User login
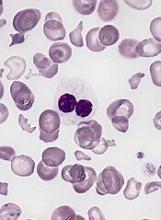
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.