User login
The Emerging Role of Liquid Biopsy in the Diagnosis and Management of CRC
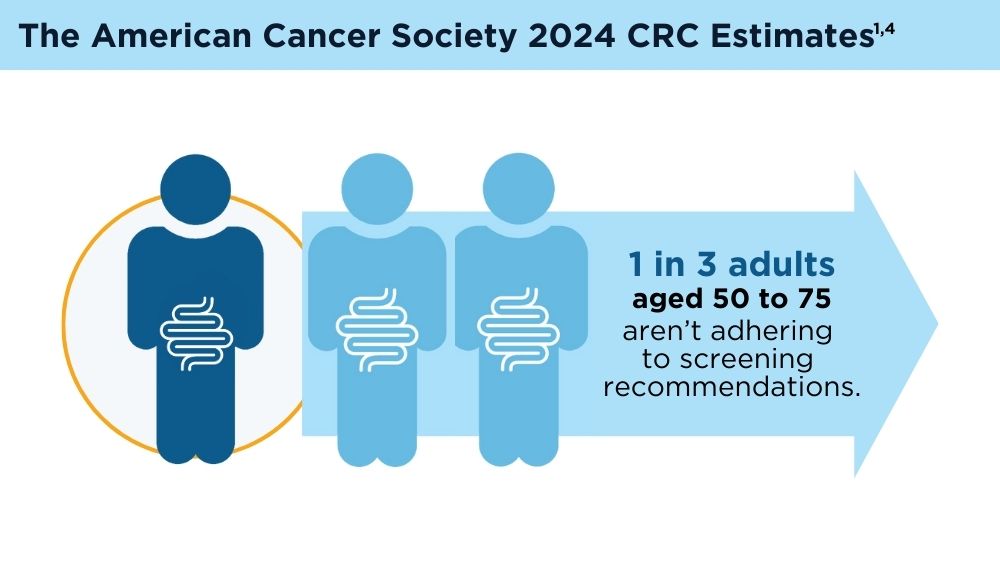
Key statistics for colorectal cancer. American Cancer Society. Revised January 13, 2023. Accessed November 30, 2023. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
Mazouji O, Ouhajjou A, Incitti R, Mansour H. Updates on clinical use of liquid biopsy in colorectal cancer screening, diagnosis, follow-up, and treatment guidance. Front Cell Dev Biol. 2021;9:660924. doi:10.3389/fcell.2021.660924
Vacante M, Ciuni R, Basile F, Biondi A. The liquid biopsy in the management of colorectal cancer: an overview. Biomedicines. 2020;8(9):308. doi:10.3390/biomedicines8090308
American Cancer Society. Colorectal cancer facts & figures 2020-2022. Published 2022. Accessed November 30, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/colorectal-cancer-facts-and-figures/colorectal-cancer-facts-and-figures-2020-2022.pdf
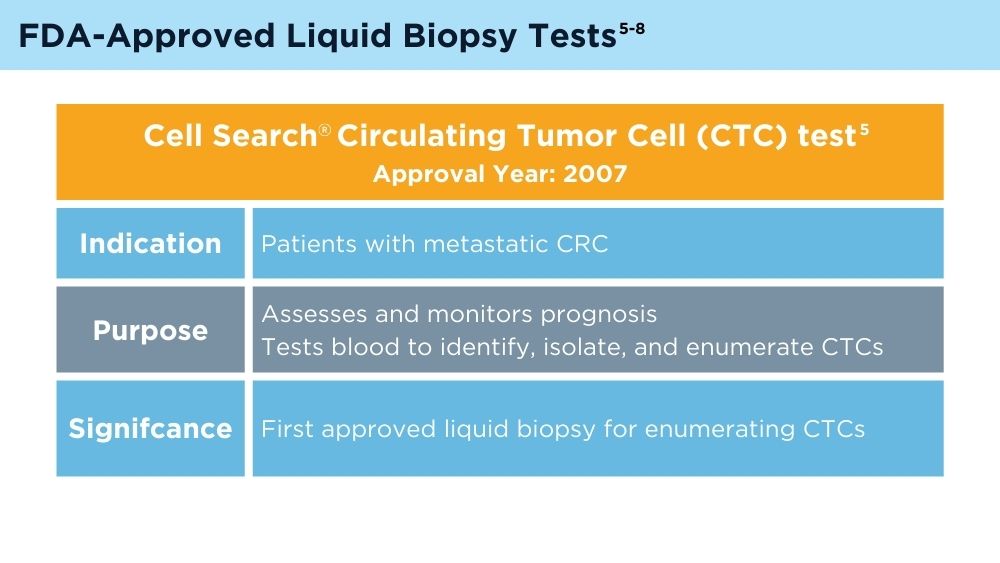
Johnson & Johnson. FDA clears Cellsearch™ circulating tumor cell test [news release]. Published February 27, 2008. Accessed November 30, 2023. https://johnsonandjohnson.gcs-web.com/news-releases/news-release-details/fda-clears-cellsearchtm-circulating-tumor-cell-test
US Food and Drug Administration. Summary of safety and effectiveness data, Epi proColon®. PMA number P130001. Published April 12, 2016. Accessed November 30, 2023. https://www.accessdata.fda.gov/cdrh_docs/pdf13/p130001b.pdf
FDA approves blood tests that can help guide cancer treatment. National Institutes of Health, National Cancer Institute. Published October 15, 2020. Accessed November 30, 2023. https://www.cancer.gov/news-events/cancer-currents-blog/2020/fda-guardant-360-foundation-one-cancer-liquid-biopsy
Foundation Medicine. US Food and Drug Administration (FDA) approves FoundationOne®LiquidCDx as a companion diagnostic for Pfizer’s BRAFTOVI® (encorafenib) in combination with cetuximab to identify patients with BRAF V600E alterations in metastatic colorectal cancer [press release]. Published June 10, 2023. Accessed November 30, 2023. https://www.foundationmedicine.com/press-releases/f9b285eb-db6d-4f61-856c-3f1edb803937
Key statistics for colorectal cancer. American Cancer Society. Revised January 13, 2023. Accessed November 30, 2023. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
Mazouji O, Ouhajjou A, Incitti R, Mansour H. Updates on clinical use of liquid biopsy in colorectal cancer screening, diagnosis, follow-up, and treatment guidance. Front Cell Dev Biol. 2021;9:660924. doi:10.3389/fcell.2021.660924
Vacante M, Ciuni R, Basile F, Biondi A. The liquid biopsy in the management of colorectal cancer: an overview. Biomedicines. 2020;8(9):308. doi:10.3390/biomedicines8090308
American Cancer Society. Colorectal cancer facts & figures 2020-2022. Published 2022. Accessed November 30, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/colorectal-cancer-facts-and-figures/colorectal-cancer-facts-and-figures-2020-2022.pdf
Johnson & Johnson. FDA clears Cellsearch™ circulating tumor cell test [news release]. Published February 27, 2008. Accessed November 30, 2023. https://johnsonandjohnson.gcs-web.com/news-releases/news-release-details/fda-clears-cellsearchtm-circulating-tumor-cell-test
US Food and Drug Administration. Summary of safety and effectiveness data, Epi proColon®. PMA number P130001. Published April 12, 2016. Accessed November 30, 2023. https://www.accessdata.fda.gov/cdrh_docs/pdf13/p130001b.pdf
FDA approves blood tests that can help guide cancer treatment. National Institutes of Health, National Cancer Institute. Published October 15, 2020. Accessed November 30, 2023. https://www.cancer.gov/news-events/cancer-currents-blog/2020/fda-guardant-360-foundation-one-cancer-liquid-biopsy
Foundation Medicine. US Food and Drug Administration (FDA) approves FoundationOne®LiquidCDx as a companion diagnostic for Pfizer’s BRAFTOVI® (encorafenib) in combination with cetuximab to identify patients with BRAF V600E alterations in metastatic colorectal cancer [press release]. Published June 10, 2023. Accessed November 30, 2023. https://www.foundationmedicine.com/press-releases/f9b285eb-db6d-4f61-856c-3f1edb803937
Key statistics for colorectal cancer. American Cancer Society. Revised January 13, 2023. Accessed November 30, 2023. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
Mazouji O, Ouhajjou A, Incitti R, Mansour H. Updates on clinical use of liquid biopsy in colorectal cancer screening, diagnosis, follow-up, and treatment guidance. Front Cell Dev Biol. 2021;9:660924. doi:10.3389/fcell.2021.660924
Vacante M, Ciuni R, Basile F, Biondi A. The liquid biopsy in the management of colorectal cancer: an overview. Biomedicines. 2020;8(9):308. doi:10.3390/biomedicines8090308
American Cancer Society. Colorectal cancer facts & figures 2020-2022. Published 2022. Accessed November 30, 2023. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/colorectal-cancer-facts-and-figures/colorectal-cancer-facts-and-figures-2020-2022.pdf
Johnson & Johnson. FDA clears Cellsearch™ circulating tumor cell test [news release]. Published February 27, 2008. Accessed November 30, 2023. https://johnsonandjohnson.gcs-web.com/news-releases/news-release-details/fda-clears-cellsearchtm-circulating-tumor-cell-test
US Food and Drug Administration. Summary of safety and effectiveness data, Epi proColon®. PMA number P130001. Published April 12, 2016. Accessed November 30, 2023. https://www.accessdata.fda.gov/cdrh_docs/pdf13/p130001b.pdf
FDA approves blood tests that can help guide cancer treatment. National Institutes of Health, National Cancer Institute. Published October 15, 2020. Accessed November 30, 2023. https://www.cancer.gov/news-events/cancer-currents-blog/2020/fda-guardant-360-foundation-one-cancer-liquid-biopsy
Foundation Medicine. US Food and Drug Administration (FDA) approves FoundationOne®LiquidCDx as a companion diagnostic for Pfizer’s BRAFTOVI® (encorafenib) in combination with cetuximab to identify patients with BRAF V600E alterations in metastatic colorectal cancer [press release]. Published June 10, 2023. Accessed November 30, 2023. https://www.foundationmedicine.com/press-releases/f9b285eb-db6d-4f61-856c-3f1edb803937
Colorectal cancer: New observations, new implications
The incidence and mortality of colorectal cancer (CRC) have declined by 3% per year over the past 10-15 years – a remarkable achievement. The decline in incidence has been dramatic for individuals over age 50 years, who are targeted by screening. However, the reduction in CRC risk does not apply to all populations in the United States. New epidemiologic trends and observations point to patient demographics and regional variation as potential risk factors. While such observations provide what I call “blurry snapshots,” they may well have important implications for our approach to screening and prevention.
What are the reasons? There are several personal and environmental factors that could be contributing. Obesity and metabolic syndrome are risk factors for CRC and have been more commonly developing in childhood over the past 40 years. Alteration of the microbiome could also potentially predispose one to developing CRC. The use of antibiotics in childhood was more common for some of these younger generations than it was for the preceding generations, and antibiotics have been introduced into the food industry to fatten animals. The introduction of food chemicals could also either alter the microbiome and/or promote inflammation, which could lead to neoplasia. Exposure to more ambient radiation may be another risk factor.
These hypotheses are biologically plausible – but untested. Nevertheless, this observational trend does have implications for clinicians. First, studies have shown that up to 20% of CRCs before age 50 years are associated with germline mutations, while others are associated with a family history of CRC. Therefore, it is important to capture and update family history. In addition, there is evidence that individuals aged 40-49 years with rectal bleeding have a higher risk of advanced adenomas, so our threshold for performing diagnostic colonoscopy should be lowered. African Americans also have a higher risk of CRC at a younger age than do other racial groups and might benefit from early screening at age 45 years. Notably, recent recommendations from the American Cancer Society call for consideration of screening everyone at age 45 years.
There is substantial state-to-state and county-to-county variation in the incidence and mortality of CRC. While some of this variation can be explained by racial variation, smoking, obesity, and social determinants of health, there are “hot-spots” that may defy easy explanation. There has been very little research about environmental factors (air, water, and ambient radiation). Two regions at particularly high risk are the Mississippi Delta region and Appalachia – areas where water pollution could be a factor. The substantial county-to-county variation within these high-risk areas points to a potential environmental culprit, but further research is needed.
For the GI community, there are several implications to be found in these changing demographics and risks. For one, we may need to consider expanding our risk concepts to include not only genetic and personal risk factors but also environmental factors. To mitigate risk, providers and public health officials may need to then target these high-risk areas for more intensive screening efforts.
Dr. Lieberman is a professor of medicine and chief of gastroenterology and hepatology at Oregon Health and Science University in Portland. He has no conflicts of interest. Dr. Lieberman made his comments during the AGA Institute Presidential Plenary at the Annual Digestive Disease Week.
The incidence and mortality of colorectal cancer (CRC) have declined by 3% per year over the past 10-15 years – a remarkable achievement. The decline in incidence has been dramatic for individuals over age 50 years, who are targeted by screening. However, the reduction in CRC risk does not apply to all populations in the United States. New epidemiologic trends and observations point to patient demographics and regional variation as potential risk factors. While such observations provide what I call “blurry snapshots,” they may well have important implications for our approach to screening and prevention.
What are the reasons? There are several personal and environmental factors that could be contributing. Obesity and metabolic syndrome are risk factors for CRC and have been more commonly developing in childhood over the past 40 years. Alteration of the microbiome could also potentially predispose one to developing CRC. The use of antibiotics in childhood was more common for some of these younger generations than it was for the preceding generations, and antibiotics have been introduced into the food industry to fatten animals. The introduction of food chemicals could also either alter the microbiome and/or promote inflammation, which could lead to neoplasia. Exposure to more ambient radiation may be another risk factor.
These hypotheses are biologically plausible – but untested. Nevertheless, this observational trend does have implications for clinicians. First, studies have shown that up to 20% of CRCs before age 50 years are associated with germline mutations, while others are associated with a family history of CRC. Therefore, it is important to capture and update family history. In addition, there is evidence that individuals aged 40-49 years with rectal bleeding have a higher risk of advanced adenomas, so our threshold for performing diagnostic colonoscopy should be lowered. African Americans also have a higher risk of CRC at a younger age than do other racial groups and might benefit from early screening at age 45 years. Notably, recent recommendations from the American Cancer Society call for consideration of screening everyone at age 45 years.
There is substantial state-to-state and county-to-county variation in the incidence and mortality of CRC. While some of this variation can be explained by racial variation, smoking, obesity, and social determinants of health, there are “hot-spots” that may defy easy explanation. There has been very little research about environmental factors (air, water, and ambient radiation). Two regions at particularly high risk are the Mississippi Delta region and Appalachia – areas where water pollution could be a factor. The substantial county-to-county variation within these high-risk areas points to a potential environmental culprit, but further research is needed.
For the GI community, there are several implications to be found in these changing demographics and risks. For one, we may need to consider expanding our risk concepts to include not only genetic and personal risk factors but also environmental factors. To mitigate risk, providers and public health officials may need to then target these high-risk areas for more intensive screening efforts.
Dr. Lieberman is a professor of medicine and chief of gastroenterology and hepatology at Oregon Health and Science University in Portland. He has no conflicts of interest. Dr. Lieberman made his comments during the AGA Institute Presidential Plenary at the Annual Digestive Disease Week.
The incidence and mortality of colorectal cancer (CRC) have declined by 3% per year over the past 10-15 years – a remarkable achievement. The decline in incidence has been dramatic for individuals over age 50 years, who are targeted by screening. However, the reduction in CRC risk does not apply to all populations in the United States. New epidemiologic trends and observations point to patient demographics and regional variation as potential risk factors. While such observations provide what I call “blurry snapshots,” they may well have important implications for our approach to screening and prevention.
What are the reasons? There are several personal and environmental factors that could be contributing. Obesity and metabolic syndrome are risk factors for CRC and have been more commonly developing in childhood over the past 40 years. Alteration of the microbiome could also potentially predispose one to developing CRC. The use of antibiotics in childhood was more common for some of these younger generations than it was for the preceding generations, and antibiotics have been introduced into the food industry to fatten animals. The introduction of food chemicals could also either alter the microbiome and/or promote inflammation, which could lead to neoplasia. Exposure to more ambient radiation may be another risk factor.
These hypotheses are biologically plausible – but untested. Nevertheless, this observational trend does have implications for clinicians. First, studies have shown that up to 20% of CRCs before age 50 years are associated with germline mutations, while others are associated with a family history of CRC. Therefore, it is important to capture and update family history. In addition, there is evidence that individuals aged 40-49 years with rectal bleeding have a higher risk of advanced adenomas, so our threshold for performing diagnostic colonoscopy should be lowered. African Americans also have a higher risk of CRC at a younger age than do other racial groups and might benefit from early screening at age 45 years. Notably, recent recommendations from the American Cancer Society call for consideration of screening everyone at age 45 years.
There is substantial state-to-state and county-to-county variation in the incidence and mortality of CRC. While some of this variation can be explained by racial variation, smoking, obesity, and social determinants of health, there are “hot-spots” that may defy easy explanation. There has been very little research about environmental factors (air, water, and ambient radiation). Two regions at particularly high risk are the Mississippi Delta region and Appalachia – areas where water pollution could be a factor. The substantial county-to-county variation within these high-risk areas points to a potential environmental culprit, but further research is needed.
For the GI community, there are several implications to be found in these changing demographics and risks. For one, we may need to consider expanding our risk concepts to include not only genetic and personal risk factors but also environmental factors. To mitigate risk, providers and public health officials may need to then target these high-risk areas for more intensive screening efforts.
Dr. Lieberman is a professor of medicine and chief of gastroenterology and hepatology at Oregon Health and Science University in Portland. He has no conflicts of interest. Dr. Lieberman made his comments during the AGA Institute Presidential Plenary at the Annual Digestive Disease Week.