT to autologous HCT for MCL is worthy of further investigation.
After remission induction by initial therapy, maintenance rituximab therapy has been evaluated for patients who have received chemotherapy only or those with chemotherapy and autologous HCT.
Currently, the only prospective trial showing overall survival (OS) benefit is in the nontransplant setting following R-CHOP or R-FC (rituximab/fludarabine/cyclophosphamide) chemotherapy performed by the European Mantle Cell Lymphoma Network. This study showed a 4-year OS of 87% for those receiving rituximab maintenance compared with 63% for those receiving interferon alpha maintenance.14
In the autologous HCT setting, support for rituximab maintenance therapy comes from a number of sources. The CALGB 59909 study was a single-arm study showing the efficacy of rituximab along with induction therapy and dose-intensive therapy with autologous HCT followed by a short course of rituximab maintenance. This study showed the feasibility of additional rituximab with 2-year and 5-year PFS of 76% and 56%, respectively.11
Using a preemptive approach, the Nordic MCL-2 study showed both feasibility and a suggestion of delayed time to clinical relapse for intervention with rituximab in those patients who showed molecular relapse. In this study, molecular relapse was defined by increasing PCR-detectable markers following induction and autologous HCT using a BEAM transplant regimen.15 The prospective randomized French GOELAMS LyMa trial compared rituximab maintenance therapy for 3 years compared with no further therapy following first-line autologous HCT. This trial has recently closed and the results have not yet been presented.
While we currently await results of the LyMa trial, it is not possible to uniformly recommend rituximab maintenance to all patients following autologous HCT. Nonetheless, the Nordic MCL-2 study with intervention for molecular relapse and the demonstrated benefit in the nontransplant setting in older patients are compelling, and the generally well-tolerated administration of rituximab, all suggest consideration of rituximab maintenance in select patients until the outcome of the LyMa study is available for review.
Other agents that have demonstrated activity in MCL and have been considered as maintenance following autologous HCT include bortezomib, lenalidomide, and ibrutinib, with lenalidomide being currently studied by the Italian Lymphoma Foundation.
Other Considerations
For those patients who relapse following initial chemotherapy, autologous HCT can be considered following effective debulking chemotherapy. While historically, this group of patients was considered incurable with either chemotherapy or autologous HCT, newer evidence suggests that certain subsets of those patients can be effectively treated with autologous HCT.16 Depending on the number of adverse factors identified, the 5-year progression-free and overall survivals can range from 58% to 15% and from 76% to 32%, respectively.
For those patients who relapse following front-line autologous HCT, select patients with responsive disease, good performance status, and an available donor can be considered for reduced-intensity allogeneic transplantation.17
With the addition of new drugs and potential combinations, it is possible that dose intensification with autologous HCT will come to play a smaller role in the overall therapy of patients with MCL. However, this will require careful assessment in prospective randomized trials, along with better identification of specific patient subsets as well as a more thorough understanding of molecular prognostic and predictive factors.
For patients beyond first remission, autologous HCT can still be of value in those without prior HCT, and in select situations, reduced-intensity allogeneic transplantation also can be considered. Given all these issues, it is strongly encouraged that treating physicians work in concert with HCT programs soon after initial diagnosis so that decisions regarding initial therapy and timing of transplantation can be optimized.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
At 5% to 6%, mantle cell lymphoma (MCL) is diagnosed in a relatively small proportion of patients with non-Hodgkin lymphoma. However, MCL is important to recognize because of its relatively poorer prognosis and the important role of autologous hematopoietic stem cell transplantation (HCT) as an adjunct to first-line treatment and, to a lesser extent, in later lines of therapy.
Treatment Options
Though pathologic features are beyond the scope of this manuscript, when a definitive diagnosis is made, it is important to differentiate the more aggressive blastoid variant from the more typical pathologic patterns. The indolent form of MCL is diagnosed by clinical presentation as described below. In addition, quantitation of Ki-67 can add prognostic value.1-6 Patients with tumors that express higher levels of Ki-67 have higher relapse rates and shorter overall survivals.2,3,5 The Mantle Cell Lymphoma International Prognostic Index (MIPI) segregates patients into low-, intermediate-, and high-risk groups based on the clinical factors of patient age, performance status, serum lactate dehydrogenase, and total white blood cell count (WBC) (Table). Use of the MIPI at both initial diagnosis and before first-line autologous HCT can also offer significant prognostic value.1-7 Patients with higher MIPI scores have shorter overall survivals.7,8
For patients who present with indolent clinical features such as a stable leukemic phase, splenomegaly without adenopathy, and low tumor burden, watchful waiting can be utilized. However, approximately 80% of patients will require initial treatment with chemotherapy.1-6
For younger patients and those with good performance status and physiologic reserve, randomized trials have not clearly identified a preferred initial regimen, though initial therapy is typically with the hyperCVAD regimen along with the addition of rituximab.5,9 This regimen is fairly aggressive, requires inpatient hospitalization, is associated with cytopenia and risk of infection, and has not been rigorously proven to be superior in prospective randomized studies, but based on select single-arm studies and retrospective controls, this is commonly used as first-line therapy in the U.S.5,9
Of note, the recent SWOG 1106 U.S. Intergroup study comparing initial therapy with R-hyperCVAD to rituximab+bendamustine was closed early due to poor peripheral blood stem cell (PBSC) mobilization in the R-hyperCVAD arm. R-CHOP or R-bendamustine are considered less aggressive alternative regimens for older patients and for those with a poorer overall performance status.
For younger patients, the incorporation of high-dose cytarabine in various combinations during induction has been consistently identified as superior to those regimens without high-dose cytarabine. In general, the comparative studies have rather complex treatment regimens and are not routinely used in the U.S.
Other drugs with proven activity, though currently without a clear therapeutic sequence, include bortezomib, lenalidomide, bendamustine, temsirolimus, and most recently ibrutinib.1-5
Autologous HCT Recommendations
Following initial chemotherapy, autologous HCT is recommended for patients aged < 65 years and with good performance status. Earlier single-arm trials showed that the addition of dose intensification with autologous HCT led to more durable remissions. Both the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) support dose intensification with autologous HCT in first remission.
There are no absolute age restrictions for autologous HCT, though patients must have adequate physiologic reserve and good overall performance status. Prognostic physiologic parameters are not as well characterized for autologous HCT as they are for reduced-intensity allogeneic transplantation and autologous HCT for multiple myeloma. However, risk indices are being developed for patients with non-Hodgkin lymphoma undergoing autologous HCT.
While the addition of rituximab has improved the overall response rate of all chemotherapy regimens in patients with MCL, the most convincing survival plateaus still occur with autologous HCT in first remission. Nonetheless, the best first-line therapy has not been proven in prospective randomized fashion.1-6,9-13
Single-arm studies have shown that the R-HyperCVAD regimen can induce complete responses of 58% to 87% of first-line patients.1-3,5 From a practical perspective, for patients receiving R-hyperCVAD and proceeding with autologous HCT, PBSC are typically harvested after the completion of cycle 1B and patients proceed with autologous HCT after cycle 2B.5,9
The best preparative regimen for autologous HCT has not been clearly identified. Options for dose intensification include the more traditional total body irradiation (TBI)-based regimen as well as chemotherapy only, such as the BEAM (carmustine, etoposide, cytarabine, melphalan) regimen. While there are no comparative studies, a small retrospective analysis suggested benefit for a TBI-based preparative regimen, with a larger and recent European Group for Blood and Marrow Transplantation (EBMT) review suggesting that the benefit of TBI may be limited to those patients who have not achieved complete remission (CR) before autologous HCT.3
Mantle cell lymphoma is known to be a radiosensitive malignancy, and the use of radioimmunotherapy (RIT) along with HCT has shown promising results in single-arm studies when compared with historical control groups. The current unavailability of radioiodine-based RIT (tositumomab) and the unproven benefit of yttrium-based RIT (ibritumomab tiuxetan) makes this approach still of uncertain benefit. Nonetheless, the suggestion of benefit based on retrospective case control studies suggests that the addition of RIT to autologous HCT for MCL is worthy of further investigation.
After remission induction by initial therapy, maintenance rituximab therapy has been evaluated for patients who have received chemotherapy only or those with chemotherapy and autologous HCT.
Currently, the only prospective trial showing overall survival (OS) benefit is in the nontransplant setting following R-CHOP or R-FC (rituximab/fludarabine/cyclophosphamide) chemotherapy performed by the European Mantle Cell Lymphoma Network. This study showed a 4-year OS of 87% for those receiving rituximab maintenance compared with 63% for those receiving interferon alpha maintenance.14
In the autologous HCT setting, support for rituximab maintenance therapy comes from a number of sources. The CALGB 59909 study was a single-arm study showing the efficacy of rituximab along with induction therapy and dose-intensive therapy with autologous HCT followed by a short course of rituximab maintenance. This study showed the feasibility of additional rituximab with 2-year and 5-year PFS of 76% and 56%, respectively.11
Using a preemptive approach, the Nordic MCL-2 study showed both feasibility and a suggestion of delayed time to clinical relapse for intervention with rituximab in those patients who showed molecular relapse. In this study, molecular relapse was defined by increasing PCR-detectable markers following induction and autologous HCT using a BEAM transplant regimen.15 The prospective randomized French GOELAMS LyMa trial compared rituximab maintenance therapy for 3 years compared with no further therapy following first-line autologous HCT. This trial has recently closed and the results have not yet been presented.
While we currently await results of the LyMa trial, it is not possible to uniformly recommend rituximab maintenance to all patients following autologous HCT. Nonetheless, the Nordic MCL-2 study with intervention for molecular relapse and the demonstrated benefit in the nontransplant setting in older patients are compelling, and the generally well-tolerated administration of rituximab, all suggest consideration of rituximab maintenance in select patients until the outcome of the LyMa study is available for review.
Other agents that have demonstrated activity in MCL and have been considered as maintenance following autologous HCT include bortezomib, lenalidomide, and ibrutinib, with lenalidomide being currently studied by the Italian Lymphoma Foundation.
Other Considerations
For those patients who relapse following initial chemotherapy, autologous HCT can be considered following effective debulking chemotherapy. While historically, this group of patients was considered incurable with either chemotherapy or autologous HCT, newer evidence suggests that certain subsets of those patients can be effectively treated with autologous HCT.16 Depending on the number of adverse factors identified, the 5-year progression-free and overall survivals can range from 58% to 15% and from 76% to 32%, respectively.
For those patients who relapse following front-line autologous HCT, select patients with responsive disease, good performance status, and an available donor can be considered for reduced-intensity allogeneic transplantation.17
With the addition of new drugs and potential combinations, it is possible that dose intensification with autologous HCT will come to play a smaller role in the overall therapy of patients with MCL. However, this will require careful assessment in prospective randomized trials, along with better identification of specific patient subsets as well as a more thorough understanding of molecular prognostic and predictive factors.
For patients beyond first remission, autologous HCT can still be of value in those without prior HCT, and in select situations, reduced-intensity allogeneic transplantation also can be considered. Given all these issues, it is strongly encouraged that treating physicians work in concert with HCT programs soon after initial diagnosis so that decisions regarding initial therapy and timing of transplantation can be optimized.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Dreyling M, Ferrero S, Hermine O. How I manage mantle cell lymphoma [published online ahead of print May 23, 2014]. Leukemia.
2. Gordon LI, Bernstein SH, Jares P, Kahl BS, Witzig TE, Dreyling M. Recent advances in mantle cell lymphoma: Report of the 2013 Mantle Cell Lymphoma Consortium Workshop [published online ahead of print April 17, 2014]. Leuk Lymphoma.
3. Dreyling M, Kluin-Nelemans HC, Beà S, et al; European MCL Network. Update on the molecular pathogenesis and clinical treatment of mantle cell lymphoma: Report of the 11th annual conference of the European Mantle Cell Lymphoma Network. Leuk Lymphoma. 2013;54(4):699-707.
4. Dreyling M, Thieblemont C, Gallamini A, et al. ESMO Consensus conferences: Guidelines on malignant lymphoma. Part 2: Marginal zone lymphoma, mantle cell lymphoma, peripheral T-cell lymphoma. Ann Oncol. 2013;24(4):857-877.
5. Williams ME. Transplantation for mantle cell lymphoma: Is it the right thing to do? Hematology Am Soc Hematol Educ Program. 2013;2013(1):568-574.
6. Dreyling M, Hiddemann W; European MCL Network. Current treatment standards and emerging strategies in mantle cell lymphoma. Hematology Am Soc Hematol Educ Program. 2009;2009(1):542-551.
7. Budde LE, Guthrie KA, Till BG, et al. Mantle cell lymphoma international prognostic index but not pretransplantation induction regimen predicts survival for patients with mantle-cell lymphoma receiving high-dose therapy and autologous stem-cell transplantation. J Clin Oncol. 2011;29(22):3023-3029.
8. Hoster E, Dreyling M, Klapper W, et al; German Low Grade Lymphoma Study Group (GLSG), European MCL Network. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood. 2008;111(2):558-565.
9. Geisler CH, Kolstad A, Laurell A, et al; Nordic Lymphoma Group. Long-term progression-free survival of mantle cell lymphoma after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: A nonrandomized phase 2 multicenter study by the Nordic Lymphoma Group. Blood. 2008;112(7):2687-2693.
10. Delarue R, Haioun C, Ribrag V, et al; Groupe d’Etude des Lymphomes de l’Adulte (GELA). CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: A phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood. 2013;121(1):48-53.
11. Damon LE, Johnson JL, Niedzwiecki D, et al. Immunochemotherapy and autologous stem-cell transplantation for untreated patients with mantle-cell lymphoma: CALGB 59909. J Clin Oncol. 2009;27(36):6101-6108.
12. Dreyling M, Lenz G, Hoster E, et al. Early consolidation by myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission significantly prolongs progression-free survival in mantle-cell lymphoma:Results of a prospective randomized trial of the European MCL Network. Blood. 2005;105(7):2677-2684.
13. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: Results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol. 2005;23(9):1984-1992.
14. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med. 2012;367(6):520-531.
15. Andersen NS, Pedersen LB, Laurell A, et al. Pre-emptive treatment with rituximab of molecular relapse after autologous stem cell transplantation in mantle cell lymphoma. J Clin Oncol. 2009;27(26):4365-4370.
16. Cassaday RD, Guthrie KA, Budde EL, et al. Specific features identify patients with relapsed or refractory mantle cell lymphoma benefitting from autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2013;19(9):1403-1406.
17. Maris MB, Sandmaier BM, Storer BE, et al. Allogeneic hematopoietic cell transplantation after fludarabine and 2 Gy total body irradiation for relapsed and refractory mantle cell lymphoma. Blood. 2004;104(12):3535-3542.
1. Dreyling M, Ferrero S, Hermine O. How I manage mantle cell lymphoma [published online ahead of print May 23, 2014]. Leukemia.
2. Gordon LI, Bernstein SH, Jares P, Kahl BS, Witzig TE, Dreyling M. Recent advances in mantle cell lymphoma: Report of the 2013 Mantle Cell Lymphoma Consortium Workshop [published online ahead of print April 17, 2014]. Leuk Lymphoma.
3. Dreyling M, Kluin-Nelemans HC, Beà S, et al; European MCL Network. Update on the molecular pathogenesis and clinical treatment of mantle cell lymphoma: Report of the 11th annual conference of the European Mantle Cell Lymphoma Network. Leuk Lymphoma. 2013;54(4):699-707.
4. Dreyling M, Thieblemont C, Gallamini A, et al. ESMO Consensus conferences: Guidelines on malignant lymphoma. Part 2: Marginal zone lymphoma, mantle cell lymphoma, peripheral T-cell lymphoma. Ann Oncol. 2013;24(4):857-877.
5. Williams ME. Transplantation for mantle cell lymphoma: Is it the right thing to do? Hematology Am Soc Hematol Educ Program. 2013;2013(1):568-574.
6. Dreyling M, Hiddemann W; European MCL Network. Current treatment standards and emerging strategies in mantle cell lymphoma. Hematology Am Soc Hematol Educ Program. 2009;2009(1):542-551.
7. Budde LE, Guthrie KA, Till BG, et al. Mantle cell lymphoma international prognostic index but not pretransplantation induction regimen predicts survival for patients with mantle-cell lymphoma receiving high-dose therapy and autologous stem-cell transplantation. J Clin Oncol. 2011;29(22):3023-3029.
8. Hoster E, Dreyling M, Klapper W, et al; German Low Grade Lymphoma Study Group (GLSG), European MCL Network. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood. 2008;111(2):558-565.
9. Geisler CH, Kolstad A, Laurell A, et al; Nordic Lymphoma Group. Long-term progression-free survival of mantle cell lymphoma after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: A nonrandomized phase 2 multicenter study by the Nordic Lymphoma Group. Blood. 2008;112(7):2687-2693.
10. Delarue R, Haioun C, Ribrag V, et al; Groupe d’Etude des Lymphomes de l’Adulte (GELA). CHOP and DHAP plus rituximab followed by autologous stem cell transplantation in mantle cell lymphoma: A phase 2 study from the Groupe d’Etude des Lymphomes de l’Adulte. Blood. 2013;121(1):48-53.
11. Damon LE, Johnson JL, Niedzwiecki D, et al. Immunochemotherapy and autologous stem-cell transplantation for untreated patients with mantle-cell lymphoma: CALGB 59909. J Clin Oncol. 2009;27(36):6101-6108.
12. Dreyling M, Lenz G, Hoster E, et al. Early consolidation by myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission significantly prolongs progression-free survival in mantle-cell lymphoma:Results of a prospective randomized trial of the European MCL Network. Blood. 2005;105(7):2677-2684.
13. Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: Results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol. 2005;23(9):1984-1992.
14. Kluin-Nelemans HC, Hoster E, Hermine O, et al. Treatment of older patients with mantle-cell lymphoma. N Engl J Med. 2012;367(6):520-531.
15. Andersen NS, Pedersen LB, Laurell A, et al. Pre-emptive treatment with rituximab of molecular relapse after autologous stem cell transplantation in mantle cell lymphoma. J Clin Oncol. 2009;27(26):4365-4370.
16. Cassaday RD, Guthrie KA, Budde EL, et al. Specific features identify patients with relapsed or refractory mantle cell lymphoma benefitting from autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2013;19(9):1403-1406.
17. Maris MB, Sandmaier BM, Storer BE, et al. Allogeneic hematopoietic cell transplantation after fludarabine and 2 Gy total body irradiation for relapsed and refractory mantle cell lymphoma. Blood. 2004;104(12):3535-3542.