User login
Gastroesophageal reflux disease (GERD) is a chronic and common medical problem, with up to 40% of the population experiencing its symptoms at least once per month.1 The condition develops when the reflux of stomach contents causes troublesome symptoms or complications.2
GERD symptoms can range from heartburn and regurgitation to cough and hoarseness. While many patients’ symptoms respond to medical treatment, the diagnosis and treatment in those whose symptoms do not respond to a proton pump inhibitor (PPI) may be challenging.
This article reviews the diagnosis and treatment options for GERD.
SYMPTOMS: TYPICAL, ATYPICAL, AND ALARM
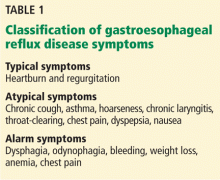
Symptoms of GERD (Table 1) can be classified as typical (heartburn and regurgitation) or atypical (cough, asthma, hoarseness, chronic laryngitis, throat-clearing, chest pain, dyspepsia, and nausea). Atypical symptoms are more likely to be due to GERD if patients also have typical symptoms and if the symptoms respond to a trial of a PPI.3
Alarm symptoms. Keep in mind that extraesophageal presentations may be multifactorial, and it may be difficult to establish that reflux, even if present, is actually the cause. While chest pain may be due to GERD, it is important to rule out cardiac chest pain before considering GERD as a cause. Similarly, dysphagia along with typical or atypical symptoms warrants investigation for potential complications such as underlying motility disorder, esophageal stricture, esophageal ring, or malignancy.4 Other alarm symptoms include odynophagia, bleeding, weight loss, and anemia.
DIAGNOSING GERD: RESPONSE TO A PPI IS DIAGNOSTIC
Patients with typical symptoms that respond to PPI therapy need no further evaluation for a diagnosis of GERD to be made.5 On the other hand, further testing should be undertaken in patients with typical symptoms that do not respond to PPI therapy, in patients presenting with atypical symptoms, and in patients in whom antireflux surgery is being considered. Figure 1 shows our proposed algorithm.
Try a PPI for 6–8 weeks
Relief of heartburn and regurgitation after a 6- to 8-week course of a PPI strongly suggests GERD.6 However, a negative trial of a PPI does not rule out GERD, as this approach has been found to have a sensitivity of 78% and specificity of 54%.6
Despite this limitation, a trial of PPI therapy should be offered to patients presenting with typical symptoms and no alarm features. This approach has been found to be more cost-effective than proceeding directly to diagnostic testing.7
Endoscopy
Endoscopic findings in GERD can include erosive esophagitis, peptic stricture, and Barrett esophagus. Esophageal erosions are a highly specific sign of GERD; the Los Angeles classification system, a standardized scale for grading the severity of erosive esophagitis (from A to D, with D the most severe) provides an objective way to assess severity.8 However, most patients with heartburn and regurgitation do not have erosive disease, thus limiting the sensitivity of upper endoscopy as an initial diagnostic test in patients with suspected GERD.9
We recommend endoscopy for patients who present with alarm symptoms, patients with noncardiac chest pain, PPI nonresponders, and patients with chronic GERD symptoms and multiple risk factors for Barrett esophagus besides GERD, such as older age, male sex, white race, overweight, and smoking.10
Ambulatory pH and impedance monitoring
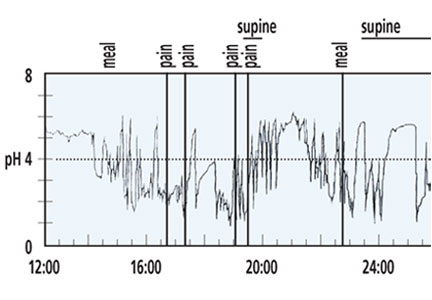
Ambulatory pH monitoring is the gold standard test for pathologic acid exposure in the esophagus. pH testing is indicated in PPI nonresponders, patients presenting with atypical symptoms, and before antireflux surgery.
In general, pH testing should be performed after the patient has been off PPI therapy for at least 7 days, as the test is highly unlikely to be abnormal while a patient is on a PPI.11 It is done either with a transnasal catheter for 24 hours, or with a wireless capsule (Bravo pH System, Given Imaging Ltd, Duluth, GA), which collects 48 to 96 hours of data. Studies of the wireless system have shown that its sensitivity increases 12% to 25% when it is performed for 48 hours compared with 24 hours.12,13
The pH test can be combined with impedance testing to evaluate for nonacid reflux.14 However, the clinical significance of nonacid reflux remains controversial, and for this reason the Esophageal Diagnostic Advisory Panel recommends that the decision to perform antireflux surgery should not be based on abnormal impedance testing.15
During pH and impedance testing, special software can calculate how closely the patient’s symptoms correlate with esophageal acid exposure. The symptom index (SI) and symptom association probability (SAP) are the symptom measurements most commonly used in practice. The SI measures the overall strength of the relationship, and an SI greater than 50% is considered positive.16 The SAP determines whether this relationship could have occurred by chance, and an SAP greater than 95% is statistically significant.17 In patients with normal levels of esophageal acid exposure, an elevated SI or SAP may indicate a component of esophageal hypersensitivity in symptom generation.
At our institution, we generally perform pH-only transnasal or wireless testing off PPI therapy to establish that the patient has pathologic acid exposure in the distal esophagus. Combined pH-impedance testing is typically reserved for patients with atypical symptoms unresponsive to PPI therapy and abnormal results on previous pH testing, which allows for correlation of nonacid reflux and symptoms.
Other tests
Esophageal manometry and barium esophagography have limited value in the primary diagnosis of GERD. However, they should be considered to rule out achalasia and other esophageal motility disorders in patients whose symptoms do not respond to PPIs. For this reason, esophageal manometry should be performed before considering antireflux surgery.
MANAGING GERD
Table 2 summarizes the various treatments for GERD.
Lifestyle modifications
Lifestyle modifications are the first-line therapy for GERD. Modifications that have been studied include weight loss, head-of-bed elevation, and avoidance of tobacco, alcohol, and late-night meals. Another modification that has been suggested is avoiding foods that can aggravate reflux symptoms—eg, caffeine, coffee, chocolate, spicy foods, highly acidic foods (oranges, tomatoes), and fatty foods. Of these, only weight loss and head-of-bed elevation have been proven effective.18
Three randomized controlled trials demonstrated that GERD symptoms and esophageal pH values improved with head-of-bed elevation using blocks or incline foam wedges.19–21 Several cohort studies demonstrated reduction in GERD symptoms with weight loss.22,23 Recently, a prospective cohort study also found that smoking cessation significantly improved GERD symptoms in patients with normal body mass index and severe symptoms.24
Antacids
Several antacids (eg, sodium bicarbonate, calcium carbonate, magnesium hydroxide, aluminum hydroxide) are available over the counter.
Antacids were thought to control heartburn symptoms by increasing the pH of gastric contents that might subsequently reflux into the esophagus. However, well-controlled studies have shown that they relieve heartburn by neutralizing acid in the esophagus, with no significant effect on gastric pH.25,26
Antacids provide rapid but short-lived relief from an existing episode of heartburn. Because they do not significantly raise the gastric pH, they do not prevent subsequent reflux episodes from repeatedly exposing the esophagus to gastric acid and causing heartburn. Additionally, antacids have not been shown to contribute to healing of erosive esophagitis.27 Hence, they may not be optimal for treating frequent reflux heartburn.
Sodium alginate
Gastric acid pockets are unbuffered pools of acid that float on top of ingested food.28 They develop as a result of poor mixing of newly secreted acid and food in the proximal stomach, which remains relatively quiescent after a meal compared with the distal stomach.29 In GERD, proximal extension of the acid pocket above the diaphragm increases the risk of acid reflux.30 The acid pocket is therefore an important source of postprandial acid in GERD and, as such, represents a unique therapeutic target.
Emerging evidence suggests that alginates may act directly on the acid pocket. Alginates are natural polysaccharide polymers that, on contact with gastric acid, precipitate within minutes into a low-density viscous gel of near-neutral pH. The change in pH triggers the sodium bicarbonate in the formulation to release carbon dioxide that becomes trapped in the alginate gel, causing it to float to the top of the gastric contents like a raft.31
A randomized controlled trial demonstrated that sodium alginate was as effective as omeprazole in relieving symptoms in patients with nonerosive reflux disease.32 Alginate has also been shown to provide more postprandial reflux relief than antacids.33
Histamine-2 receptor antagonists
Histamine-2 receptor antagonists act more swiftly and increase postprandial gastric pH more rapidly than PPIs, thus making them a good option for prophylaxis against postprandial GERD.34 Taking these drugs at bedtime may help in patients with objective nighttime reflux despite optimal PPI use. However, tachyphylaxis may occur as early as 1 week after starting combination therapy.35
Proton pump inhibitors
There are currently seven available PPIs, including four that can be obtained over the counter (omeprazole, lansoprazole, esomeprazole, and omeprazole-sodium bicarbonate) and three available only by prescription (rabeprazole, pantoprazole, and dexlansoprazole). Studies have shown than a standard 6- to 8-week course of a PPI provides complete symptom relief in 70% to 80% of patients with erosive reflux disease and in 60% of patients with nonerosive reflux disease.36,37 Clinically, PPIs all appear to be similar in their symptom relief.38
Most PPIs should be taken 30 to 60 minutes before meals. Exceptions are omeprazole-sodium bicarbonate and dexlansoprazole, which can be taken without regard to meals. At our institution, we usually start PPIs in a once-daily standard dose for 6 to 8 weeks and consider increasing to twice-daily dosing if symptoms do not respond completely. Patients with mild intermittent GERD symptoms may benefit from “on-demand” use of PPIs. This approach is best suited for patients with nonerosive reflux disease without evidence of Barrett esophagus on endoscopy.
Safety and adverse effects of PPIs
PPIs are generally safe but can cause adverse effects (Table 3).
Osteoporosis. In 2010, the US Food and Drug Administration issued warnings regarding the potential for wrist, hip, and spine fractures in PPI users.26 Most recent evidence suggests that PPIs may be associated with a small increase in risk of hip fractures in patients already at high risk.39,40 However, the 2013 American College of Gastroenterology (ACG) guidelines say that patients with known osteoporosis can remain on PPI therapy, and concern for hip fractures and osteoporosis should not affect the decision to use PPIs long-term except in patients with other risk factors for hip fracture.41
Community-acquired pneumonia. An increased risk of community-acquired pneumonia cannot be clearly documented in association with PPI therapy. Multiple studies, including randomized controlled trials, investigated this potential correlation. However, evidence suggests that short-term but not long-term PPI use may be associated with an overall increased risk of community-acquired pneumonia.42,43 Current guidelines suggest that in patients who need a PPI, the drug should not be withheld on the basis of a potential risk of community-acquired pneumonia.41
Clostridium difficile infection. In theory, PPIs may increase the risk of C difficile infection by increasing the ability of the spore to convert to the vegetative form and to survive intraluminally. In fact, studies and meta-analyses have suggested that PPIs do increase the risk of development and recurrence of C difficile infection.44,45 Therefore, PPIs should be used with care in patients who are at risk.41
Interaction with clopidogrel. The antiplatelet activity of clopidogrel requires activation by CYP2C19, the same pathway required for metabolism of some PPIs. Concern was raised about decreased antiplatelet activity of clopidogrel in the presence of PPIs. This was extensively studied, and there now appears to be no increased risk of adverse cardiovascular events in patients on PPIs, based on data from well-controlled randomized trials.46,47 A consensus panel of the American College of Cardiology Foundation, the American Heart Association, and the ACG said that PPIs may be used for appropriate indications in patients taking clopidogrel.47
Hypomagnesemia. By an unknown molecular mechanism, PPIs are thought to reduce intestinal magnesium absorption, leading to hypomagnesemia. A meta-analysis published in 2011 showed that PPI-induced hypomagnesemia is a drug-class effect and occurred after a median of 5.5 years of PPI use. Stopping the PPI resulted in magnesium recovery in 4 days, and rechallenge led to recurrence within 4 days.48
Hence, to avoid putting patients on long-term PPI therapy at risk, clinicians should anticipate this problem. Our practice is to check the magnesium level before starting a patient on long-term PPI therapy, and then to repeat the measurement every 1 to 2 years.
Baclofen
Transient lower esophageal sphincter relaxation has been shown to be a cause of reflux in healthy people and in patients with GERD.49
Baclofen, a muscle relaxant with selective gamma-aminobutyric acid receptor class B agonist properties, reduces transient lower esophageal sphincter relaxation in humans.50 In a well-designed, double-blind, randomized controlled trial, baclofen was associated with a significant decrease in upright reflux on 24-hour pH monitoring and significant improvement in belching and overall reflux symptoms.51 However, baclofen is not approved by the US Food and Drug Administration for the treatment of GERD, and its use may be limited by side effects such as somnolence and dizziness.
Antireflux surgery
Antireflux surgery is a reasonable option for selected patients with chronic GERD. The main types of surgery are laparoscopic fundoplication and, for obese patients, gastric bypass. Reasons to consider antireflux surgery include desire to stop PPI therapy, esophagitis not healed by PPIs, symptomatic hiatal hernia, and refractory reflux documented by pH testing.41
In general, surgical therapy may be considered in patients who respond to PPIs, but patients who do not respond to PPIs are less likely to respond to antireflux surgery.15 Other patients less likely to respond are those with symptoms of dyspepsia, such as nausea, vomiting, and epigastric pain.41
Common adverse effects of antireflux surgery include gas-bloat syndrome (up to 85% of patients), dysphagia (10% to 50% of patients), diarrhea (18% to 33% of patients), and recurrent heartburn (10% to 62% of patients).52
Endoscopic and minimally invasive antireflux procedures include endoscopic plication of the lower esophageal sphincter, radiofrequency augmentation of the lower esophageal sphincter, and sphincter augmentation by a string of titanium beads. While some have shown promise, they are not recommended by the most recent ACG guidelines, given lack of long-term data.41
REFRACTORY GERD
There is no consensus on the definition of refractory GERD. However, for the sake of simplicity, we can define it as persistence of suspected GERD symptoms despite treatment with a PPI. This may vary from a partial response to PPI therapy to a complete absence of response.
It is extremely important to rule out non-GERD causes of the ongoing symptoms, such as achalasia, gastroparesis, eosinophilic esophagitis, rumination, and aerophagia. PPI nonresponders are more likely to be obese, poorly compliant, and have extraesophageal symptoms.53–56 As previously discussed, PPIs should be taken 30 to 60 minutes before meals. For patients whose symptoms fail to respond to standard-dose daily PPI therapy, switching to another PPI or doubling the dose is common, although data to support this practice are limited. Of note, omeprazole-sodium bicarbonate has been shown to provide more symptom relief in nocturnal GERD.57 Additionally, adding a nighttime histamine-2 receptor antagonist may also help in patients with objective nighttime reflux.41
After noncompliance and suboptimal PPI dosing have been ruled out, PPI nonresponders with typical symptoms should undergo upper endoscopy and subsequent pH monitoring. Normal esophageal acid exposure on pH testing suggests functional heartburn or functional dyspepsia. Negative pH testing in a patient with atypical symptoms suggests a non-GERD cause of symptoms, and referral to an otolaryngologist, pulmonologist, or allergist is often warranted.
While antireflux surgery can be considered for patients with nonacid reflux on impedance testing, it should again be noted that GERD in patients with no response to PPIs is less likely to respond to antireflux surgery.15
TAKE-HOME POINTS
- GERD is a common medical condition, affecting up to 40% of US adults at least once monthly.
- GERD can result in a wide variety of symptoms, including typical heartburn and regurgitation as well as atypical symptoms such as cough.
- On the other hand, keep in mind that multiple non-GERD causes of heartburn and regurgitation may exist.
- Testing for GERD includes endoscopy and pH testing as well as functional testing such as esophageal manometry.
- While in most patients GERD will respond to lifestyle changes and antisecretory therapy such as a PPI, careful attention must be given to patients with symptoms refractory to PPI therapy.
- For a subset of patients, antireflux surgery may be a reasonable option, but care must be taken to exclude patients with a lower likelihood of responding to surgery.
- Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology 1997; 112:1448–1456.
- Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R; Globale Konsensusgrupp. [The Montreal definition and classification of gastroesophageal reflux disease: a global, evidence-based consensus paper]. Z Gastroenterol 2007; 45:1125–1240. In German.
- Gerson LB, Kahrilas PJ, Fass R. Insights into gastroesophageal reflux disease-associated dyspeptic symptoms. Clin Gastroenterol Hepatol 2011; 9:824–833.
- Vakil NB, Traxler B, Levine D. Dysphagia in patients with erosive esophagitis: prevalence, severity, and response to proton pump inhibitor treatment. Clin Gastroenterol Hepatol 2004; 2:665–668.
- Kahrilas PJ, Shaheen NJ, aezi MF, et al; American Gastroenterological Association. American Gastroenterological Association Medical Position Statement on the management of gastroesophageal reflux disease. Gastroenterology 2008; 135:1383–1391 e1–5.
- Numans ME, Lau J, de Wit NJ, Bonis PA. Short-term treatment with proton-pump inhibitors as a test for gastroesophageal reflux disease: a meta-analysis of diagnostic test characteristics. Ann Intern Med 2004; 140:518–527.
- Fass R. Empirical trials in treatment of gastroesophageal reflux disease. Dig Dis 2000; 18:20–26.
- Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 1999; 45:172–180.
- Johansson KE, Ask P, Boeryd B, Fransson SG, Tibbling L. Oesophagitis, signs of reflux, and gastric acid secretion in patients with symptoms of gastro-oesophageal reflux disease. Scand J Gastroenterol 1986; 21:837–847.
- Becher A, Dent J. Systematic review: ageing and gastro-oesophageal reflux disease symptoms, oesophageal function and reflux oesophagitis. Aliment Pharmacol Ther 2011; 33:442–454.
- Charbel S, Khandwala F, Vaezi MF. The role of esophageal pH monitoring in symptomatic patients on PPI therapy. Am J Gastroenterol 2005; 100:283–289.
- Pandolfino JE, Richter JE, Ours T, Guardino JM, Chapman J, Kahrilas PJ. Ambulatory esophageal pH monitoring using a wireless system. Am J Gastroenterol 2003; 98:740–749.
- Prakash C, Clouse RE. Value of extended recording time with wireless pH monitoring in evaluating gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2005; 3:329–334.
- Hirano I, Richter JE; Practice Parameters Committee of the American College of Gastroenterology. ACG practice guidelines: esophageal reflux testing. Am J Gastroenterol 2007; 102:668–685.
- Jobe BA, Richter JE, Hoppo T, et al. Preoperative diagnostic workup before antireflux surgery: an evidence and experience-based consensus of the Esophageal Diagnostic Advisory Panel. J Am Coll Surg 2013; 217:586–597.
- Singh S, Richter JE, Bradley LA, Haile JM. The symptom index. Differential usefulness in suspected acid-related complaints of heartburn and chest pain. Dig Dis Sci 1993; 38:1402–1408.
- Weusten BL, Roelofs JM, Akkermans LM, Van Berge-Henegouwen GP, Smout AJ. The symptom-association probability: an improved method for symptom analysis of 24-hour esophageal pH data. Gastroenterology 1994; 107:1741–1745.
- Kaltenbach T, Crockett S, Gerson LB. Are lifestyle measures effective in patients with gastroesophageal reflux disease? An evidence-based approach. Arch Intern Med 2006; 166:965–971.
- Hamilton JW, Boisen RJ, Yamamoto DT, Wagner JL, Reichelderfer M. Sleeping on a wedge diminishes exposure of the esophagus to refluxed acid. Dig Dis Sci 1988; 33:518–522.
- Pollmann H, Zillessen E, Pohl J, et al. [Effect of elevated head position in bed in therapy of gastroesophageal reflux]. Z Gastroenterol 1996; 34(suppl 2):93–99. In German.
- Stanciu C, Bennett JR. Effects of posture on gastro-oesophageal reflux. Digestion 1977; 15:104–109.
- Fraser-Moodie CA, Norton B, Gornall C, Magnago S, Weale AR, Holmes GK. Weight loss has an independent beneficial effect on symptoms of gastro-oesophageal reflux in patients who are overweight. Scand J Gastroenterol 1999; 34:337–340.
- Mathus-Vliegen LM, Tytgat GN. Twenty-four-hour pH measurements in morbid obesity: effects of massive overweight, weight loss and gastric distension. Eur J Gastroenterol Hepatol 1996; 8:635–640.
- Ness-Jensen E, Lindam A, Lagergren J, Hveem K. Tobacco smoking cessation and improved gastroesophageal reflux: a prospective population-based cohort study: the HUNT study. Am J Gastroenterol 2014; 109:171–177.
- Collings KL, Rodriguez-Stanley S, Proskin HM, Robinson M, Miner PB Jr. Clinical effectiveness of a new antacid chewing gum on heartburn and oesophageal pH control. Aliment Pharmacol Ther 2002; 16:2029–2035.
- Decktor DL, Robinson M, Maton PN, Lanza FL, Gottlieb S. Effects of aluminum/magnesium hydroxide and calcium carbonate on esophageal and gastric pH in subjects with heartburn. Am J Ther 1995; 2:546–552.
- Pettit M. Treatment of gastroesophageal reflux disease. Pharm World Sci 2005; 27:432–435.
- Fletcher J, Wirz A, Young J, Vallance R, McColl KE. Unbuffered highly acidic gastric juice exists at the gastroesophageal junction after a meal. Gastroenterology 2001; 121:775–783.
- Sauter M, Curcic J, Menne D, et al. Measuring the interaction of meal and gastric secretion: a combined quantitative magnetic resonance imaging and pharmacokinetic modeling approach. Neurogastroenterol Motil 2012; 24:632–638, e272–e273.
- Beaumont H, Bennink RJ, de Jong J, Boeckxstaens GE. The position of the acid pocket as a major risk factor for acidic reflux in healthy subjects and patients with GORD. Gut 2010; 59:441–451.
- Tytgat GN, Simoneau G. Clinical and laboratory studies of the antacid and raft-forming properties of Rennie alginate suspension. Aliment Pharmacol Ther 2006; 23:759–765.
- Chiu CT, Hsu CM, Wang CC, et al. Randomised clinical trial: sodium alginate oral suspension is non-inferior to omeprazole in the treatment of patients with non-erosive gastroesophageal disease. Aliment Pharmacol Ther 2013; 38:1054–1064.
- Rohof WO, Bennink RJ, Smout AJ, Thomas E, Boeckxstaens GE. An alginate-antacid formulation localizes to the acid pocket to reduce acid reflux in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2013; 11:1585–1591.
- Khoury RM, Katz PO, Castell DO. Post-prandial ranitidine is superior to post-prandial omeprazole in control of gastric acidity in healthy volunteers. Aliment Pharmacol Ther 1999; 13:1211–1214.
- Fackler WK, Ours TM, Vaezi MF, Richter JE. Long-term effect of H2RA therapy on nocturnal gastric acid breakthrough. Gastroenterology 2002; 122:625–632.
- Robinson M, Sahba B, Avner D, Jhala N, Greski-Rose PA, Jennings DE. A comparison of lansoprazole and ranitidine in the treatment of erosive oesophagitis. Multicentre Investigational Group. Aliment Pharmacol Ther 1995; 9:25–31.
- Vantrappen G, Rutgeerts L, Schurmans P, Coenegrachts JL. Omeprazole (40 mg) is superior to ranitidine in short-term treatment of ulcerative reflux esophagitis. Dig Dis Sci 1988; 33:523–529.
- Gralnek IM, Dulai GS, Fennerty MB, Spiegel BM. Esomeprazole versus other proton pump inhibitors in erosive esophagitis: a meta-analysis of randomized clinical trials. Clin Gastroenterol Hepatol 2006; 4:1452–1458.
- Targownik LE, Lix LM, Leung S, Leslie WD. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010; 138:896–904.
- Corley DA, Kubo A, Zhao W, Quesenberry C. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology 2010; 139:93–101.
- Katz PO, Gerson LB, Vela MF. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol 2013; 108:308–328.
- Giuliano C, Wilhelm SM, Kale-Pradhan PB. Are proton pump inhibitors associated with the development of community-acquired pneumonia? A meta-analysis. Expert Rev Clin Pharmacol 2012; 5:337–344.
- Hermos JA, Young MM, Fonda JR, Gagnon DR, Fiore LD, Lawler EV. Risk of community-acquired pneumonia in veteran patients to whom proton pump inhibitors were dispensed. Clin Infect Dis 2012; 54:33–42.
- Linsky A, Gupta K, Lawler EV, Fonda JR, Hermos JA. Proton pump inhibitors and risk for recurrent Clostridium difficile infection. Arch Intern Med 2010; 170:772–778.
- Bavishi C, Dupont HL. Systematic review: the use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment Pharmacol Ther 2011; 34:1269–1281.
- Bhatt DL, Cryer BL, Contant CF, et al; COGENT Investigators. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med 2010; 363:1909–1917.
- O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet 2009; 374:989–997.
- Hess MW, Hoenderop JG, Bindels RJ, Drenth JP. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther 2012; 36:405–413.
- Mittal RK, McCallum RW. Characteristics and frequency of transient relaxations of the lower esophageal sphincter in patients with reflux esophagitis. Gastroenterology 1988; 95:593–599.
- Lidums I, Lehmann A, Checklin H, Dent J, Holloway RH. Control of transient lower esophageal sphincter relaxations and reflux by the GABA(B) agonist baclofen in normal subjects. Gastroenterology 2000; 118:7–13.
- Cossentino MJ, Mann K, Armbruster SP, Lake JM, Maydonovitch C, Wong RK. Randomised clinical trial: the effect of baclofen in patients with gastro-oesophageal reflux—a randomised prospective study. Aliment Pharmacol Ther 2012; 35:1036–1044.
- Richter JE. Gastroesophageal reflux disease treatment: side effects and complications of fundoplication. Clin Gastroenterol Hepatol 2013; 11:465–471.
- Dickman R, Boaz M, Aizic S, Beniashvili Z, Fass R, Niv Y. Comparison of clinical characteristics of patients with gastroesophageal reflux disease who failed proton pump inhibitor therapy versus those who fully responded. J Neurogastroenterol Motil 2011; 17:387–394.
- Chan WW, Chiou E, Obstein KL, Tignor AS, Whitlock TL. The efficacy of proton pump inhibitors for the treatment of asthma in adults: a meta-analysis. Arch Intern Med 2011; 171:620–629.
- Chang AB, Lasserson TJ, Gaffney J, Connor FL, Garske LA. Gastro-oesophageal reflux treatment for prolonged non-specific cough in children and adults. Cochrane Database Syst Rev 2011; 1:CD004823.
- Qadeer MA, Phillips CO, Lopez AR, et al. Proton pump inhibitor therapy for suspected GERD-related chronic laryngitis: a meta-analysis of randomized controlled trials. Am J Gastroenterol 2006; 101:2646–2654.
- Gerson LB, Mitra S, Bleker WF, Yeung P. Control of intra-oesophageal pH in patients with Barrett's oesophagus on omeprazole-sodium bicarbonate therapy. Aliment Pharmacol Ther 2012; 35:803–809.
Gastroesophageal reflux disease (GERD) is a chronic and common medical problem, with up to 40% of the population experiencing its symptoms at least once per month.1 The condition develops when the reflux of stomach contents causes troublesome symptoms or complications.2
GERD symptoms can range from heartburn and regurgitation to cough and hoarseness. While many patients’ symptoms respond to medical treatment, the diagnosis and treatment in those whose symptoms do not respond to a proton pump inhibitor (PPI) may be challenging.
This article reviews the diagnosis and treatment options for GERD.
SYMPTOMS: TYPICAL, ATYPICAL, AND ALARM
Symptoms of GERD (Table 1) can be classified as typical (heartburn and regurgitation) or atypical (cough, asthma, hoarseness, chronic laryngitis, throat-clearing, chest pain, dyspepsia, and nausea). Atypical symptoms are more likely to be due to GERD if patients also have typical symptoms and if the symptoms respond to a trial of a PPI.3
Alarm symptoms. Keep in mind that extraesophageal presentations may be multifactorial, and it may be difficult to establish that reflux, even if present, is actually the cause. While chest pain may be due to GERD, it is important to rule out cardiac chest pain before considering GERD as a cause. Similarly, dysphagia along with typical or atypical symptoms warrants investigation for potential complications such as underlying motility disorder, esophageal stricture, esophageal ring, or malignancy.4 Other alarm symptoms include odynophagia, bleeding, weight loss, and anemia.
DIAGNOSING GERD: RESPONSE TO A PPI IS DIAGNOSTIC
Patients with typical symptoms that respond to PPI therapy need no further evaluation for a diagnosis of GERD to be made.5 On the other hand, further testing should be undertaken in patients with typical symptoms that do not respond to PPI therapy, in patients presenting with atypical symptoms, and in patients in whom antireflux surgery is being considered. Figure 1 shows our proposed algorithm.
Try a PPI for 6–8 weeks
Relief of heartburn and regurgitation after a 6- to 8-week course of a PPI strongly suggests GERD.6 However, a negative trial of a PPI does not rule out GERD, as this approach has been found to have a sensitivity of 78% and specificity of 54%.6
Despite this limitation, a trial of PPI therapy should be offered to patients presenting with typical symptoms and no alarm features. This approach has been found to be more cost-effective than proceeding directly to diagnostic testing.7
Endoscopy
Endoscopic findings in GERD can include erosive esophagitis, peptic stricture, and Barrett esophagus. Esophageal erosions are a highly specific sign of GERD; the Los Angeles classification system, a standardized scale for grading the severity of erosive esophagitis (from A to D, with D the most severe) provides an objective way to assess severity.8 However, most patients with heartburn and regurgitation do not have erosive disease, thus limiting the sensitivity of upper endoscopy as an initial diagnostic test in patients with suspected GERD.9
We recommend endoscopy for patients who present with alarm symptoms, patients with noncardiac chest pain, PPI nonresponders, and patients with chronic GERD symptoms and multiple risk factors for Barrett esophagus besides GERD, such as older age, male sex, white race, overweight, and smoking.10
Ambulatory pH and impedance monitoring
Ambulatory pH monitoring is the gold standard test for pathologic acid exposure in the esophagus. pH testing is indicated in PPI nonresponders, patients presenting with atypical symptoms, and before antireflux surgery.
In general, pH testing should be performed after the patient has been off PPI therapy for at least 7 days, as the test is highly unlikely to be abnormal while a patient is on a PPI.11 It is done either with a transnasal catheter for 24 hours, or with a wireless capsule (Bravo pH System, Given Imaging Ltd, Duluth, GA), which collects 48 to 96 hours of data. Studies of the wireless system have shown that its sensitivity increases 12% to 25% when it is performed for 48 hours compared with 24 hours.12,13
The pH test can be combined with impedance testing to evaluate for nonacid reflux.14 However, the clinical significance of nonacid reflux remains controversial, and for this reason the Esophageal Diagnostic Advisory Panel recommends that the decision to perform antireflux surgery should not be based on abnormal impedance testing.15
During pH and impedance testing, special software can calculate how closely the patient’s symptoms correlate with esophageal acid exposure. The symptom index (SI) and symptom association probability (SAP) are the symptom measurements most commonly used in practice. The SI measures the overall strength of the relationship, and an SI greater than 50% is considered positive.16 The SAP determines whether this relationship could have occurred by chance, and an SAP greater than 95% is statistically significant.17 In patients with normal levels of esophageal acid exposure, an elevated SI or SAP may indicate a component of esophageal hypersensitivity in symptom generation.
At our institution, we generally perform pH-only transnasal or wireless testing off PPI therapy to establish that the patient has pathologic acid exposure in the distal esophagus. Combined pH-impedance testing is typically reserved for patients with atypical symptoms unresponsive to PPI therapy and abnormal results on previous pH testing, which allows for correlation of nonacid reflux and symptoms.
Other tests
Esophageal manometry and barium esophagography have limited value in the primary diagnosis of GERD. However, they should be considered to rule out achalasia and other esophageal motility disorders in patients whose symptoms do not respond to PPIs. For this reason, esophageal manometry should be performed before considering antireflux surgery.
MANAGING GERD
Table 2 summarizes the various treatments for GERD.
Lifestyle modifications
Lifestyle modifications are the first-line therapy for GERD. Modifications that have been studied include weight loss, head-of-bed elevation, and avoidance of tobacco, alcohol, and late-night meals. Another modification that has been suggested is avoiding foods that can aggravate reflux symptoms—eg, caffeine, coffee, chocolate, spicy foods, highly acidic foods (oranges, tomatoes), and fatty foods. Of these, only weight loss and head-of-bed elevation have been proven effective.18
Three randomized controlled trials demonstrated that GERD symptoms and esophageal pH values improved with head-of-bed elevation using blocks or incline foam wedges.19–21 Several cohort studies demonstrated reduction in GERD symptoms with weight loss.22,23 Recently, a prospective cohort study also found that smoking cessation significantly improved GERD symptoms in patients with normal body mass index and severe symptoms.24
Antacids
Several antacids (eg, sodium bicarbonate, calcium carbonate, magnesium hydroxide, aluminum hydroxide) are available over the counter.
Antacids were thought to control heartburn symptoms by increasing the pH of gastric contents that might subsequently reflux into the esophagus. However, well-controlled studies have shown that they relieve heartburn by neutralizing acid in the esophagus, with no significant effect on gastric pH.25,26
Antacids provide rapid but short-lived relief from an existing episode of heartburn. Because they do not significantly raise the gastric pH, they do not prevent subsequent reflux episodes from repeatedly exposing the esophagus to gastric acid and causing heartburn. Additionally, antacids have not been shown to contribute to healing of erosive esophagitis.27 Hence, they may not be optimal for treating frequent reflux heartburn.
Sodium alginate
Gastric acid pockets are unbuffered pools of acid that float on top of ingested food.28 They develop as a result of poor mixing of newly secreted acid and food in the proximal stomach, which remains relatively quiescent after a meal compared with the distal stomach.29 In GERD, proximal extension of the acid pocket above the diaphragm increases the risk of acid reflux.30 The acid pocket is therefore an important source of postprandial acid in GERD and, as such, represents a unique therapeutic target.
Emerging evidence suggests that alginates may act directly on the acid pocket. Alginates are natural polysaccharide polymers that, on contact with gastric acid, precipitate within minutes into a low-density viscous gel of near-neutral pH. The change in pH triggers the sodium bicarbonate in the formulation to release carbon dioxide that becomes trapped in the alginate gel, causing it to float to the top of the gastric contents like a raft.31
A randomized controlled trial demonstrated that sodium alginate was as effective as omeprazole in relieving symptoms in patients with nonerosive reflux disease.32 Alginate has also been shown to provide more postprandial reflux relief than antacids.33
Histamine-2 receptor antagonists
Histamine-2 receptor antagonists act more swiftly and increase postprandial gastric pH more rapidly than PPIs, thus making them a good option for prophylaxis against postprandial GERD.34 Taking these drugs at bedtime may help in patients with objective nighttime reflux despite optimal PPI use. However, tachyphylaxis may occur as early as 1 week after starting combination therapy.35
Proton pump inhibitors
There are currently seven available PPIs, including four that can be obtained over the counter (omeprazole, lansoprazole, esomeprazole, and omeprazole-sodium bicarbonate) and three available only by prescription (rabeprazole, pantoprazole, and dexlansoprazole). Studies have shown than a standard 6- to 8-week course of a PPI provides complete symptom relief in 70% to 80% of patients with erosive reflux disease and in 60% of patients with nonerosive reflux disease.36,37 Clinically, PPIs all appear to be similar in their symptom relief.38
Most PPIs should be taken 30 to 60 minutes before meals. Exceptions are omeprazole-sodium bicarbonate and dexlansoprazole, which can be taken without regard to meals. At our institution, we usually start PPIs in a once-daily standard dose for 6 to 8 weeks and consider increasing to twice-daily dosing if symptoms do not respond completely. Patients with mild intermittent GERD symptoms may benefit from “on-demand” use of PPIs. This approach is best suited for patients with nonerosive reflux disease without evidence of Barrett esophagus on endoscopy.
Safety and adverse effects of PPIs
PPIs are generally safe but can cause adverse effects (Table 3).
Osteoporosis. In 2010, the US Food and Drug Administration issued warnings regarding the potential for wrist, hip, and spine fractures in PPI users.26 Most recent evidence suggests that PPIs may be associated with a small increase in risk of hip fractures in patients already at high risk.39,40 However, the 2013 American College of Gastroenterology (ACG) guidelines say that patients with known osteoporosis can remain on PPI therapy, and concern for hip fractures and osteoporosis should not affect the decision to use PPIs long-term except in patients with other risk factors for hip fracture.41
Community-acquired pneumonia. An increased risk of community-acquired pneumonia cannot be clearly documented in association with PPI therapy. Multiple studies, including randomized controlled trials, investigated this potential correlation. However, evidence suggests that short-term but not long-term PPI use may be associated with an overall increased risk of community-acquired pneumonia.42,43 Current guidelines suggest that in patients who need a PPI, the drug should not be withheld on the basis of a potential risk of community-acquired pneumonia.41
Clostridium difficile infection. In theory, PPIs may increase the risk of C difficile infection by increasing the ability of the spore to convert to the vegetative form and to survive intraluminally. In fact, studies and meta-analyses have suggested that PPIs do increase the risk of development and recurrence of C difficile infection.44,45 Therefore, PPIs should be used with care in patients who are at risk.41
Interaction with clopidogrel. The antiplatelet activity of clopidogrel requires activation by CYP2C19, the same pathway required for metabolism of some PPIs. Concern was raised about decreased antiplatelet activity of clopidogrel in the presence of PPIs. This was extensively studied, and there now appears to be no increased risk of adverse cardiovascular events in patients on PPIs, based on data from well-controlled randomized trials.46,47 A consensus panel of the American College of Cardiology Foundation, the American Heart Association, and the ACG said that PPIs may be used for appropriate indications in patients taking clopidogrel.47
Hypomagnesemia. By an unknown molecular mechanism, PPIs are thought to reduce intestinal magnesium absorption, leading to hypomagnesemia. A meta-analysis published in 2011 showed that PPI-induced hypomagnesemia is a drug-class effect and occurred after a median of 5.5 years of PPI use. Stopping the PPI resulted in magnesium recovery in 4 days, and rechallenge led to recurrence within 4 days.48
Hence, to avoid putting patients on long-term PPI therapy at risk, clinicians should anticipate this problem. Our practice is to check the magnesium level before starting a patient on long-term PPI therapy, and then to repeat the measurement every 1 to 2 years.
Baclofen
Transient lower esophageal sphincter relaxation has been shown to be a cause of reflux in healthy people and in patients with GERD.49
Baclofen, a muscle relaxant with selective gamma-aminobutyric acid receptor class B agonist properties, reduces transient lower esophageal sphincter relaxation in humans.50 In a well-designed, double-blind, randomized controlled trial, baclofen was associated with a significant decrease in upright reflux on 24-hour pH monitoring and significant improvement in belching and overall reflux symptoms.51 However, baclofen is not approved by the US Food and Drug Administration for the treatment of GERD, and its use may be limited by side effects such as somnolence and dizziness.
Antireflux surgery
Antireflux surgery is a reasonable option for selected patients with chronic GERD. The main types of surgery are laparoscopic fundoplication and, for obese patients, gastric bypass. Reasons to consider antireflux surgery include desire to stop PPI therapy, esophagitis not healed by PPIs, symptomatic hiatal hernia, and refractory reflux documented by pH testing.41
In general, surgical therapy may be considered in patients who respond to PPIs, but patients who do not respond to PPIs are less likely to respond to antireflux surgery.15 Other patients less likely to respond are those with symptoms of dyspepsia, such as nausea, vomiting, and epigastric pain.41
Common adverse effects of antireflux surgery include gas-bloat syndrome (up to 85% of patients), dysphagia (10% to 50% of patients), diarrhea (18% to 33% of patients), and recurrent heartburn (10% to 62% of patients).52
Endoscopic and minimally invasive antireflux procedures include endoscopic plication of the lower esophageal sphincter, radiofrequency augmentation of the lower esophageal sphincter, and sphincter augmentation by a string of titanium beads. While some have shown promise, they are not recommended by the most recent ACG guidelines, given lack of long-term data.41
REFRACTORY GERD
There is no consensus on the definition of refractory GERD. However, for the sake of simplicity, we can define it as persistence of suspected GERD symptoms despite treatment with a PPI. This may vary from a partial response to PPI therapy to a complete absence of response.
It is extremely important to rule out non-GERD causes of the ongoing symptoms, such as achalasia, gastroparesis, eosinophilic esophagitis, rumination, and aerophagia. PPI nonresponders are more likely to be obese, poorly compliant, and have extraesophageal symptoms.53–56 As previously discussed, PPIs should be taken 30 to 60 minutes before meals. For patients whose symptoms fail to respond to standard-dose daily PPI therapy, switching to another PPI or doubling the dose is common, although data to support this practice are limited. Of note, omeprazole-sodium bicarbonate has been shown to provide more symptom relief in nocturnal GERD.57 Additionally, adding a nighttime histamine-2 receptor antagonist may also help in patients with objective nighttime reflux.41
After noncompliance and suboptimal PPI dosing have been ruled out, PPI nonresponders with typical symptoms should undergo upper endoscopy and subsequent pH monitoring. Normal esophageal acid exposure on pH testing suggests functional heartburn or functional dyspepsia. Negative pH testing in a patient with atypical symptoms suggests a non-GERD cause of symptoms, and referral to an otolaryngologist, pulmonologist, or allergist is often warranted.
While antireflux surgery can be considered for patients with nonacid reflux on impedance testing, it should again be noted that GERD in patients with no response to PPIs is less likely to respond to antireflux surgery.15
TAKE-HOME POINTS
- GERD is a common medical condition, affecting up to 40% of US adults at least once monthly.
- GERD can result in a wide variety of symptoms, including typical heartburn and regurgitation as well as atypical symptoms such as cough.
- On the other hand, keep in mind that multiple non-GERD causes of heartburn and regurgitation may exist.
- Testing for GERD includes endoscopy and pH testing as well as functional testing such as esophageal manometry.
- While in most patients GERD will respond to lifestyle changes and antisecretory therapy such as a PPI, careful attention must be given to patients with symptoms refractory to PPI therapy.
- For a subset of patients, antireflux surgery may be a reasonable option, but care must be taken to exclude patients with a lower likelihood of responding to surgery.
Gastroesophageal reflux disease (GERD) is a chronic and common medical problem, with up to 40% of the population experiencing its symptoms at least once per month.1 The condition develops when the reflux of stomach contents causes troublesome symptoms or complications.2
GERD symptoms can range from heartburn and regurgitation to cough and hoarseness. While many patients’ symptoms respond to medical treatment, the diagnosis and treatment in those whose symptoms do not respond to a proton pump inhibitor (PPI) may be challenging.
This article reviews the diagnosis and treatment options for GERD.
SYMPTOMS: TYPICAL, ATYPICAL, AND ALARM
Symptoms of GERD (Table 1) can be classified as typical (heartburn and regurgitation) or atypical (cough, asthma, hoarseness, chronic laryngitis, throat-clearing, chest pain, dyspepsia, and nausea). Atypical symptoms are more likely to be due to GERD if patients also have typical symptoms and if the symptoms respond to a trial of a PPI.3
Alarm symptoms. Keep in mind that extraesophageal presentations may be multifactorial, and it may be difficult to establish that reflux, even if present, is actually the cause. While chest pain may be due to GERD, it is important to rule out cardiac chest pain before considering GERD as a cause. Similarly, dysphagia along with typical or atypical symptoms warrants investigation for potential complications such as underlying motility disorder, esophageal stricture, esophageal ring, or malignancy.4 Other alarm symptoms include odynophagia, bleeding, weight loss, and anemia.
DIAGNOSING GERD: RESPONSE TO A PPI IS DIAGNOSTIC
Patients with typical symptoms that respond to PPI therapy need no further evaluation for a diagnosis of GERD to be made.5 On the other hand, further testing should be undertaken in patients with typical symptoms that do not respond to PPI therapy, in patients presenting with atypical symptoms, and in patients in whom antireflux surgery is being considered. Figure 1 shows our proposed algorithm.
Try a PPI for 6–8 weeks
Relief of heartburn and regurgitation after a 6- to 8-week course of a PPI strongly suggests GERD.6 However, a negative trial of a PPI does not rule out GERD, as this approach has been found to have a sensitivity of 78% and specificity of 54%.6
Despite this limitation, a trial of PPI therapy should be offered to patients presenting with typical symptoms and no alarm features. This approach has been found to be more cost-effective than proceeding directly to diagnostic testing.7
Endoscopy
Endoscopic findings in GERD can include erosive esophagitis, peptic stricture, and Barrett esophagus. Esophageal erosions are a highly specific sign of GERD; the Los Angeles classification system, a standardized scale for grading the severity of erosive esophagitis (from A to D, with D the most severe) provides an objective way to assess severity.8 However, most patients with heartburn and regurgitation do not have erosive disease, thus limiting the sensitivity of upper endoscopy as an initial diagnostic test in patients with suspected GERD.9
We recommend endoscopy for patients who present with alarm symptoms, patients with noncardiac chest pain, PPI nonresponders, and patients with chronic GERD symptoms and multiple risk factors for Barrett esophagus besides GERD, such as older age, male sex, white race, overweight, and smoking.10
Ambulatory pH and impedance monitoring
Ambulatory pH monitoring is the gold standard test for pathologic acid exposure in the esophagus. pH testing is indicated in PPI nonresponders, patients presenting with atypical symptoms, and before antireflux surgery.
In general, pH testing should be performed after the patient has been off PPI therapy for at least 7 days, as the test is highly unlikely to be abnormal while a patient is on a PPI.11 It is done either with a transnasal catheter for 24 hours, or with a wireless capsule (Bravo pH System, Given Imaging Ltd, Duluth, GA), which collects 48 to 96 hours of data. Studies of the wireless system have shown that its sensitivity increases 12% to 25% when it is performed for 48 hours compared with 24 hours.12,13
The pH test can be combined with impedance testing to evaluate for nonacid reflux.14 However, the clinical significance of nonacid reflux remains controversial, and for this reason the Esophageal Diagnostic Advisory Panel recommends that the decision to perform antireflux surgery should not be based on abnormal impedance testing.15
During pH and impedance testing, special software can calculate how closely the patient’s symptoms correlate with esophageal acid exposure. The symptom index (SI) and symptom association probability (SAP) are the symptom measurements most commonly used in practice. The SI measures the overall strength of the relationship, and an SI greater than 50% is considered positive.16 The SAP determines whether this relationship could have occurred by chance, and an SAP greater than 95% is statistically significant.17 In patients with normal levels of esophageal acid exposure, an elevated SI or SAP may indicate a component of esophageal hypersensitivity in symptom generation.
At our institution, we generally perform pH-only transnasal or wireless testing off PPI therapy to establish that the patient has pathologic acid exposure in the distal esophagus. Combined pH-impedance testing is typically reserved for patients with atypical symptoms unresponsive to PPI therapy and abnormal results on previous pH testing, which allows for correlation of nonacid reflux and symptoms.
Other tests
Esophageal manometry and barium esophagography have limited value in the primary diagnosis of GERD. However, they should be considered to rule out achalasia and other esophageal motility disorders in patients whose symptoms do not respond to PPIs. For this reason, esophageal manometry should be performed before considering antireflux surgery.
MANAGING GERD
Table 2 summarizes the various treatments for GERD.
Lifestyle modifications
Lifestyle modifications are the first-line therapy for GERD. Modifications that have been studied include weight loss, head-of-bed elevation, and avoidance of tobacco, alcohol, and late-night meals. Another modification that has been suggested is avoiding foods that can aggravate reflux symptoms—eg, caffeine, coffee, chocolate, spicy foods, highly acidic foods (oranges, tomatoes), and fatty foods. Of these, only weight loss and head-of-bed elevation have been proven effective.18
Three randomized controlled trials demonstrated that GERD symptoms and esophageal pH values improved with head-of-bed elevation using blocks or incline foam wedges.19–21 Several cohort studies demonstrated reduction in GERD symptoms with weight loss.22,23 Recently, a prospective cohort study also found that smoking cessation significantly improved GERD symptoms in patients with normal body mass index and severe symptoms.24
Antacids
Several antacids (eg, sodium bicarbonate, calcium carbonate, magnesium hydroxide, aluminum hydroxide) are available over the counter.
Antacids were thought to control heartburn symptoms by increasing the pH of gastric contents that might subsequently reflux into the esophagus. However, well-controlled studies have shown that they relieve heartburn by neutralizing acid in the esophagus, with no significant effect on gastric pH.25,26
Antacids provide rapid but short-lived relief from an existing episode of heartburn. Because they do not significantly raise the gastric pH, they do not prevent subsequent reflux episodes from repeatedly exposing the esophagus to gastric acid and causing heartburn. Additionally, antacids have not been shown to contribute to healing of erosive esophagitis.27 Hence, they may not be optimal for treating frequent reflux heartburn.
Sodium alginate
Gastric acid pockets are unbuffered pools of acid that float on top of ingested food.28 They develop as a result of poor mixing of newly secreted acid and food in the proximal stomach, which remains relatively quiescent after a meal compared with the distal stomach.29 In GERD, proximal extension of the acid pocket above the diaphragm increases the risk of acid reflux.30 The acid pocket is therefore an important source of postprandial acid in GERD and, as such, represents a unique therapeutic target.
Emerging evidence suggests that alginates may act directly on the acid pocket. Alginates are natural polysaccharide polymers that, on contact with gastric acid, precipitate within minutes into a low-density viscous gel of near-neutral pH. The change in pH triggers the sodium bicarbonate in the formulation to release carbon dioxide that becomes trapped in the alginate gel, causing it to float to the top of the gastric contents like a raft.31
A randomized controlled trial demonstrated that sodium alginate was as effective as omeprazole in relieving symptoms in patients with nonerosive reflux disease.32 Alginate has also been shown to provide more postprandial reflux relief than antacids.33
Histamine-2 receptor antagonists
Histamine-2 receptor antagonists act more swiftly and increase postprandial gastric pH more rapidly than PPIs, thus making them a good option for prophylaxis against postprandial GERD.34 Taking these drugs at bedtime may help in patients with objective nighttime reflux despite optimal PPI use. However, tachyphylaxis may occur as early as 1 week after starting combination therapy.35
Proton pump inhibitors
There are currently seven available PPIs, including four that can be obtained over the counter (omeprazole, lansoprazole, esomeprazole, and omeprazole-sodium bicarbonate) and three available only by prescription (rabeprazole, pantoprazole, and dexlansoprazole). Studies have shown than a standard 6- to 8-week course of a PPI provides complete symptom relief in 70% to 80% of patients with erosive reflux disease and in 60% of patients with nonerosive reflux disease.36,37 Clinically, PPIs all appear to be similar in their symptom relief.38
Most PPIs should be taken 30 to 60 minutes before meals. Exceptions are omeprazole-sodium bicarbonate and dexlansoprazole, which can be taken without regard to meals. At our institution, we usually start PPIs in a once-daily standard dose for 6 to 8 weeks and consider increasing to twice-daily dosing if symptoms do not respond completely. Patients with mild intermittent GERD symptoms may benefit from “on-demand” use of PPIs. This approach is best suited for patients with nonerosive reflux disease without evidence of Barrett esophagus on endoscopy.
Safety and adverse effects of PPIs
PPIs are generally safe but can cause adverse effects (Table 3).
Osteoporosis. In 2010, the US Food and Drug Administration issued warnings regarding the potential for wrist, hip, and spine fractures in PPI users.26 Most recent evidence suggests that PPIs may be associated with a small increase in risk of hip fractures in patients already at high risk.39,40 However, the 2013 American College of Gastroenterology (ACG) guidelines say that patients with known osteoporosis can remain on PPI therapy, and concern for hip fractures and osteoporosis should not affect the decision to use PPIs long-term except in patients with other risk factors for hip fracture.41
Community-acquired pneumonia. An increased risk of community-acquired pneumonia cannot be clearly documented in association with PPI therapy. Multiple studies, including randomized controlled trials, investigated this potential correlation. However, evidence suggests that short-term but not long-term PPI use may be associated with an overall increased risk of community-acquired pneumonia.42,43 Current guidelines suggest that in patients who need a PPI, the drug should not be withheld on the basis of a potential risk of community-acquired pneumonia.41
Clostridium difficile infection. In theory, PPIs may increase the risk of C difficile infection by increasing the ability of the spore to convert to the vegetative form and to survive intraluminally. In fact, studies and meta-analyses have suggested that PPIs do increase the risk of development and recurrence of C difficile infection.44,45 Therefore, PPIs should be used with care in patients who are at risk.41
Interaction with clopidogrel. The antiplatelet activity of clopidogrel requires activation by CYP2C19, the same pathway required for metabolism of some PPIs. Concern was raised about decreased antiplatelet activity of clopidogrel in the presence of PPIs. This was extensively studied, and there now appears to be no increased risk of adverse cardiovascular events in patients on PPIs, based on data from well-controlled randomized trials.46,47 A consensus panel of the American College of Cardiology Foundation, the American Heart Association, and the ACG said that PPIs may be used for appropriate indications in patients taking clopidogrel.47
Hypomagnesemia. By an unknown molecular mechanism, PPIs are thought to reduce intestinal magnesium absorption, leading to hypomagnesemia. A meta-analysis published in 2011 showed that PPI-induced hypomagnesemia is a drug-class effect and occurred after a median of 5.5 years of PPI use. Stopping the PPI resulted in magnesium recovery in 4 days, and rechallenge led to recurrence within 4 days.48
Hence, to avoid putting patients on long-term PPI therapy at risk, clinicians should anticipate this problem. Our practice is to check the magnesium level before starting a patient on long-term PPI therapy, and then to repeat the measurement every 1 to 2 years.
Baclofen
Transient lower esophageal sphincter relaxation has been shown to be a cause of reflux in healthy people and in patients with GERD.49
Baclofen, a muscle relaxant with selective gamma-aminobutyric acid receptor class B agonist properties, reduces transient lower esophageal sphincter relaxation in humans.50 In a well-designed, double-blind, randomized controlled trial, baclofen was associated with a significant decrease in upright reflux on 24-hour pH monitoring and significant improvement in belching and overall reflux symptoms.51 However, baclofen is not approved by the US Food and Drug Administration for the treatment of GERD, and its use may be limited by side effects such as somnolence and dizziness.
Antireflux surgery
Antireflux surgery is a reasonable option for selected patients with chronic GERD. The main types of surgery are laparoscopic fundoplication and, for obese patients, gastric bypass. Reasons to consider antireflux surgery include desire to stop PPI therapy, esophagitis not healed by PPIs, symptomatic hiatal hernia, and refractory reflux documented by pH testing.41
In general, surgical therapy may be considered in patients who respond to PPIs, but patients who do not respond to PPIs are less likely to respond to antireflux surgery.15 Other patients less likely to respond are those with symptoms of dyspepsia, such as nausea, vomiting, and epigastric pain.41
Common adverse effects of antireflux surgery include gas-bloat syndrome (up to 85% of patients), dysphagia (10% to 50% of patients), diarrhea (18% to 33% of patients), and recurrent heartburn (10% to 62% of patients).52
Endoscopic and minimally invasive antireflux procedures include endoscopic plication of the lower esophageal sphincter, radiofrequency augmentation of the lower esophageal sphincter, and sphincter augmentation by a string of titanium beads. While some have shown promise, they are not recommended by the most recent ACG guidelines, given lack of long-term data.41
REFRACTORY GERD
There is no consensus on the definition of refractory GERD. However, for the sake of simplicity, we can define it as persistence of suspected GERD symptoms despite treatment with a PPI. This may vary from a partial response to PPI therapy to a complete absence of response.
It is extremely important to rule out non-GERD causes of the ongoing symptoms, such as achalasia, gastroparesis, eosinophilic esophagitis, rumination, and aerophagia. PPI nonresponders are more likely to be obese, poorly compliant, and have extraesophageal symptoms.53–56 As previously discussed, PPIs should be taken 30 to 60 minutes before meals. For patients whose symptoms fail to respond to standard-dose daily PPI therapy, switching to another PPI or doubling the dose is common, although data to support this practice are limited. Of note, omeprazole-sodium bicarbonate has been shown to provide more symptom relief in nocturnal GERD.57 Additionally, adding a nighttime histamine-2 receptor antagonist may also help in patients with objective nighttime reflux.41
After noncompliance and suboptimal PPI dosing have been ruled out, PPI nonresponders with typical symptoms should undergo upper endoscopy and subsequent pH monitoring. Normal esophageal acid exposure on pH testing suggests functional heartburn or functional dyspepsia. Negative pH testing in a patient with atypical symptoms suggests a non-GERD cause of symptoms, and referral to an otolaryngologist, pulmonologist, or allergist is often warranted.
While antireflux surgery can be considered for patients with nonacid reflux on impedance testing, it should again be noted that GERD in patients with no response to PPIs is less likely to respond to antireflux surgery.15
TAKE-HOME POINTS
- GERD is a common medical condition, affecting up to 40% of US adults at least once monthly.
- GERD can result in a wide variety of symptoms, including typical heartburn and regurgitation as well as atypical symptoms such as cough.
- On the other hand, keep in mind that multiple non-GERD causes of heartburn and regurgitation may exist.
- Testing for GERD includes endoscopy and pH testing as well as functional testing such as esophageal manometry.
- While in most patients GERD will respond to lifestyle changes and antisecretory therapy such as a PPI, careful attention must be given to patients with symptoms refractory to PPI therapy.
- For a subset of patients, antireflux surgery may be a reasonable option, but care must be taken to exclude patients with a lower likelihood of responding to surgery.
- Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology 1997; 112:1448–1456.
- Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R; Globale Konsensusgrupp. [The Montreal definition and classification of gastroesophageal reflux disease: a global, evidence-based consensus paper]. Z Gastroenterol 2007; 45:1125–1240. In German.
- Gerson LB, Kahrilas PJ, Fass R. Insights into gastroesophageal reflux disease-associated dyspeptic symptoms. Clin Gastroenterol Hepatol 2011; 9:824–833.
- Vakil NB, Traxler B, Levine D. Dysphagia in patients with erosive esophagitis: prevalence, severity, and response to proton pump inhibitor treatment. Clin Gastroenterol Hepatol 2004; 2:665–668.
- Kahrilas PJ, Shaheen NJ, aezi MF, et al; American Gastroenterological Association. American Gastroenterological Association Medical Position Statement on the management of gastroesophageal reflux disease. Gastroenterology 2008; 135:1383–1391 e1–5.
- Numans ME, Lau J, de Wit NJ, Bonis PA. Short-term treatment with proton-pump inhibitors as a test for gastroesophageal reflux disease: a meta-analysis of diagnostic test characteristics. Ann Intern Med 2004; 140:518–527.
- Fass R. Empirical trials in treatment of gastroesophageal reflux disease. Dig Dis 2000; 18:20–26.
- Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 1999; 45:172–180.
- Johansson KE, Ask P, Boeryd B, Fransson SG, Tibbling L. Oesophagitis, signs of reflux, and gastric acid secretion in patients with symptoms of gastro-oesophageal reflux disease. Scand J Gastroenterol 1986; 21:837–847.
- Becher A, Dent J. Systematic review: ageing and gastro-oesophageal reflux disease symptoms, oesophageal function and reflux oesophagitis. Aliment Pharmacol Ther 2011; 33:442–454.
- Charbel S, Khandwala F, Vaezi MF. The role of esophageal pH monitoring in symptomatic patients on PPI therapy. Am J Gastroenterol 2005; 100:283–289.
- Pandolfino JE, Richter JE, Ours T, Guardino JM, Chapman J, Kahrilas PJ. Ambulatory esophageal pH monitoring using a wireless system. Am J Gastroenterol 2003; 98:740–749.
- Prakash C, Clouse RE. Value of extended recording time with wireless pH monitoring in evaluating gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2005; 3:329–334.
- Hirano I, Richter JE; Practice Parameters Committee of the American College of Gastroenterology. ACG practice guidelines: esophageal reflux testing. Am J Gastroenterol 2007; 102:668–685.
- Jobe BA, Richter JE, Hoppo T, et al. Preoperative diagnostic workup before antireflux surgery: an evidence and experience-based consensus of the Esophageal Diagnostic Advisory Panel. J Am Coll Surg 2013; 217:586–597.
- Singh S, Richter JE, Bradley LA, Haile JM. The symptom index. Differential usefulness in suspected acid-related complaints of heartburn and chest pain. Dig Dis Sci 1993; 38:1402–1408.
- Weusten BL, Roelofs JM, Akkermans LM, Van Berge-Henegouwen GP, Smout AJ. The symptom-association probability: an improved method for symptom analysis of 24-hour esophageal pH data. Gastroenterology 1994; 107:1741–1745.
- Kaltenbach T, Crockett S, Gerson LB. Are lifestyle measures effective in patients with gastroesophageal reflux disease? An evidence-based approach. Arch Intern Med 2006; 166:965–971.
- Hamilton JW, Boisen RJ, Yamamoto DT, Wagner JL, Reichelderfer M. Sleeping on a wedge diminishes exposure of the esophagus to refluxed acid. Dig Dis Sci 1988; 33:518–522.
- Pollmann H, Zillessen E, Pohl J, et al. [Effect of elevated head position in bed in therapy of gastroesophageal reflux]. Z Gastroenterol 1996; 34(suppl 2):93–99. In German.
- Stanciu C, Bennett JR. Effects of posture on gastro-oesophageal reflux. Digestion 1977; 15:104–109.
- Fraser-Moodie CA, Norton B, Gornall C, Magnago S, Weale AR, Holmes GK. Weight loss has an independent beneficial effect on symptoms of gastro-oesophageal reflux in patients who are overweight. Scand J Gastroenterol 1999; 34:337–340.
- Mathus-Vliegen LM, Tytgat GN. Twenty-four-hour pH measurements in morbid obesity: effects of massive overweight, weight loss and gastric distension. Eur J Gastroenterol Hepatol 1996; 8:635–640.
- Ness-Jensen E, Lindam A, Lagergren J, Hveem K. Tobacco smoking cessation and improved gastroesophageal reflux: a prospective population-based cohort study: the HUNT study. Am J Gastroenterol 2014; 109:171–177.
- Collings KL, Rodriguez-Stanley S, Proskin HM, Robinson M, Miner PB Jr. Clinical effectiveness of a new antacid chewing gum on heartburn and oesophageal pH control. Aliment Pharmacol Ther 2002; 16:2029–2035.
- Decktor DL, Robinson M, Maton PN, Lanza FL, Gottlieb S. Effects of aluminum/magnesium hydroxide and calcium carbonate on esophageal and gastric pH in subjects with heartburn. Am J Ther 1995; 2:546–552.
- Pettit M. Treatment of gastroesophageal reflux disease. Pharm World Sci 2005; 27:432–435.
- Fletcher J, Wirz A, Young J, Vallance R, McColl KE. Unbuffered highly acidic gastric juice exists at the gastroesophageal junction after a meal. Gastroenterology 2001; 121:775–783.
- Sauter M, Curcic J, Menne D, et al. Measuring the interaction of meal and gastric secretion: a combined quantitative magnetic resonance imaging and pharmacokinetic modeling approach. Neurogastroenterol Motil 2012; 24:632–638, e272–e273.
- Beaumont H, Bennink RJ, de Jong J, Boeckxstaens GE. The position of the acid pocket as a major risk factor for acidic reflux in healthy subjects and patients with GORD. Gut 2010; 59:441–451.
- Tytgat GN, Simoneau G. Clinical and laboratory studies of the antacid and raft-forming properties of Rennie alginate suspension. Aliment Pharmacol Ther 2006; 23:759–765.
- Chiu CT, Hsu CM, Wang CC, et al. Randomised clinical trial: sodium alginate oral suspension is non-inferior to omeprazole in the treatment of patients with non-erosive gastroesophageal disease. Aliment Pharmacol Ther 2013; 38:1054–1064.
- Rohof WO, Bennink RJ, Smout AJ, Thomas E, Boeckxstaens GE. An alginate-antacid formulation localizes to the acid pocket to reduce acid reflux in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2013; 11:1585–1591.
- Khoury RM, Katz PO, Castell DO. Post-prandial ranitidine is superior to post-prandial omeprazole in control of gastric acidity in healthy volunteers. Aliment Pharmacol Ther 1999; 13:1211–1214.
- Fackler WK, Ours TM, Vaezi MF, Richter JE. Long-term effect of H2RA therapy on nocturnal gastric acid breakthrough. Gastroenterology 2002; 122:625–632.
- Robinson M, Sahba B, Avner D, Jhala N, Greski-Rose PA, Jennings DE. A comparison of lansoprazole and ranitidine in the treatment of erosive oesophagitis. Multicentre Investigational Group. Aliment Pharmacol Ther 1995; 9:25–31.
- Vantrappen G, Rutgeerts L, Schurmans P, Coenegrachts JL. Omeprazole (40 mg) is superior to ranitidine in short-term treatment of ulcerative reflux esophagitis. Dig Dis Sci 1988; 33:523–529.
- Gralnek IM, Dulai GS, Fennerty MB, Spiegel BM. Esomeprazole versus other proton pump inhibitors in erosive esophagitis: a meta-analysis of randomized clinical trials. Clin Gastroenterol Hepatol 2006; 4:1452–1458.
- Targownik LE, Lix LM, Leung S, Leslie WD. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010; 138:896–904.
- Corley DA, Kubo A, Zhao W, Quesenberry C. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology 2010; 139:93–101.
- Katz PO, Gerson LB, Vela MF. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol 2013; 108:308–328.
- Giuliano C, Wilhelm SM, Kale-Pradhan PB. Are proton pump inhibitors associated with the development of community-acquired pneumonia? A meta-analysis. Expert Rev Clin Pharmacol 2012; 5:337–344.
- Hermos JA, Young MM, Fonda JR, Gagnon DR, Fiore LD, Lawler EV. Risk of community-acquired pneumonia in veteran patients to whom proton pump inhibitors were dispensed. Clin Infect Dis 2012; 54:33–42.
- Linsky A, Gupta K, Lawler EV, Fonda JR, Hermos JA. Proton pump inhibitors and risk for recurrent Clostridium difficile infection. Arch Intern Med 2010; 170:772–778.
- Bavishi C, Dupont HL. Systematic review: the use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment Pharmacol Ther 2011; 34:1269–1281.
- Bhatt DL, Cryer BL, Contant CF, et al; COGENT Investigators. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med 2010; 363:1909–1917.
- O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet 2009; 374:989–997.
- Hess MW, Hoenderop JG, Bindels RJ, Drenth JP. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther 2012; 36:405–413.
- Mittal RK, McCallum RW. Characteristics and frequency of transient relaxations of the lower esophageal sphincter in patients with reflux esophagitis. Gastroenterology 1988; 95:593–599.
- Lidums I, Lehmann A, Checklin H, Dent J, Holloway RH. Control of transient lower esophageal sphincter relaxations and reflux by the GABA(B) agonist baclofen in normal subjects. Gastroenterology 2000; 118:7–13.
- Cossentino MJ, Mann K, Armbruster SP, Lake JM, Maydonovitch C, Wong RK. Randomised clinical trial: the effect of baclofen in patients with gastro-oesophageal reflux—a randomised prospective study. Aliment Pharmacol Ther 2012; 35:1036–1044.
- Richter JE. Gastroesophageal reflux disease treatment: side effects and complications of fundoplication. Clin Gastroenterol Hepatol 2013; 11:465–471.
- Dickman R, Boaz M, Aizic S, Beniashvili Z, Fass R, Niv Y. Comparison of clinical characteristics of patients with gastroesophageal reflux disease who failed proton pump inhibitor therapy versus those who fully responded. J Neurogastroenterol Motil 2011; 17:387–394.
- Chan WW, Chiou E, Obstein KL, Tignor AS, Whitlock TL. The efficacy of proton pump inhibitors for the treatment of asthma in adults: a meta-analysis. Arch Intern Med 2011; 171:620–629.
- Chang AB, Lasserson TJ, Gaffney J, Connor FL, Garske LA. Gastro-oesophageal reflux treatment for prolonged non-specific cough in children and adults. Cochrane Database Syst Rev 2011; 1:CD004823.
- Qadeer MA, Phillips CO, Lopez AR, et al. Proton pump inhibitor therapy for suspected GERD-related chronic laryngitis: a meta-analysis of randomized controlled trials. Am J Gastroenterol 2006; 101:2646–2654.
- Gerson LB, Mitra S, Bleker WF, Yeung P. Control of intra-oesophageal pH in patients with Barrett's oesophagus on omeprazole-sodium bicarbonate therapy. Aliment Pharmacol Ther 2012; 35:803–809.
- Locke GR 3rd, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ 3rd. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology 1997; 112:1448–1456.
- Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R; Globale Konsensusgrupp. [The Montreal definition and classification of gastroesophageal reflux disease: a global, evidence-based consensus paper]. Z Gastroenterol 2007; 45:1125–1240. In German.
- Gerson LB, Kahrilas PJ, Fass R. Insights into gastroesophageal reflux disease-associated dyspeptic symptoms. Clin Gastroenterol Hepatol 2011; 9:824–833.
- Vakil NB, Traxler B, Levine D. Dysphagia in patients with erosive esophagitis: prevalence, severity, and response to proton pump inhibitor treatment. Clin Gastroenterol Hepatol 2004; 2:665–668.
- Kahrilas PJ, Shaheen NJ, aezi MF, et al; American Gastroenterological Association. American Gastroenterological Association Medical Position Statement on the management of gastroesophageal reflux disease. Gastroenterology 2008; 135:1383–1391 e1–5.
- Numans ME, Lau J, de Wit NJ, Bonis PA. Short-term treatment with proton-pump inhibitors as a test for gastroesophageal reflux disease: a meta-analysis of diagnostic test characteristics. Ann Intern Med 2004; 140:518–527.
- Fass R. Empirical trials in treatment of gastroesophageal reflux disease. Dig Dis 2000; 18:20–26.
- Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 1999; 45:172–180.
- Johansson KE, Ask P, Boeryd B, Fransson SG, Tibbling L. Oesophagitis, signs of reflux, and gastric acid secretion in patients with symptoms of gastro-oesophageal reflux disease. Scand J Gastroenterol 1986; 21:837–847.
- Becher A, Dent J. Systematic review: ageing and gastro-oesophageal reflux disease symptoms, oesophageal function and reflux oesophagitis. Aliment Pharmacol Ther 2011; 33:442–454.
- Charbel S, Khandwala F, Vaezi MF. The role of esophageal pH monitoring in symptomatic patients on PPI therapy. Am J Gastroenterol 2005; 100:283–289.
- Pandolfino JE, Richter JE, Ours T, Guardino JM, Chapman J, Kahrilas PJ. Ambulatory esophageal pH monitoring using a wireless system. Am J Gastroenterol 2003; 98:740–749.
- Prakash C, Clouse RE. Value of extended recording time with wireless pH monitoring in evaluating gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2005; 3:329–334.
- Hirano I, Richter JE; Practice Parameters Committee of the American College of Gastroenterology. ACG practice guidelines: esophageal reflux testing. Am J Gastroenterol 2007; 102:668–685.
- Jobe BA, Richter JE, Hoppo T, et al. Preoperative diagnostic workup before antireflux surgery: an evidence and experience-based consensus of the Esophageal Diagnostic Advisory Panel. J Am Coll Surg 2013; 217:586–597.
- Singh S, Richter JE, Bradley LA, Haile JM. The symptom index. Differential usefulness in suspected acid-related complaints of heartburn and chest pain. Dig Dis Sci 1993; 38:1402–1408.
- Weusten BL, Roelofs JM, Akkermans LM, Van Berge-Henegouwen GP, Smout AJ. The symptom-association probability: an improved method for symptom analysis of 24-hour esophageal pH data. Gastroenterology 1994; 107:1741–1745.
- Kaltenbach T, Crockett S, Gerson LB. Are lifestyle measures effective in patients with gastroesophageal reflux disease? An evidence-based approach. Arch Intern Med 2006; 166:965–971.
- Hamilton JW, Boisen RJ, Yamamoto DT, Wagner JL, Reichelderfer M. Sleeping on a wedge diminishes exposure of the esophagus to refluxed acid. Dig Dis Sci 1988; 33:518–522.
- Pollmann H, Zillessen E, Pohl J, et al. [Effect of elevated head position in bed in therapy of gastroesophageal reflux]. Z Gastroenterol 1996; 34(suppl 2):93–99. In German.
- Stanciu C, Bennett JR. Effects of posture on gastro-oesophageal reflux. Digestion 1977; 15:104–109.
- Fraser-Moodie CA, Norton B, Gornall C, Magnago S, Weale AR, Holmes GK. Weight loss has an independent beneficial effect on symptoms of gastro-oesophageal reflux in patients who are overweight. Scand J Gastroenterol 1999; 34:337–340.
- Mathus-Vliegen LM, Tytgat GN. Twenty-four-hour pH measurements in morbid obesity: effects of massive overweight, weight loss and gastric distension. Eur J Gastroenterol Hepatol 1996; 8:635–640.
- Ness-Jensen E, Lindam A, Lagergren J, Hveem K. Tobacco smoking cessation and improved gastroesophageal reflux: a prospective population-based cohort study: the HUNT study. Am J Gastroenterol 2014; 109:171–177.
- Collings KL, Rodriguez-Stanley S, Proskin HM, Robinson M, Miner PB Jr. Clinical effectiveness of a new antacid chewing gum on heartburn and oesophageal pH control. Aliment Pharmacol Ther 2002; 16:2029–2035.
- Decktor DL, Robinson M, Maton PN, Lanza FL, Gottlieb S. Effects of aluminum/magnesium hydroxide and calcium carbonate on esophageal and gastric pH in subjects with heartburn. Am J Ther 1995; 2:546–552.
- Pettit M. Treatment of gastroesophageal reflux disease. Pharm World Sci 2005; 27:432–435.
- Fletcher J, Wirz A, Young J, Vallance R, McColl KE. Unbuffered highly acidic gastric juice exists at the gastroesophageal junction after a meal. Gastroenterology 2001; 121:775–783.
- Sauter M, Curcic J, Menne D, et al. Measuring the interaction of meal and gastric secretion: a combined quantitative magnetic resonance imaging and pharmacokinetic modeling approach. Neurogastroenterol Motil 2012; 24:632–638, e272–e273.
- Beaumont H, Bennink RJ, de Jong J, Boeckxstaens GE. The position of the acid pocket as a major risk factor for acidic reflux in healthy subjects and patients with GORD. Gut 2010; 59:441–451.
- Tytgat GN, Simoneau G. Clinical and laboratory studies of the antacid and raft-forming properties of Rennie alginate suspension. Aliment Pharmacol Ther 2006; 23:759–765.
- Chiu CT, Hsu CM, Wang CC, et al. Randomised clinical trial: sodium alginate oral suspension is non-inferior to omeprazole in the treatment of patients with non-erosive gastroesophageal disease. Aliment Pharmacol Ther 2013; 38:1054–1064.
- Rohof WO, Bennink RJ, Smout AJ, Thomas E, Boeckxstaens GE. An alginate-antacid formulation localizes to the acid pocket to reduce acid reflux in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol 2013; 11:1585–1591.
- Khoury RM, Katz PO, Castell DO. Post-prandial ranitidine is superior to post-prandial omeprazole in control of gastric acidity in healthy volunteers. Aliment Pharmacol Ther 1999; 13:1211–1214.
- Fackler WK, Ours TM, Vaezi MF, Richter JE. Long-term effect of H2RA therapy on nocturnal gastric acid breakthrough. Gastroenterology 2002; 122:625–632.
- Robinson M, Sahba B, Avner D, Jhala N, Greski-Rose PA, Jennings DE. A comparison of lansoprazole and ranitidine in the treatment of erosive oesophagitis. Multicentre Investigational Group. Aliment Pharmacol Ther 1995; 9:25–31.
- Vantrappen G, Rutgeerts L, Schurmans P, Coenegrachts JL. Omeprazole (40 mg) is superior to ranitidine in short-term treatment of ulcerative reflux esophagitis. Dig Dis Sci 1988; 33:523–529.
- Gralnek IM, Dulai GS, Fennerty MB, Spiegel BM. Esomeprazole versus other proton pump inhibitors in erosive esophagitis: a meta-analysis of randomized clinical trials. Clin Gastroenterol Hepatol 2006; 4:1452–1458.
- Targownik LE, Lix LM, Leung S, Leslie WD. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010; 138:896–904.
- Corley DA, Kubo A, Zhao W, Quesenberry C. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology 2010; 139:93–101.
- Katz PO, Gerson LB, Vela MF. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol 2013; 108:308–328.
- Giuliano C, Wilhelm SM, Kale-Pradhan PB. Are proton pump inhibitors associated with the development of community-acquired pneumonia? A meta-analysis. Expert Rev Clin Pharmacol 2012; 5:337–344.
- Hermos JA, Young MM, Fonda JR, Gagnon DR, Fiore LD, Lawler EV. Risk of community-acquired pneumonia in veteran patients to whom proton pump inhibitors were dispensed. Clin Infect Dis 2012; 54:33–42.
- Linsky A, Gupta K, Lawler EV, Fonda JR, Hermos JA. Proton pump inhibitors and risk for recurrent Clostridium difficile infection. Arch Intern Med 2010; 170:772–778.
- Bavishi C, Dupont HL. Systematic review: the use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment Pharmacol Ther 2011; 34:1269–1281.
- Bhatt DL, Cryer BL, Contant CF, et al; COGENT Investigators. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med 2010; 363:1909–1917.
- O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet 2009; 374:989–997.
- Hess MW, Hoenderop JG, Bindels RJ, Drenth JP. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther 2012; 36:405–413.
- Mittal RK, McCallum RW. Characteristics and frequency of transient relaxations of the lower esophageal sphincter in patients with reflux esophagitis. Gastroenterology 1988; 95:593–599.
- Lidums I, Lehmann A, Checklin H, Dent J, Holloway RH. Control of transient lower esophageal sphincter relaxations and reflux by the GABA(B) agonist baclofen in normal subjects. Gastroenterology 2000; 118:7–13.
- Cossentino MJ, Mann K, Armbruster SP, Lake JM, Maydonovitch C, Wong RK. Randomised clinical trial: the effect of baclofen in patients with gastro-oesophageal reflux—a randomised prospective study. Aliment Pharmacol Ther 2012; 35:1036–1044.
- Richter JE. Gastroesophageal reflux disease treatment: side effects and complications of fundoplication. Clin Gastroenterol Hepatol 2013; 11:465–471.
- Dickman R, Boaz M, Aizic S, Beniashvili Z, Fass R, Niv Y. Comparison of clinical characteristics of patients with gastroesophageal reflux disease who failed proton pump inhibitor therapy versus those who fully responded. J Neurogastroenterol Motil 2011; 17:387–394.
- Chan WW, Chiou E, Obstein KL, Tignor AS, Whitlock TL. The efficacy of proton pump inhibitors for the treatment of asthma in adults: a meta-analysis. Arch Intern Med 2011; 171:620–629.
- Chang AB, Lasserson TJ, Gaffney J, Connor FL, Garske LA. Gastro-oesophageal reflux treatment for prolonged non-specific cough in children and adults. Cochrane Database Syst Rev 2011; 1:CD004823.
- Qadeer MA, Phillips CO, Lopez AR, et al. Proton pump inhibitor therapy for suspected GERD-related chronic laryngitis: a meta-analysis of randomized controlled trials. Am J Gastroenterol 2006; 101:2646–2654.
- Gerson LB, Mitra S, Bleker WF, Yeung P. Control of intra-oesophageal pH in patients with Barrett's oesophagus on omeprazole-sodium bicarbonate therapy. Aliment Pharmacol Ther 2012; 35:803–809.
KEY POINTS
- GERD symptoms may be typical (eg, heartburn, regurgitation) or atypical (eg, cough, chest pain, hoarseness).
- In patients with typical symptoms, a 6- to 8-week trial of a PPI is a reasonable and cost-effective approach to diagnosing GERD.
- Endoscopy is indicated for patients who have alarm symptoms such as dysphagia, weight loss, and bleeding; it is unnecessary in patients who have typical GERD symptoms.
- Ambulatory pH monitoring should be used in patients whose symptoms do not respond to a PPI and those in whom antireflux surgery is being considered.
- Weight loss and head-of-bed elevation are the only lifestyle interventions that have been proven effective for GERD.
- While risks of PPI use are rare, they should be discussed with patients on long-term therapy.
- Symptoms that do not respond to a PPI are less likely to improve with antireflux surgery.